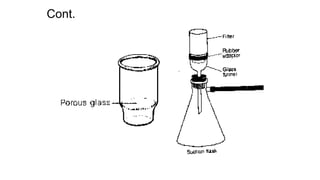
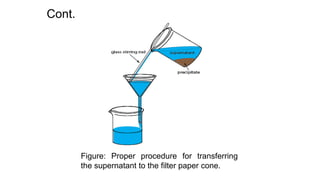
This document discusses gravimetric analysis, which is a quantitative analytical technique where the amount of analyte is determined by measuring its mass. There are several types of gravimetric methods including precipitation gravimetry, where the analyte is separated as a precipitate and weighed. An ideal precipitate would be easily filtered, insoluble, and of known composition when dried or ignited. The document outlines the general procedure for precipitation gravimetry and factors that influence precipitate formation and particle size such as supersaturation and coagulation of colloids.




































![Indicators for Argentometric titrations
Iron(III) ion (The Volhard Method)
Silver ions are titrated with a standard solution of thio-cyanate ion:
Ag+ + SCN- ↔ AgSCN (s)
• Iron(III) serves as the indicator
• Solution turns red with the first slight excess of thio-cyanate ion:
Fe3+ + SCN- ↔ FeSCN2+
• Titration must be carried out in acidic solution to prevent precipitation of Fe(III)
as the hydrated oxide
• [Indicator] is not critical and can be between 0.002M – 1.6M](https://image.slidesharecdn.com/nche211unit3gravimetry-240326124404-69580799/85/NCHE-211-UNIT3-GRAVIMETRY-UIVERSITYY-pdf-41-320.jpg)


