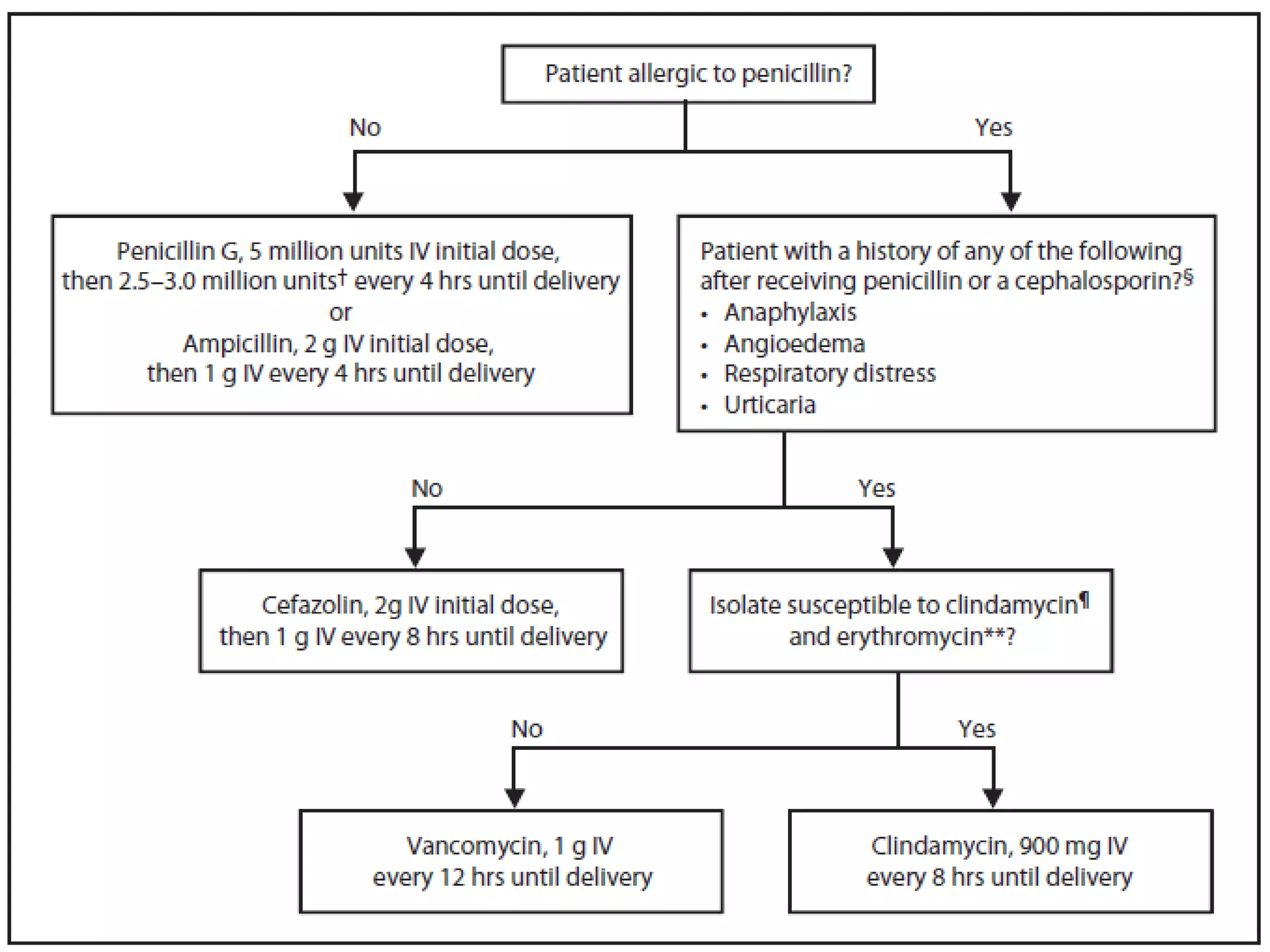
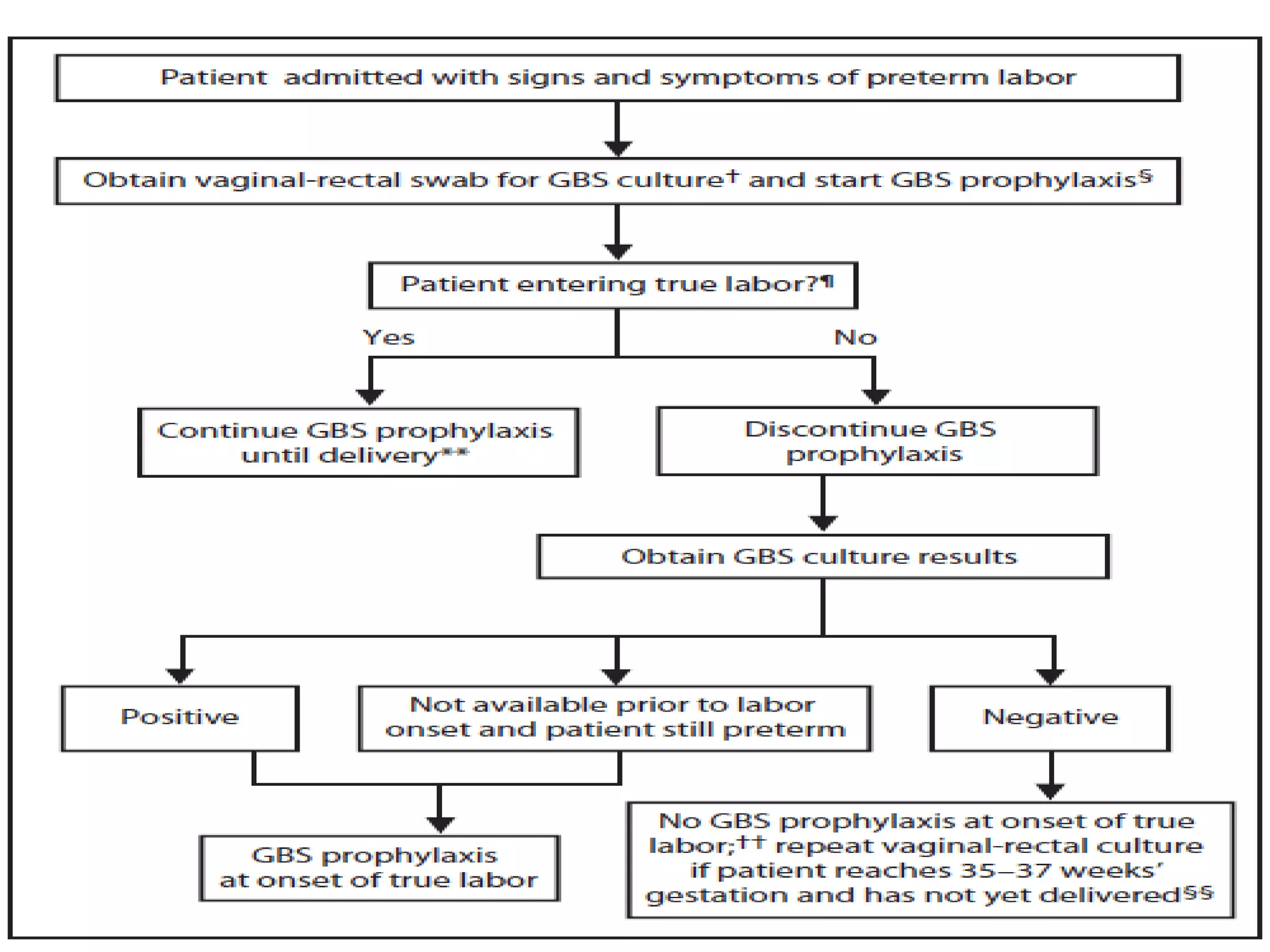
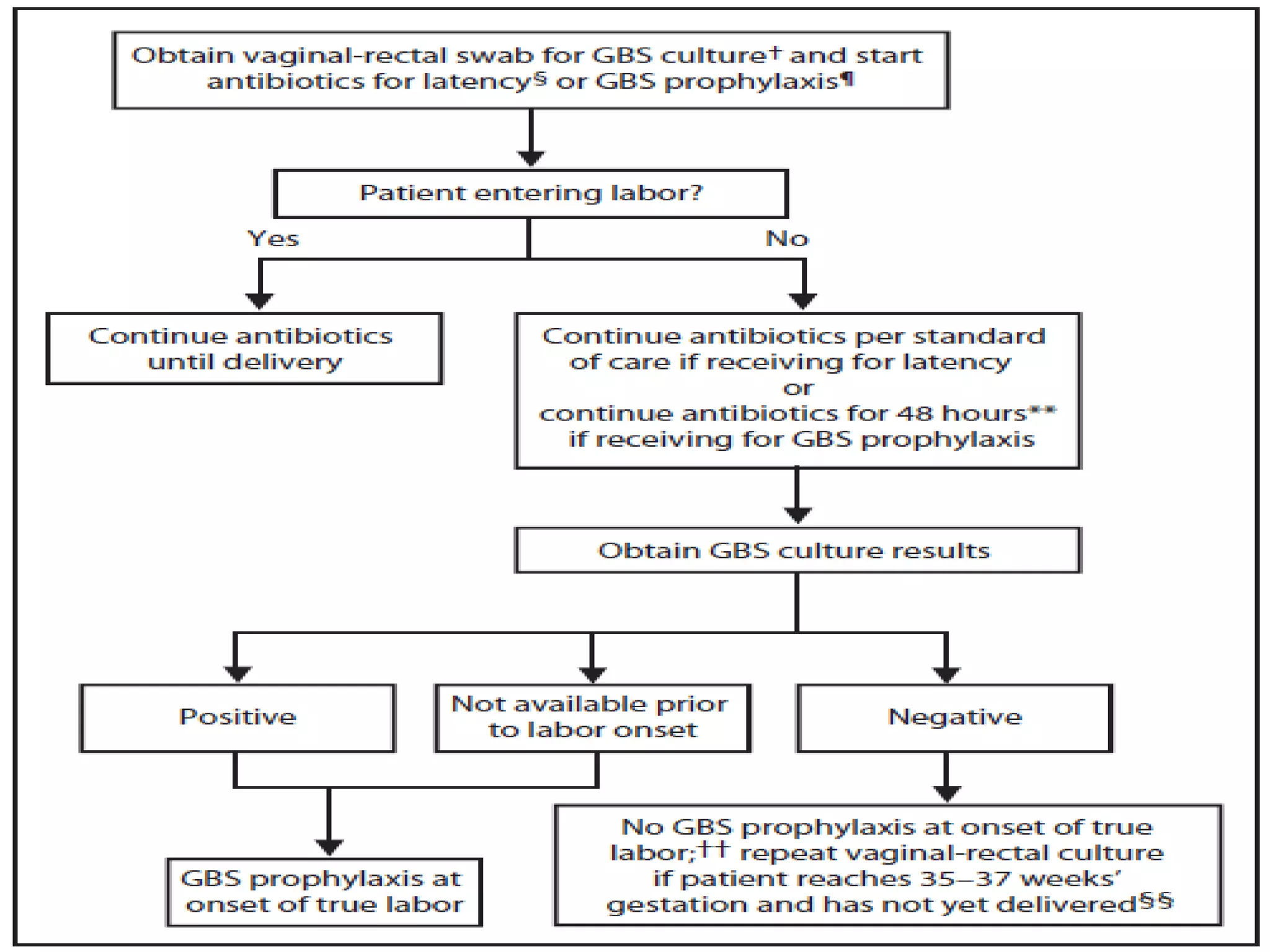
