Elements and compounds
•Download as PPT, PDF•
0 likes•62 views
This document defines key chemistry concepts such as elements, compounds, mixtures, acids, bases, and pH. It explains that elements cannot be broken down further, while compounds are formed by chemical bonds between elements. Mixtures do not form new chemical bonds. Acids and bases are defined using indicators like litmus, and the importance of pH is described for areas like the human body, food processing, and plant growth. Common acids and bases are outlined along with safety precautions for handling them. Neutralization is also introduced.
Report
Share
Report
Share
Recommended
Elements Compounds and Mixtures (by Deven)

This document summarizes key chemistry concepts including elements, compounds, and mixtures. An element is a substance made of only one type of atom that cannot be broken down further. Compounds are substances made of two or more elements chemically bonded together in fixed proportions that cannot be separated by physical means. Mixtures contain two or more substances mixed but not chemically combined, and can be either homogeneous, with substances fully mixed, or heterogeneous, with substances remaining separate. Examples of each are provided.
Elements Compounds And Mixtures

This document provides an overview of acids, bases, and solutions. It defines acids as substances that release hydrogen ions in water, causing solutions to have a sour taste and be able to conduct electricity. Bases are defined as substances that accept hydrogen ions in water, releasing hydroxide ions and causing solutions to feel slippery and taste bitter. The document also discusses pH scale, acid and base strengths, indicators, and neutralization reactions between acids and bases.
Mixtures, solutions, elements, compounds

Mixtures and pure substances can be categorized and separated in different ways. Mixtures are combinations of substances that are not chemically combined and can be physically separated. Pure substances include elements, which consist of only one type of atom, and compounds, which are formed by chemical combination of two or more elements. Mixtures include heterogeneous mixtures where the parts can be easily distinguished, like mixtures, and homogeneous mixtures where the parts are evenly distributed and appear uniform, like solutions.
Elements compounds and mixtures notes

This document discusses the differences between elements, compounds, and mixtures. It defines each term and provides examples. Mixtures are combinations of substances that are not chemically combined and can be separated through physical means. Solutions, suspensions, and colloids are types of mixtures. Elements are pure substances that cannot be broken down further, while compounds are pure substances composed of two or more chemically bonded elements.
Preformulation (chemical properties)

The document discusses various chemical properties and processes that can affect drug stability during preformulation, including oxidation, hydrolysis, racemization, and polymerization. It provides details on the mechanisms and factors that influence these processes, as well as methods to prevent or reduce degradation, such as adjusting pH, adding antioxidants or chelating agents, and controlling temperature and light exposure. Racemization can impact a drug's pharmacological and toxicological properties due to differences between its enantiomers. Polymerization involves monomers reacting to form polymer chains or networks and is exemplified by the darkening of glucose solutions.
Element,compound and mixture

The document discusses elements, compounds, and mixtures. It defines each term and provides examples. Elements cannot be broken down further through chemical reactions, while compounds can be broken down into their constituent elements. Mixtures do not undergo chemical reactions and can be separated into their original substances. Compounds are uniform throughout while mixtures can be heterogeneous or homogeneous.
3.1 What is a solution? 

This chapter discusses different types of substances and mixtures. It defines a substance as matter that has a fixed composition and cannot be separated into simpler substances by physical processes like boiling or filtering. Compounds have a fixed ratio of atoms, while mixtures can have varying proportions and their components can be separated. Solutions are homogeneous mixtures that are evenly mixed on a molecular level. The chapter also describes how solutions form and the different types of solutions.
basics of ionic liquids and emulsions

in this presentation basic information regarding ionic liquids and emulsions is shared which includes pros, cons of ionic liquids, there applications, applications of emulsions process of ,making one ionic liquid is also included
Recommended
Elements Compounds and Mixtures (by Deven)

This document summarizes key chemistry concepts including elements, compounds, and mixtures. An element is a substance made of only one type of atom that cannot be broken down further. Compounds are substances made of two or more elements chemically bonded together in fixed proportions that cannot be separated by physical means. Mixtures contain two or more substances mixed but not chemically combined, and can be either homogeneous, with substances fully mixed, or heterogeneous, with substances remaining separate. Examples of each are provided.
Elements Compounds And Mixtures

This document provides an overview of acids, bases, and solutions. It defines acids as substances that release hydrogen ions in water, causing solutions to have a sour taste and be able to conduct electricity. Bases are defined as substances that accept hydrogen ions in water, releasing hydroxide ions and causing solutions to feel slippery and taste bitter. The document also discusses pH scale, acid and base strengths, indicators, and neutralization reactions between acids and bases.
Mixtures, solutions, elements, compounds

Mixtures and pure substances can be categorized and separated in different ways. Mixtures are combinations of substances that are not chemically combined and can be physically separated. Pure substances include elements, which consist of only one type of atom, and compounds, which are formed by chemical combination of two or more elements. Mixtures include heterogeneous mixtures where the parts can be easily distinguished, like mixtures, and homogeneous mixtures where the parts are evenly distributed and appear uniform, like solutions.
Elements compounds and mixtures notes

This document discusses the differences between elements, compounds, and mixtures. It defines each term and provides examples. Mixtures are combinations of substances that are not chemically combined and can be separated through physical means. Solutions, suspensions, and colloids are types of mixtures. Elements are pure substances that cannot be broken down further, while compounds are pure substances composed of two or more chemically bonded elements.
Preformulation (chemical properties)

The document discusses various chemical properties and processes that can affect drug stability during preformulation, including oxidation, hydrolysis, racemization, and polymerization. It provides details on the mechanisms and factors that influence these processes, as well as methods to prevent or reduce degradation, such as adjusting pH, adding antioxidants or chelating agents, and controlling temperature and light exposure. Racemization can impact a drug's pharmacological and toxicological properties due to differences between its enantiomers. Polymerization involves monomers reacting to form polymer chains or networks and is exemplified by the darkening of glucose solutions.
Element,compound and mixture

The document discusses elements, compounds, and mixtures. It defines each term and provides examples. Elements cannot be broken down further through chemical reactions, while compounds can be broken down into their constituent elements. Mixtures do not undergo chemical reactions and can be separated into their original substances. Compounds are uniform throughout while mixtures can be heterogeneous or homogeneous.
3.1 What is a solution? 

