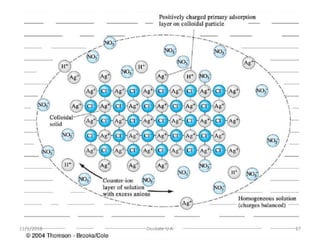
Gravimetric analysis determines the amount of analyte by measuring the mass of a pure precipitate containing the analyte. There are two main types - precipitation and volatilization. For a successful analysis, the precipitate must be completely precipitated, of known composition, pure, and easily filtered. Factors like particle size, purity, and co-precipitation must be considered. The general steps involve preparing a solution, precipitating, filtering, washing, drying, and weighing the precipitate.