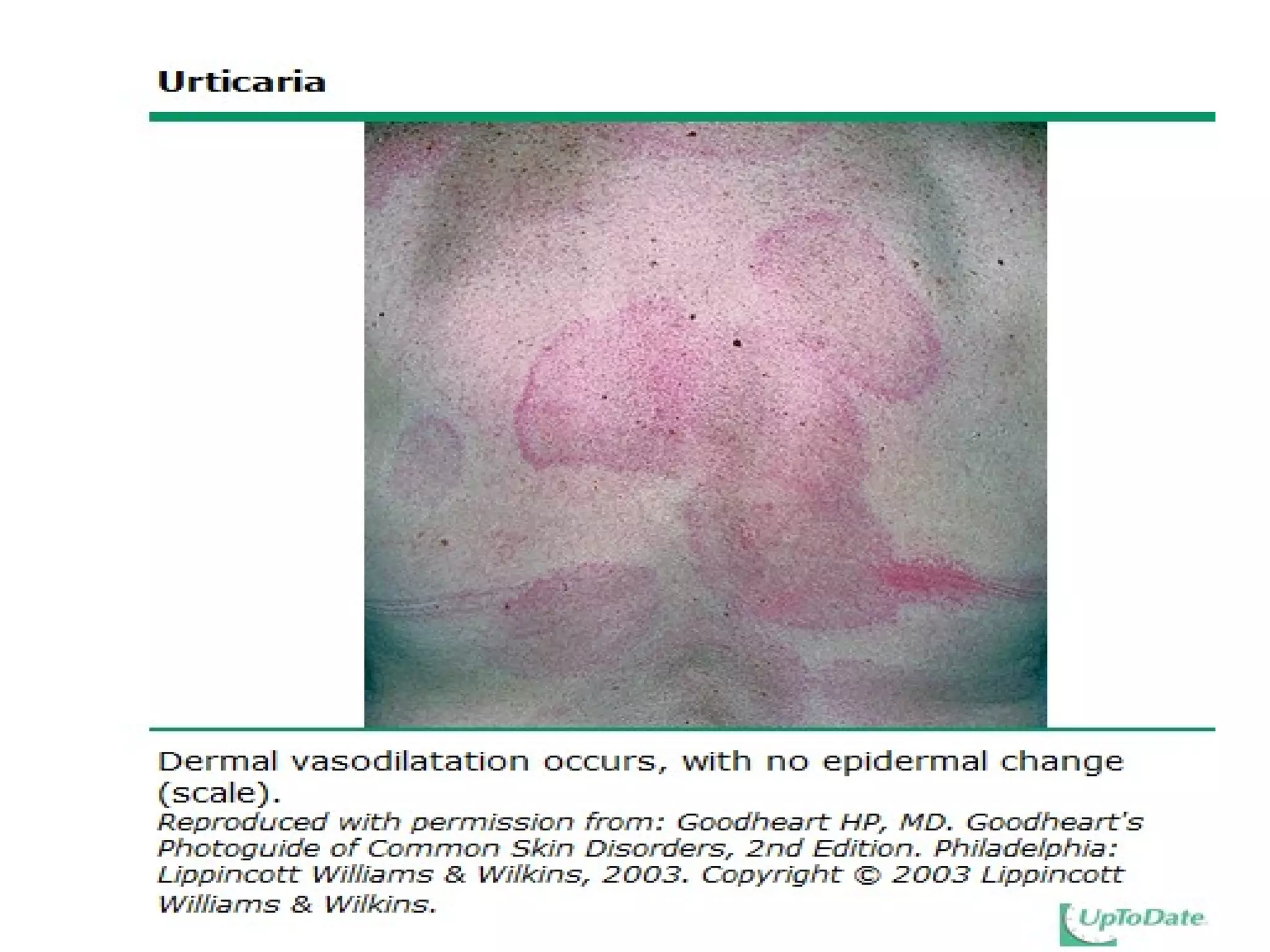
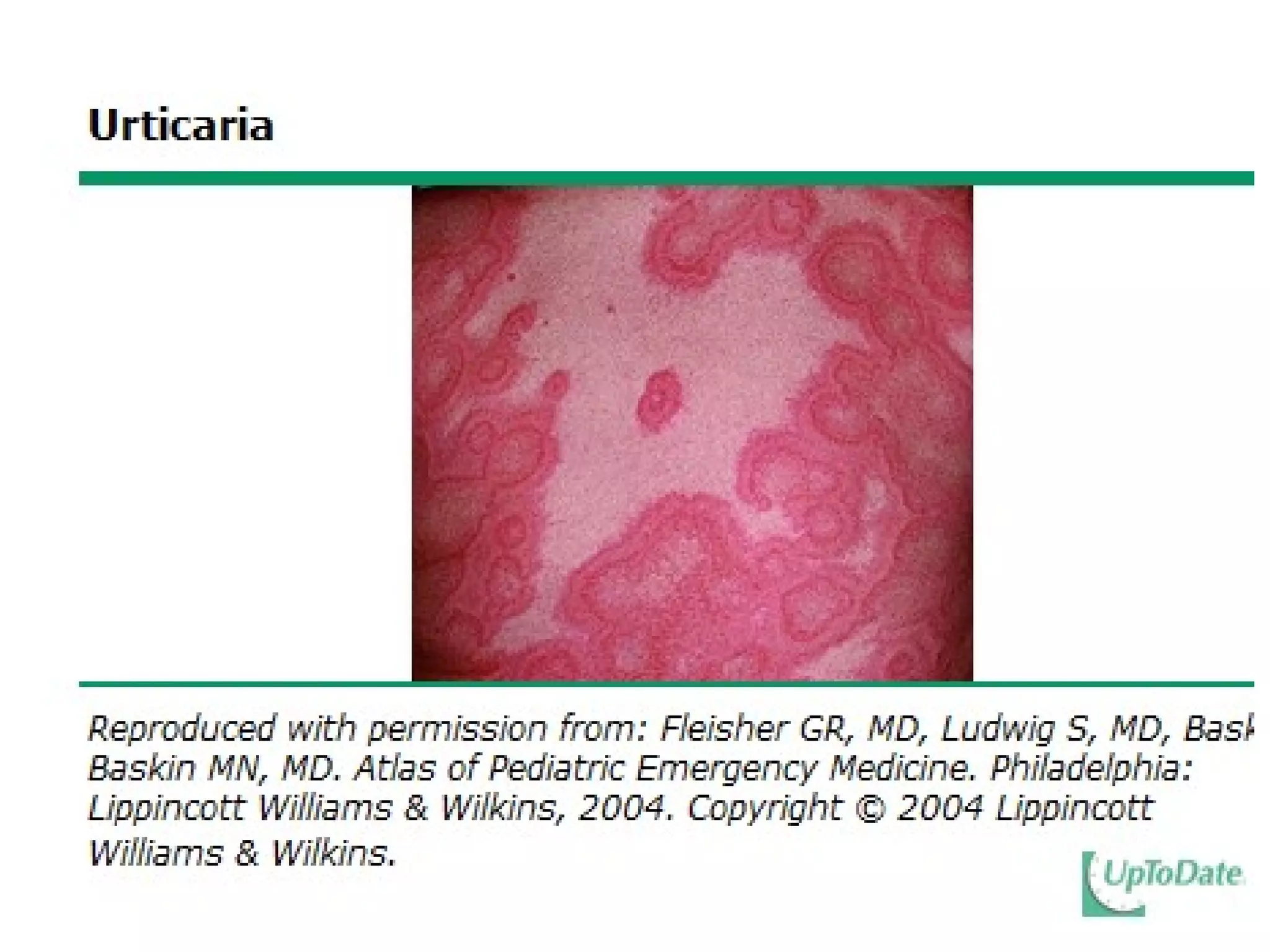
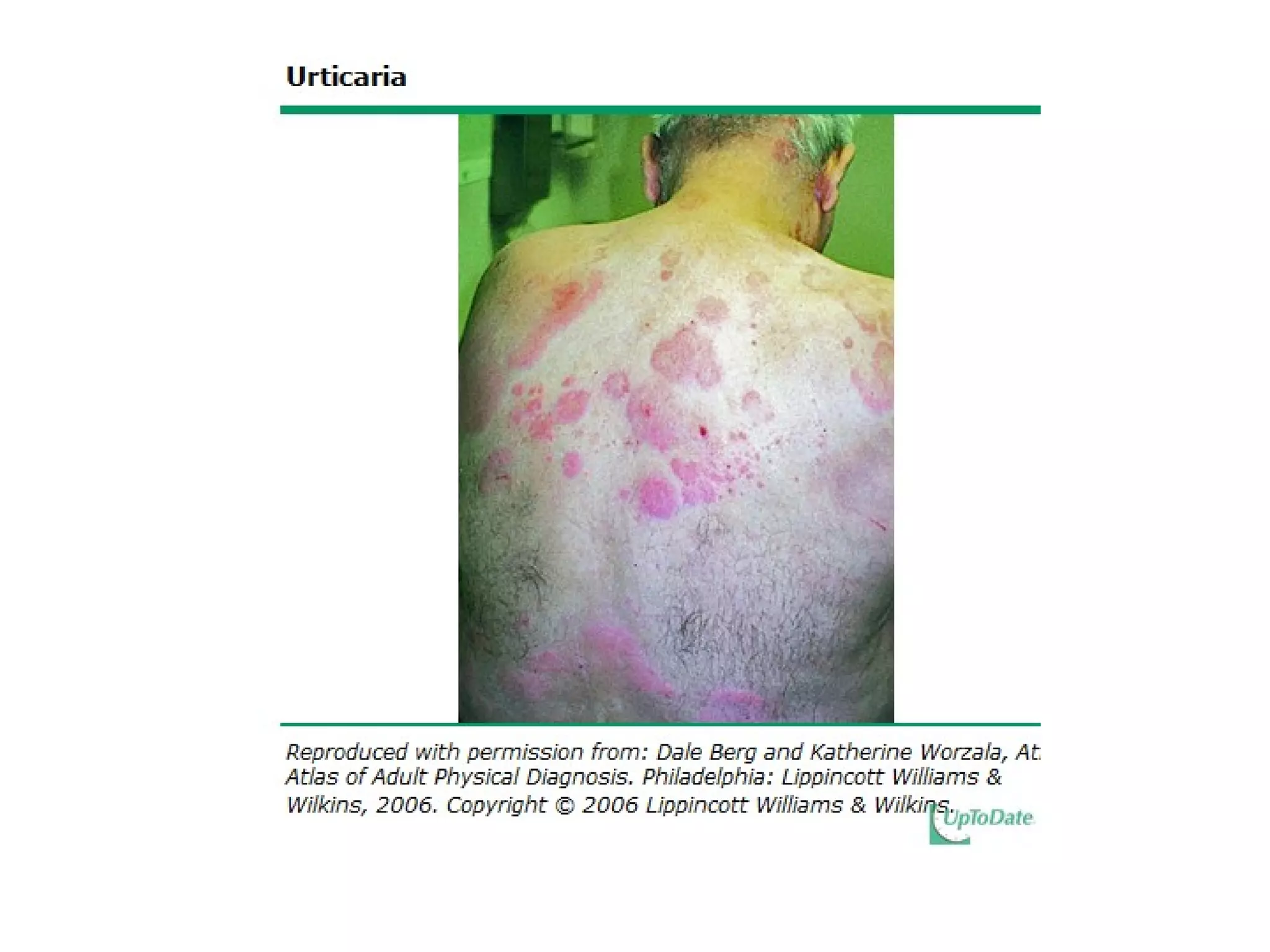