This chapter discusses different types of substances and mixtures. It defines a substance as matter that has a fixed composition and cannot be separated into simpler substances by physical processes like boiling or filtering. Compounds have a fixed ratio of atoms, while mixtures can have varying proportions and their components can be separated. Solutions are homogeneous mixtures that are evenly mixed on a molecular level. The chapter also describes how solutions form and the different types of solutions.
basics of ionic liquids and emulsions

in this presentation basic information regarding ionic liquids and emulsions is shared which includes pros, cons of ionic liquids, there applications, applications of emulsions process of ,making one ionic liquid is also included
Introduction to solutions (chapter 5)

This document introduces key concepts about solutions and mixtures. It defines a solution as a homogeneous mixture formed when a solute dissolves evenly throughout a solvent. Water is discussed as a common solvent that can dissolve many substances to form solutions. The reading also distinguishes between heterogeneous mixtures, where substances are not evenly mixed, and homogeneous mixtures or solutions, where the mixing appears uniform. An upcoming student activity is described where groups will create mixtures using water and different substances to observe solubility.
drug metabolism-hydrolysis

Hydrolysis is a phase I drug metabolism reaction that introduces a water molecule to cleave drugs like esters, amides, hydrazides, and carbamates, making them more water soluble. It works by breaking bonds between functional groups on a drug molecule and adding hydrophilic groups to increase water solubility. Examples given include the hydrolysis of atropine, cocaine, procaine, and acetylcholine. The hydrolysis reaction removes ester and amide groups from drug molecules through the addition of a water molecule, converting them into their acid and alcohol/amine components.
Elements compounds-mixtures

This document discusses the classification of matter into elements, compounds, and mixtures. It defines elements as pure substances made of only one type of atom that cannot be broken down further. Compounds are defined as pure substances made of two or more elements chemically bonded together. Mixtures are defined as a combination of two or more pure substances that are not chemically combined and can be separated physically. Examples are provided to demonstrate whether specific substances represent elements, compounds, or mixtures.
Elements, Compounds, And Mixtures

An element is a pure substance that cannot be separated into simpler substances. Elements have unique characteristic properties like melting point and reactivity that can be used to identify them. Elements are grouped into metals, nonmetals, and metalloids based on shared properties. Compounds are formed when two or more elements chemically combine to form a new substance with different properties. Mixtures are combinations of substances that do not chemically combine and can be separated physically.
Elements Compounds Mixtures

A resource do differentiate between elements, compounds and the different types of mixtures formed by matter.
Hydrolysis

This document discusses hydrolysis, beginning with its definition as the reaction of water with another chemical compound to form two or more products by ionizing the water molecule and splitting the other compound. It then provides examples of hydrolysis including the conversion of starch to glucose and saponification. The document goes on to explain the mechanism of hydrolysis where a molecule is cleaved into two parts by the addition of a water molecule, with one fragment gaining a hydrogen ion and the other a hydroxyl group. It also discusses different types of hydrolysis reactions and some common uses of hydrolysis like breaking down food, producing soap, and generating energy through the hydrolysis of ATP.
Acid Content of Common Fruits Experiment

This document contains a student's project on the role of chemistry in domestic activities. It includes an introduction describing the student, guide, and topic. It then discusses how chemistry is involved in many everyday domestic tasks like cooking, cleaning, and stain removal. Cooking relies on chemical reactions to change food as it is prepared. Cleaning agents work through the chemical properties of surfactants, chelating agents, enzymes, and other components that help dissolve dirt and grease. Bleaches and dry cleaning also use chemical processes to remove stains from clothing. The document provides examples of the underlying chemistry behind common activities in the home.
Classification of matter

The document discusses the classification of matter into pure substances and mixtures. Pure substances are either elements or compounds, both of which have a uniform composition. Mixtures contain two or more substances mixed together, and can be either homogeneous, with a uniform composition throughout, or heterogeneous, with a non-uniform composition. Common examples of pure substances and mixtures are provided.
Hydrolysis (2)

This document discusses chemical degradation, specifically hydrolysis, as a type of chemical degradation of drugs. It defines hydrolysis as the reaction of water with another chemical compound to form two or more products, involving the ionization of water and splitting of the other compound. The document outlines the main types of hydrolysis as ester hydrolysis, amide hydrolysis, and ring hydrolysis. It provides examples of drugs that undergo each type of hydrolysis, such as tetracaine undergoing ester hydrolysis and dibucaine undergoing amide hydrolysis. The document also notes that hydrolysis can be retarded through various approaches to protect drugs against this degradation pathway.
Decomposition and stabilization of pharmaceutical products

Drug stability:Stabilization of medicinal agents against common reactions like hydrolysis & oxidation. Accelerated stability testing in expiration dating of pharmaceutical dosage forms. Photolytic degradation and its prevention.
Classificationofmatter notes

1. Matter is classified as either pure substances or mixtures. Pure substances are uniform and consist of elements or compounds, while mixtures contain two or more substances mixed together.
2. Elements are the simplest pure substances made of only one type of atom, while compounds are formed by chemical bonds between different atoms in specific ratios.
3. Mixtures can be either homogeneous, with substances mixed uniformly, or heterogeneous, with distinct parts. Homogeneous mixtures include solutions and colloids, while heterogeneous mixtures have visible parts that settle over time like suspensions.
Antidotes

The document defines antidotes as therapeutic substances that counteract the toxic actions of xenobiotics. Antidotes are classified based on their mode of action into physiological, physical, and chemical antidotes. Physiological antidotes counteract poisons through opposing pharmacological effects, while physical antidotes interfere with poisons through adsorption, coating, or dissolving. Chemical antidotes specifically interact with or neutralize toxins through complex formation or metabolic conversion into less toxic products. Common ways antidotes work include inert complex formation, accelerated detoxification, reduced toxic conversion, receptor site competition, receptor site blockage, and bypassing toxic effects.
effect of various environment and processing on stability of formulations

The document discusses various factors that can affect the stability of pharmaceutical formulations, including moisture, heat, light, radiation, and discusses physical and chemical degradation pathways like hydrolysis, oxidation, and techniques to stabilize formulations against degradation. It provides examples of drugs that can undergo hydrolysis or oxidation and discusses preventive measures like using buffers, complexation, suppressing solubility, antioxidants, chelating agents, and appropriate vehicles to inhibit degradation reactions like hydrolysis and oxidation.
oxidation of drugs

it gives you a brief description that how oxidation can affect the stability of drug and causes degradation
Ch8. colloids system

This document is a chapter about colloidal systems from a book by Aliyi Gerina. It begins with an outline and learning objectives for the chapter. The introduction defines dispersed systems and classifies them as molecular dispersions, colloidal dispersions, or coarse dispersions based on particle size. Colloids can be further classified by the physical state of the dispersed and continuous phases. The chapter also describes the properties of lyophilic colloids, lyophobic colloids, and association colloids. It compares the solvation, preparation, viscosity, electrolyte effects, and other properties of these different types of colloidal systems.
Pharmaceutical Degradation

Pharmaceutical degradation can occur through physical, chemical, or microbiological processes. Physical degradation includes changes in appearance, properties like hardness or consistency, and polymorphic changes. Chemical degradation involves reactions like hydrolysis, oxidation, decarboxylation, isomerization, and polymerization that break down the drug. Microbial degradation is caused by microbial growth contaminating the product. Proper storage conditions and formulation design can help prevent degradation through control of factors like temperature, humidity, light exposure, and microbial contamination.
Incompatibility l1

This document discusses different types of incompatibilities that can occur when mixing ingredients in prescriptions. There are three main types: physical, chemical, and therapeutic incompatibilities. Physical incompatibilities involve a visible change, such as insolubility or immiscibility, resulting in an uneven mixture. Chemical incompatibilities occur via reactions like pH changes that alter the chemical properties. Therapeutic incompatibilities change the intended medical effects. The document provides examples and remedies for each type, such as altering solvents, volumes, or adding emulsifying agents to overcome physical incompatibilities. Care must be taken when mixing ingredients to avoid unwanted interactions.
Pharmaceutical incompatibility

This document discusses pharmaceutical incompatibilities, which occur when ingredients in a prescription interact in undesirable ways. It classifies incompatibilities as physical, chemical, or therapeutic. Physical incompatibilities involve changes in properties like solubility from mixing solvents or ingredients. Chemical incompatibilities involve immediate reactions like oxidation, acid-base reactions, or hydrolysis. Therapeutic incompatibilities modify a drug's intended effects when combined with another drug or excipient. The document provides examples like immiscibility, insolubility, oxidation-reduction reactions, and contraindicated drugs. Rectifying methods include vigorous shaking, choice of emulsifying agents, use of antioxidants, and avoiding certain drug combinations.
Ch4. solubility and distribution phenomena

This document discusses solubility and distribution phenomena and was written by Aliyi Gerina from Bule Hora University. It defines key terms like solute, solvent, solution and solubility. It explains that solubility depends on interactions between solute and solvent molecules. Polar solutes dissolve best in polar solvents due to interactions like hydrogen bonding and dipole-dipole attractions. The document outlines factors that influence solubility such as temperature, pressure, and the ratio of polar to nonpolar groups in a molecule. It also discusses solubility of different forms of matter like gases in liquids, liquids in liquids, and solids in liquids.
Drug Incompatibility.

This presentation discusses drug incompatibility, which occurs when two or more substances that are mixed produce an undesirable effect. It defines and classifies incompatibility, including physical/chemical incompatibility and therapeutic incompatibility. Physical incompatibility can result in changes in properties like color, odor, or solubility. Chemical incompatibility involves chemical reactions between substances. Therapeutic incompatibility refers to interactions that occur in the body. The presentation also discusses types of drug interactions like drug-drug and drug-food, and provides examples. It explains how to prevent drug interactions by informing doctors of all medications and supplements and carefully reading drug labels.
Introduction to biochemistry

This biology course covers topics like biochemistry, metabolism, genetics, homeostasis, and ecology. It examines the structure and functions of living matter at the molecular level, including biomolecules like proteins, lipids, nucleic acids, and carbohydrates. Key concepts covered are the levels of protein structure, structures and functions of animal, plant and prokaryotic cells, pH and buffering, and chemical reactions in living systems like oxidation, reduction, hydrolysis, and condensation.
collection 1 of solution and pH 1111.ppt