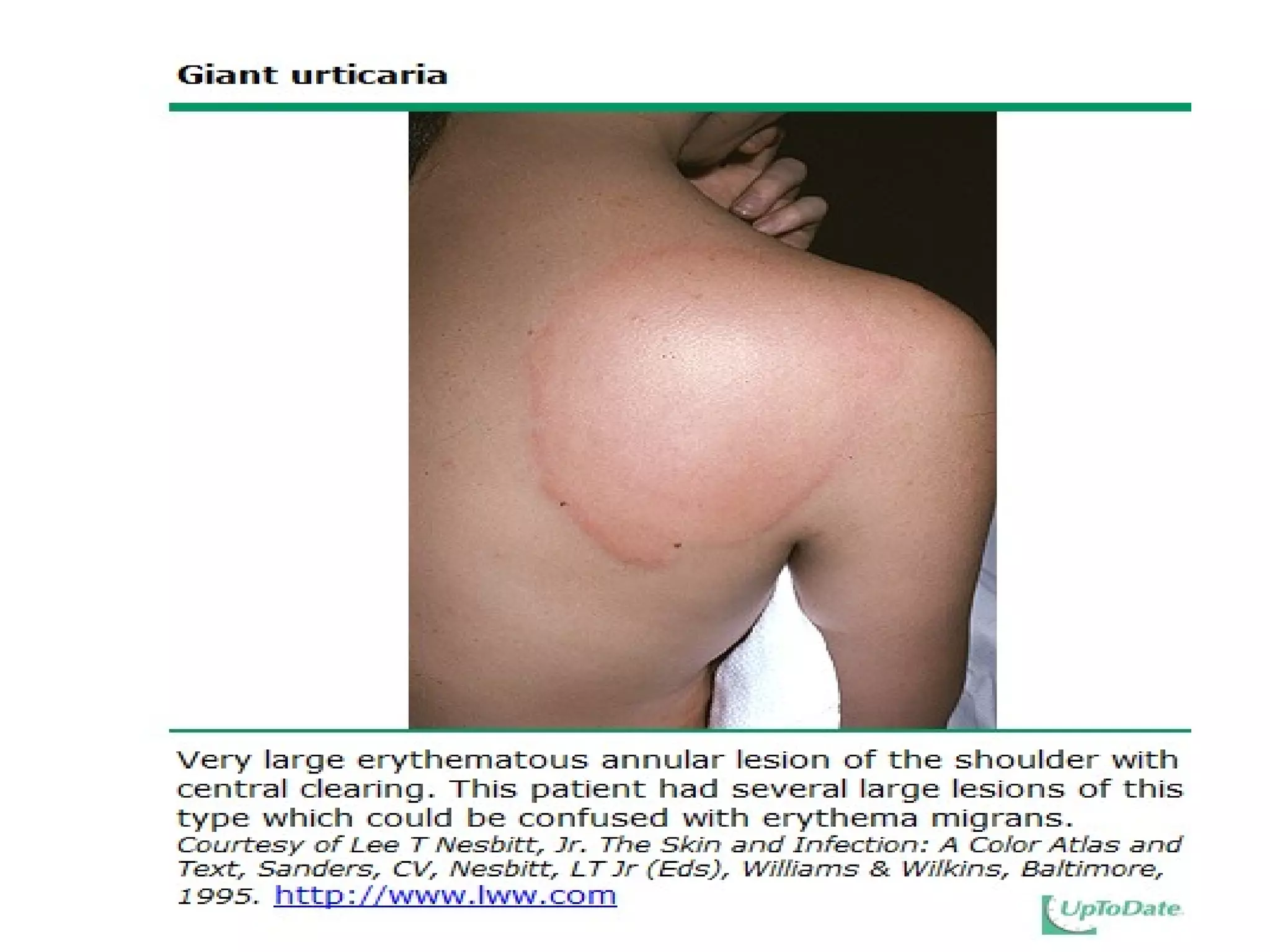
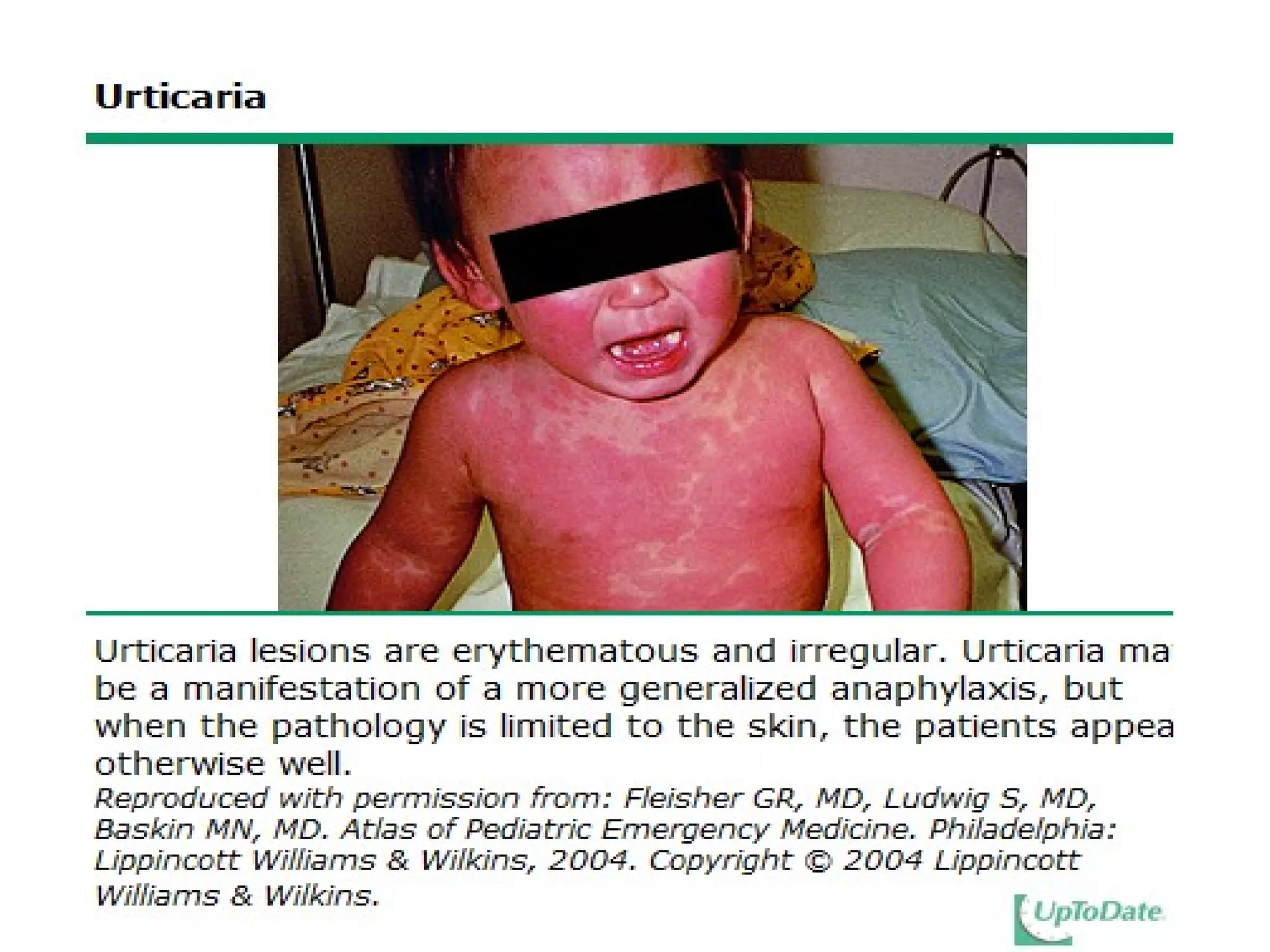
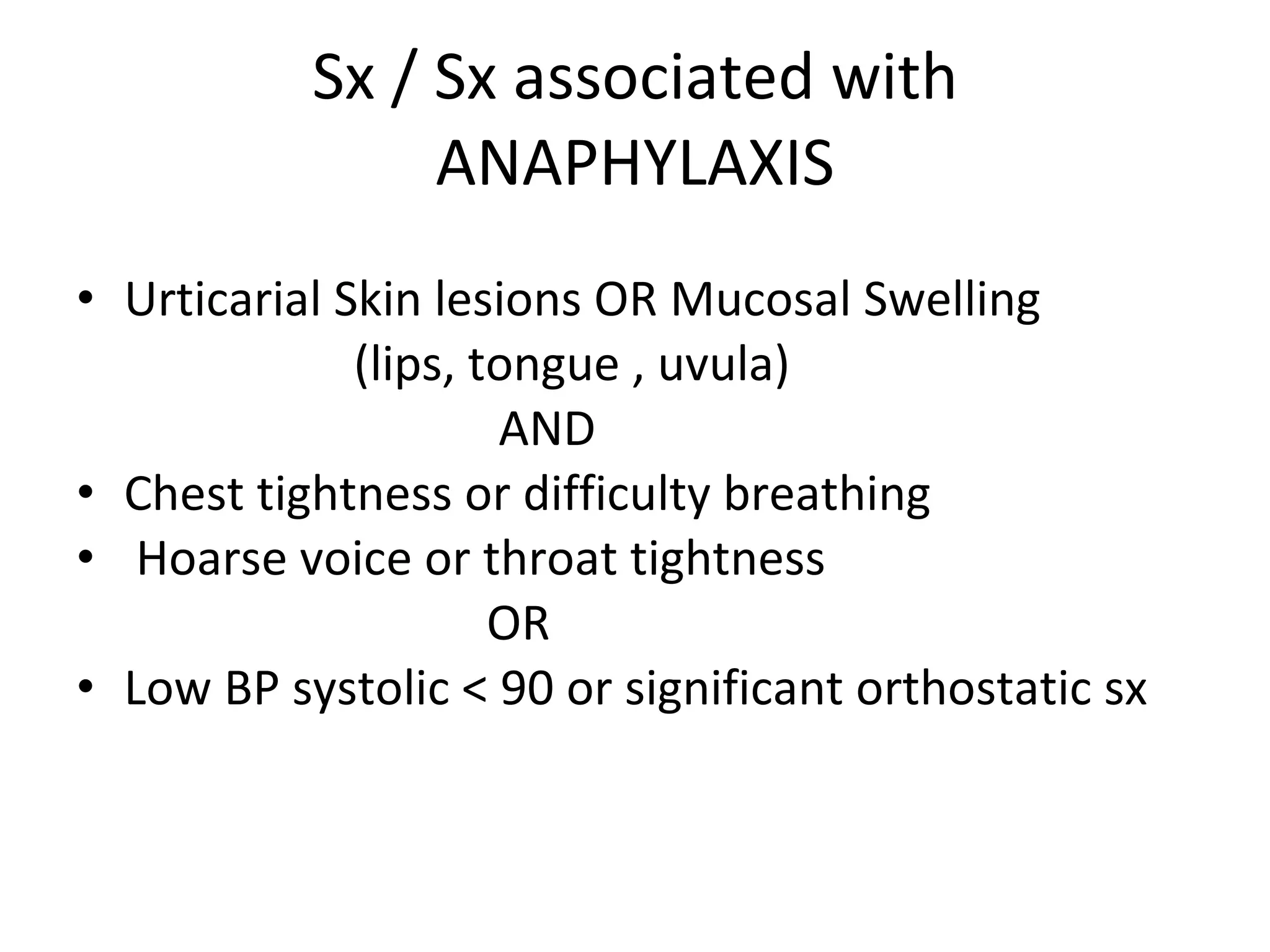
The document summarizes key changes to CDC guidelines for prevention of early-onset group B Streptococcus (GBS) disease. These include expanded laboratory detection options for GBS, revised thresholds for reporting GBS in urine cultures, clarified definitions of adequate antibiotic prophylaxis, and recommendations for neonatal management and treatment of penicillin-allergic pregnant women. Laboratories are no longer required to report low levels of GBS in urine cultures but providers should still consider these women GBS positive. Clindamycin susceptibility testing is important for penicillin-allergic high-risk patients.