The document provides information on matter, mixtures, and solutions. It defines matter as any substance that has mass and takes up space. Mixtures are either homogeneous, with a uniform composition, or heterogeneous. Solutions are homogeneous mixtures composed of solutes dissolved in solvents. True solutions are also called crystalloids and have particle sizes between 0.01 and 1 nm, making them transparent and unable to be separated by filtration.
More Related Content
What's hot
Introduction to solutions (chapter 5)

This document introduces key concepts about solutions and mixtures. It defines a solution as a homogeneous mixture formed when a solute dissolves evenly throughout a solvent. Water is discussed as a common solvent that can dissolve many substances to form solutions. The reading also distinguishes between heterogeneous mixtures, where substances are not evenly mixed, and homogeneous mixtures or solutions, where the mixing appears uniform. An upcoming student activity is described where groups will create mixtures using water and different substances to observe solubility.
drug metabolism-hydrolysis

Hydrolysis is a phase I drug metabolism reaction that introduces a water molecule to cleave drugs like esters, amides, hydrazides, and carbamates, making them more water soluble. It works by breaking bonds between functional groups on a drug molecule and adding hydrophilic groups to increase water solubility. Examples given include the hydrolysis of atropine, cocaine, procaine, and acetylcholine. The hydrolysis reaction removes ester and amide groups from drug molecules through the addition of a water molecule, converting them into their acid and alcohol/amine components.
Elements compounds-mixtures

This document discusses the classification of matter into elements, compounds, and mixtures. It defines elements as pure substances made of only one type of atom that cannot be broken down further. Compounds are defined as pure substances made of two or more elements chemically bonded together. Mixtures are defined as a combination of two or more pure substances that are not chemically combined and can be separated physically. Examples are provided to demonstrate whether specific substances represent elements, compounds, or mixtures.
Elements, Compounds, And Mixtures

An element is a pure substance that cannot be separated into simpler substances. Elements have unique characteristic properties like melting point and reactivity that can be used to identify them. Elements are grouped into metals, nonmetals, and metalloids based on shared properties. Compounds are formed when two or more elements chemically combine to form a new substance with different properties. Mixtures are combinations of substances that do not chemically combine and can be separated physically.
Elements Compounds Mixtures

A resource do differentiate between elements, compounds and the different types of mixtures formed by matter.
Hydrolysis

This document discusses hydrolysis, beginning with its definition as the reaction of water with another chemical compound to form two or more products by ionizing the water molecule and splitting the other compound. It then provides examples of hydrolysis including the conversion of starch to glucose and saponification. The document goes on to explain the mechanism of hydrolysis where a molecule is cleaved into two parts by the addition of a water molecule, with one fragment gaining a hydrogen ion and the other a hydroxyl group. It also discusses different types of hydrolysis reactions and some common uses of hydrolysis like breaking down food, producing soap, and generating energy through the hydrolysis of ATP.
Acid Content of Common Fruits Experiment

This document contains a student's project on the role of chemistry in domestic activities. It includes an introduction describing the student, guide, and topic. It then discusses how chemistry is involved in many everyday domestic tasks like cooking, cleaning, and stain removal. Cooking relies on chemical reactions to change food as it is prepared. Cleaning agents work through the chemical properties of surfactants, chelating agents, enzymes, and other components that help dissolve dirt and grease. Bleaches and dry cleaning also use chemical processes to remove stains from clothing. The document provides examples of the underlying chemistry behind common activities in the home.
Classification of matter

The document discusses the classification of matter into pure substances and mixtures. Pure substances are either elements or compounds, both of which have a uniform composition. Mixtures contain two or more substances mixed together, and can be either homogeneous, with a uniform composition throughout, or heterogeneous, with a non-uniform composition. Common examples of pure substances and mixtures are provided.
Hydrolysis (2)

This document discusses chemical degradation, specifically hydrolysis, as a type of chemical degradation of drugs. It defines hydrolysis as the reaction of water with another chemical compound to form two or more products, involving the ionization of water and splitting of the other compound. The document outlines the main types of hydrolysis as ester hydrolysis, amide hydrolysis, and ring hydrolysis. It provides examples of drugs that undergo each type of hydrolysis, such as tetracaine undergoing ester hydrolysis and dibucaine undergoing amide hydrolysis. The document also notes that hydrolysis can be retarded through various approaches to protect drugs against this degradation pathway.
Decomposition and stabilization of pharmaceutical products

Drug stability:Stabilization of medicinal agents against common reactions like hydrolysis & oxidation. Accelerated stability testing in expiration dating of pharmaceutical dosage forms. Photolytic degradation and its prevention.
Classificationofmatter notes

1. Matter is classified as either pure substances or mixtures. Pure substances are uniform and consist of elements or compounds, while mixtures contain two or more substances mixed together.
2. Elements are the simplest pure substances made of only one type of atom, while compounds are formed by chemical bonds between different atoms in specific ratios.
3. Mixtures can be either homogeneous, with substances mixed uniformly, or heterogeneous, with distinct parts. Homogeneous mixtures include solutions and colloids, while heterogeneous mixtures have visible parts that settle over time like suspensions.
Antidotes

The document defines antidotes as therapeutic substances that counteract the toxic actions of xenobiotics. Antidotes are classified based on their mode of action into physiological, physical, and chemical antidotes. Physiological antidotes counteract poisons through opposing pharmacological effects, while physical antidotes interfere with poisons through adsorption, coating, or dissolving. Chemical antidotes specifically interact with or neutralize toxins through complex formation or metabolic conversion into less toxic products. Common ways antidotes work include inert complex formation, accelerated detoxification, reduced toxic conversion, receptor site competition, receptor site blockage, and bypassing toxic effects.
effect of various environment and processing on stability of formulations

The document discusses various factors that can affect the stability of pharmaceutical formulations, including moisture, heat, light, radiation, and discusses physical and chemical degradation pathways like hydrolysis, oxidation, and techniques to stabilize formulations against degradation. It provides examples of drugs that can undergo hydrolysis or oxidation and discusses preventive measures like using buffers, complexation, suppressing solubility, antioxidants, chelating agents, and appropriate vehicles to inhibit degradation reactions like hydrolysis and oxidation.
oxidation of drugs

it gives you a brief description that how oxidation can affect the stability of drug and causes degradation
Ch8. colloids system

This document is a chapter about colloidal systems from a book by Aliyi Gerina. It begins with an outline and learning objectives for the chapter. The introduction defines dispersed systems and classifies them as molecular dispersions, colloidal dispersions, or coarse dispersions based on particle size. Colloids can be further classified by the physical state of the dispersed and continuous phases. The chapter also describes the properties of lyophilic colloids, lyophobic colloids, and association colloids. It compares the solvation, preparation, viscosity, electrolyte effects, and other properties of these different types of colloidal systems.
Pharmaceutical Degradation

Pharmaceutical degradation can occur through physical, chemical, or microbiological processes. Physical degradation includes changes in appearance, properties like hardness or consistency, and polymorphic changes. Chemical degradation involves reactions like hydrolysis, oxidation, decarboxylation, isomerization, and polymerization that break down the drug. Microbial degradation is caused by microbial growth contaminating the product. Proper storage conditions and formulation design can help prevent degradation through control of factors like temperature, humidity, light exposure, and microbial contamination.
Incompatibility l1

This document discusses different types of incompatibilities that can occur when mixing ingredients in prescriptions. There are three main types: physical, chemical, and therapeutic incompatibilities. Physical incompatibilities involve a visible change, such as insolubility or immiscibility, resulting in an uneven mixture. Chemical incompatibilities occur via reactions like pH changes that alter the chemical properties. Therapeutic incompatibilities change the intended medical effects. The document provides examples and remedies for each type, such as altering solvents, volumes, or adding emulsifying agents to overcome physical incompatibilities. Care must be taken when mixing ingredients to avoid unwanted interactions.
Pharmaceutical incompatibility

This document discusses pharmaceutical incompatibilities, which occur when ingredients in a prescription interact in undesirable ways. It classifies incompatibilities as physical, chemical, or therapeutic. Physical incompatibilities involve changes in properties like solubility from mixing solvents or ingredients. Chemical incompatibilities involve immediate reactions like oxidation, acid-base reactions, or hydrolysis. Therapeutic incompatibilities modify a drug's intended effects when combined with another drug or excipient. The document provides examples like immiscibility, insolubility, oxidation-reduction reactions, and contraindicated drugs. Rectifying methods include vigorous shaking, choice of emulsifying agents, use of antioxidants, and avoiding certain drug combinations.
Ch4. solubility and distribution phenomena

This document discusses solubility and distribution phenomena and was written by Aliyi Gerina from Bule Hora University. It defines key terms like solute, solvent, solution and solubility. It explains that solubility depends on interactions between solute and solvent molecules. Polar solutes dissolve best in polar solvents due to interactions like hydrogen bonding and dipole-dipole attractions. The document outlines factors that influence solubility such as temperature, pressure, and the ratio of polar to nonpolar groups in a molecule. It also discusses solubility of different forms of matter like gases in liquids, liquids in liquids, and solids in liquids.
Drug Incompatibility.

This presentation discusses drug incompatibility, which occurs when two or more substances that are mixed produce an undesirable effect. It defines and classifies incompatibility, including physical/chemical incompatibility and therapeutic incompatibility. Physical incompatibility can result in changes in properties like color, odor, or solubility. Chemical incompatibility involves chemical reactions between substances. Therapeutic incompatibility refers to interactions that occur in the body. The presentation also discusses types of drug interactions like drug-drug and drug-food, and provides examples. It explains how to prevent drug interactions by informing doctors of all medications and supplements and carefully reading drug labels.
What's hot (20)
Decomposition and stabilization of pharmaceutical products

Decomposition and stabilization of pharmaceutical products
effect of various environment and processing on stability of formulations

effect of various environment and processing on stability of formulations
Similar to Elements and compounds
Introduction to biochemistry

This biology course covers topics like biochemistry, metabolism, genetics, homeostasis, and ecology. It examines the structure and functions of living matter at the molecular level, including biomolecules like proteins, lipids, nucleic acids, and carbohydrates. Key concepts covered are the levels of protein structure, structures and functions of animal, plant and prokaryotic cells, pH and buffering, and chemical reactions in living systems like oxidation, reduction, hydrolysis, and condensation.
collection 1 of solution and pH 1111.ppt

The document provides information on matter, mixtures, and solutions. It defines matter as any substance that has mass and takes up space. Mixtures are either homogeneous, with a uniform composition, or heterogeneous. Solutions are homogeneous mixtures composed of solutes dissolved in solvents. True solutions are also called crystalloids and have particle sizes between 0.01 and 1 nm, making them transparent and unable to be separated by filtration.
Classification of Matter (General Chemistry)

Each of these classifications is based on distinct properties of matter, and they help us understand and study the world around us.
Properties of water and aqueous solutions

Akhuwat Faisalabad Institute Of Research Science and
Technology.....
This video is a course context of 1st prof Bs (hons) biotechnology.. which is prepared by momin khan niazi...........
Compounds

Compounds contain two or more elements chemically combined in fixed proportions. Compounds can be broken down into simpler substances but elements cannot. Examples of compounds include table salt, sucrose, nitrogen gas, and water. Compounds are classified as acids, bases, or salts. Acids are sour and react with metals and bases to produce hydrogen gas and water. Bases have a slippery feel and react with oils. The strength of acids and bases depends on how much they dissociate, with strong acids and bases completely dissociating and weak ones only partially. Common strong acids include sulfuric and hydrochloric acids while common bases include calcium hydroxide and magnesium hydroxide.
Hardness of water

This document discusses hardness of water. It defines hardness as being caused by calcium and magnesium ions which can be temporary (removed by boiling) or permanent. It describes common methods to determine water hardness, including soap titration and EDTA methods. Hardness is expressed in units of calcium carbonate equivalent by multiplying the weight of ions by their molecular weights.
Water and pH lecture Note .pptx

1. The document discusses water and pH, including the importance of water for life, its unique properties, and its roles as a solvent and reactant in biochemical reactions.
2. It describes the pH scale and how pH is a measure of hydrogen ion concentration, with acidic solutions having high H+ and alkaline solutions having high OH-.
3. Tight regulation of pH and water balance is critical for cellular functions and homeostasis, as pH affects enzyme activity, solute transport, and macromolecular structure.
biochemistry.ppt.Definitions of the biochem

What is biochemistry?
Biochemistry explores chemical processes related to living organisms. It is a laboratory-based science combining biology and chemistry.
Biochemists study the structure, composition, and chemical reactions of substances in living systems and, in turn, their functions and ways to control them. Biochemistry emerged as a separate discipline when scientists combined biology with organic, inorganic, and physical chemistry. They began to study areas such as:
How living things get energy from food
The chemical basis of heredity
What fundamental changes occur in disease
Biochemistry includes the sciences of molecular biology, immunochemistry, and neurochemistry, as well as bioinorganic, bioorganic, and biophysical chemistry.
What do biochemists do?
Biochemists interact with scientists from a wide variety of other disciplines, usually on problems that are a very small piece of a very large and complex system.
Biochemists in industry are interested in specific applications that will lead to marketable products
Biochemists in academia or government labs conduct more basic and less applied research
Where is biochemistry used?
Biochemistry has obvious applications in medicine, dentistry, and veterinary medicine. Other applications include:
Food Science
Biochemists determine the chemical composition of foods, research ways to develop abundant and inexpensive sources of nutritious foods, develop methods to extract nutrients from waste products, and/or invent ways to prolong the shelf life of food products.
Agriculture
Biochemists study the interaction of herbicides/insecticides with plants and pests. They examine the structure–activity relationships of compounds, determine their ability to inhibit growth, and evaluate the toxicological effects on surrounding life.
Pharmacology, Physiology, Microbiology, Toxicology, and Clinical Chemistry
Biochemists investigate the mechanisms of drug actions; engage in viral research; conduct research pertaining to organ function; or use chemical concepts, procedures, and techniques to study the diagnosis and therapy of disease and the assessment of health.
Water Quality Control & Treatment 

This document discusses water quality control and treatment. It begins by defining water and its chemical and physical properties. It then discusses concepts related to sustainable development and water resources. Various units used to measure concentration in solutions are explained, including percent composition by mass, volume percent, mole fraction, molarity, normality, and mass per unit volume. The document outlines objectives in water quality control work and provides a general classification of pollutants. Finally, it identifies and describes common sources of water pollution from industrial, municipal, agricultural, natural, stormwater, and other activities.
Environmental significance of water - S T A R K .ppt

Water has unique properties that make it essential for life on Earth. It is the universal solvent and medium for biological and chemical processes. Its ability to moderate temperature through processes like evaporation and its expansion upon freezing help stabilize temperatures for organisms. Its polarity allows it to dissolve many substances including proteins and ions, forming hydration shells. Water's dissociation into H+ and OH- ions affects pH and biochemical reactions. Its properties related to density, heat capacity, and phase changes influence global heat transfer and circulation patterns in oceans.
chemistry of water ph acid and bases. 

Water is essential for life and makes up about two-thirds of the human body. It has no taste or smell and exists in solid, liquid, and gas forms. Water regulates body temperature, transports nutrients and waste, cushions joints, and maintains pH levels. The pH scale measures hydrogen ion concentration from 0-14, with 7 being neutral. Acids donate protons and have a pH below 7, while bases accept protons and have a pH above 7. Buffers resist pH changes and are important for biological processes.
Milady chemistry-2019 (2)

Chemistry plays an important role in cosmetology. Many cosmetology services rely on chemicals, from hair color products to skin care. Chemistry is the science dealing with the composition and properties of matter and how matter changes. The document defines key chemistry concepts like matter, elements, atoms, molecules, and the three states of matter. It explains how chemistry influences cosmetology through ingredients in products like hydroxide relaxers, zinc oxide in SPF, and hydrogen peroxide in hair color. Physical and chemical properties and changes are distinguished. The roles of oxidation, reduction, combustion, pH, and common ingredients are outlined.
WATER PURIFICATION

The document discusses the chemical properties of water and common water impurities. It states that water is sometimes called the universal solvent because it can dissolve many substances. The main water impurities mentioned are total suspended solids like dust, sand and clay. There are also two types of water hardness: temporary hardness caused by bicarbonates that can be removed by boiling, and permanent hardness caused by chlorides, nitrates and sulfates that require water softening. Various water purification methods are discussed like coagulation, flocculation, sedimentation and filtration.
Acid and Base.pptx

Corrosion is the gradual destruction of a metal due to reactions with its environment. Over time, corrosion changes the appearance of the metal as it breaks down and becomes weaker. Coating metals with paint or chemicals protects them from corrosion, but damage to these coatings allows corrosion to occur. Rusting refers specifically to the corrosion of iron through a reaction of iron with oxygen and water to form hydrated iron oxide. Factors like salt can increase the rate of corrosion. Methods of preventing corrosion include covering metals with grease, attaching zinc, painting, or coating with tin.
Biochemistry

The document provides information on biochemistry and chemistry concepts. It discusses the structure of atoms and defines elements, isotopes, and compounds. It then summarizes key biomolecules like carbohydrates, lipids, proteins, and nucleic acids. Finally, it explains important chemical reactions in living systems like hydrolysis and discusses how enzymes function as biological catalysts.
PHYSICAL BIOCHEMISTRY

1. Water is the universal solvent and is essential for life. It has unique properties such as hydrogen bonding that allow it to dissolve many polar substances and maintain homeostasis.
2. Solutions can be characterized by their concentration and type. Common ways to express concentration are molarity, molality, mass percent, and parts per million. Solutes influence colligative properties such as vapor pressure and boiling point.
3. Acids and bases are important classes of solutions. The pH scale quantifies the concentration of hydrogen ions and indicates if a solution is acidic or basic. Buffers resist changes in pH through chemical equilibria.
Biochemistry

The document provides an overview of biochemistry concepts including:
1) Atoms are the basic unit of matter and are composed of protons, neutrons, and electrons. Chemical bonds form molecules by sharing or transferring electrons.
2) Carbohydrates, lipids, proteins, and nucleic acids are the four major macromolecules that make up living things. They are formed by monomers linking through dehydration synthesis and broken down through hydrolysis.
3) Enzymes are protein catalysts that speed up biochemical reactions by lowering their activation energy. Factors like pH, temperature, and substrate/enzyme concentrations influence enzymatic reaction rates.
Unique properties of water

Water is a polar molecule that is essential for life. Its polarity allows it to dissolve many other polar substances and act as a solvent for biological molecules and reactions in living cells and organisms. Water has unique physical properties, such as a high heat capacity and ability to form hydrogen bonds, that make it well-suited to regulate temperature and support life processes on Earth. The polarity and hydrogen bonding of water molecules also allow water to have high surface tension and influence many other physical phenomena.
WATER QUALITY PARAMETERS-1-1.pptx

The document discusses various water quality parameters including water pollution, water quality standards, and important requirements of water for domestic use. It defines total solids as the total of all solids in a water sample, including total suspended solids, total dissolved solids, and volatile suspended solids. It also discusses how total solids are measured by weighing the solids present in a known water sample volume before and after drying to evaporate the water. The document provides classifications of impurities based on size and nature, and describes several common methods for analyzing water quality parameters.
DETERGENT PRODUCTION.pptx

The document discusses producing detergent using plantain peel ash extract. It begins with background on detergents and their properties. It then compares soaps and detergents, noting key differences. The aim is to produce detergent using plantain peel ash potassium hydroxide extract. Methods are described for preparing the ash extract, determining its pH and concentration. Production involves weighing ingredients and heating them to form the detergent. Characterization will include determining the detergent's pH, alkalinity, stability, and viscosity.
Similar to Elements and compounds (20)
Environmental significance of water - S T A R K .ppt

Environmental significance of water - S T A R K .ppt
Recently uploaded
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.The simplified electron and muon model, Oscillating Spacetime: The Foundation...

Discover the Simplified Electron and Muon Model: A New Wave-Based Approach to Understanding Particles delves into a groundbreaking theory that presents electrons and muons as rotating soliton waves within oscillating spacetime. Geared towards students, researchers, and science buffs, this book breaks down complex ideas into simple explanations. It covers topics such as electron waves, temporal dynamics, and the implications of this model on particle physics. With clear illustrations and easy-to-follow explanations, readers will gain a new outlook on the universe's fundamental nature.
How to Manage Your Lost Opportunities in Odoo 17 CRM

Odoo 17 CRM allows us to track why we lose sales opportunities with "Lost Reasons." This helps analyze our sales process and identify areas for improvement. Here's how to configure lost reasons in Odoo 17 CRM
Main Java[All of the Base Concepts}.docx

This is part 1 of my Java Learning Journey. This Contains Custom methods, classes, constructors, packages, multithreading , try- catch block, finally block and more.
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
Natural birth techniques - Mrs.Akanksha Trivedi Rama University

Natural birth techniques - Mrs.Akanksha Trivedi Rama UniversityAkanksha trivedi rama nursing college kanpur.
Natural birth techniques are various type such as/ water birth , alexender method, hypnosis, bradley method, lamaze method etcCommunity pharmacy- Social and preventive pharmacy UNIT 5

Covered community pharmacy topic of the subject Social and preventive pharmacy for Diploma and Bachelor of pharmacy
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশের অর্থনৈতিক সমীক্ষা ২০২৪ [Bangladesh Economic Review 2024 Bangla.pdf] কম্পিউটার , ট্যাব ও স্মার্ট ফোন ভার্সন সহ সম্পূর্ণ বাংলা ই-বুক বা pdf বই " সুচিপত্র ...বুকমার্ক মেনু 🔖 ও হাইপার লিংক মেনু 📝👆 যুক্ত ..
আমাদের সবার জন্য খুব খুব গুরুত্বপূর্ণ একটি বই ..বিসিএস, ব্যাংক, ইউনিভার্সিটি ভর্তি ও যে কোন প্রতিযোগিতা মূলক পরীক্ষার জন্য এর খুব ইম্পরট্যান্ট একটি বিষয় ...তাছাড়া বাংলাদেশের সাম্প্রতিক যে কোন ডাটা বা তথ্য এই বইতে পাবেন ...
তাই একজন নাগরিক হিসাবে এই তথ্য গুলো আপনার জানা প্রয়োজন ...।
বিসিএস ও ব্যাংক এর লিখিত পরীক্ষা ...+এছাড়া মাধ্যমিক ও উচ্চমাধ্যমিকের স্টুডেন্টদের জন্য অনেক কাজে আসবে ...
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

Denis is a dynamic and results-driven Chief Information Officer (CIO) with a distinguished career spanning information systems analysis and technical project management. With a proven track record of spearheading the design and delivery of cutting-edge Information Management solutions, he has consistently elevated business operations, streamlined reporting functions, and maximized process efficiency.
Certified as an ISO/IEC 27001: Information Security Management Systems (ISMS) Lead Implementer, Data Protection Officer, and Cyber Risks Analyst, Denis brings a heightened focus on data security, privacy, and cyber resilience to every endeavor.
His expertise extends across a diverse spectrum of reporting, database, and web development applications, underpinned by an exceptional grasp of data storage and virtualization technologies. His proficiency in application testing, database administration, and data cleansing ensures seamless execution of complex projects.
What sets Denis apart is his comprehensive understanding of Business and Systems Analysis technologies, honed through involvement in all phases of the Software Development Lifecycle (SDLC). From meticulous requirements gathering to precise analysis, innovative design, rigorous development, thorough testing, and successful implementation, he has consistently delivered exceptional results.
Throughout his career, he has taken on multifaceted roles, from leading technical project management teams to owning solutions that drive operational excellence. His conscientious and proactive approach is unwavering, whether he is working independently or collaboratively within a team. His ability to connect with colleagues on a personal level underscores his commitment to fostering a harmonious and productive workplace environment.
Date: May 29, 2024
Tags: Information Security, ISO/IEC 27001, ISO/IEC 42001, Artificial Intelligence, GDPR
-------------------------------------------------------------------------------
Find out more about ISO training and certification services
Training: ISO/IEC 27001 Information Security Management System - EN | PECB
ISO/IEC 42001 Artificial Intelligence Management System - EN | PECB
General Data Protection Regulation (GDPR) - Training Courses - EN | PECB
Webinars: https://pecb.com/webinars
Article: https://pecb.com/article
-------------------------------------------------------------------------------
For more information about PECB:
Website: https://pecb.com/
LinkedIn: https://www.linkedin.com/company/pecb/
Facebook: https://www.facebook.com/PECBInternational/
Slideshare: http://www.slideshare.net/PECBCERTIFICATION
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.Recently uploaded (20)
Pride Month Slides 2024 David Douglas School District

Pride Month Slides 2024 David Douglas School District
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
Liberal Approach to the Study of Indian Politics.pdf

Liberal Approach to the Study of Indian Politics.pdf
The simplified electron and muon model, Oscillating Spacetime: The Foundation...

The simplified electron and muon model, Oscillating Spacetime: The Foundation...
How to Manage Your Lost Opportunities in Odoo 17 CRM

How to Manage Your Lost Opportunities in Odoo 17 CRM
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
Natural birth techniques - Mrs.Akanksha Trivedi Rama University

Natural birth techniques - Mrs.Akanksha Trivedi Rama University
Community pharmacy- Social and preventive pharmacy UNIT 5

Community pharmacy- Social and preventive pharmacy UNIT 5
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
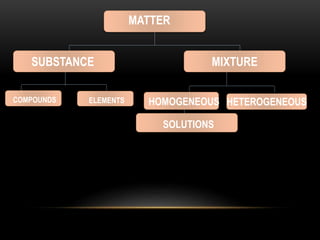
Elements and compounds
- 2. ELEMENT • An element is a substance that cannot be broken down further either by physical or chemical means. It is made up of only one kind of atom. • Each element has a unique properties. No two elements have the same properties.
- 3. Elements cannot be broken down using chemical methods. However, they can be changed into other elements through nuclear reactions. Research about the different types of nuclear reactions that change the identity of the elements.
- 4. A compound always contains the same ratio of its constituent elements. For example, one molecule of water always contains one atom of oxygen and two atoms of hydrogen. The properties of a compound are different from its constituent elements. For example sodium is a reactive metal while chlorine is a poisonous gas. However, when they combine, a safe and stable sodium chloride, (table salt) is formed. Water (H2O), Ammonia (NH3), and table salt (NaCl) are examples of compounds.
- 6. COMPOUND • A compound is a substance that can be broken down into its constituent elements only by chemical means. It is a combination of two or more elements bound by chemical bonds. For example, elements X and Y may combine to form to a compound. If one atom of element X forms a compound with two atoms of element Y, then the resulting compound is XY2
- 8. MIXTURE • A mixture forms when two or more elements or compounds are mixed but are not combined chemically. Therefore, no new chemical bonds are formed when a mixture is prepared. For example, elements X and Y may combine to form a mixture, but these elements would not be linked in a bond.
- 10. Each of the constituents of a mixture retains its characteristic properties. They can be separated by physical methods such as distillation, dialysis, electrophoresis and chromatography. These methods are based on the difference in the boiling point, size, solubility, density and electrical charge of the components.
- 11. • There are two kinds of mixtures, namely, heterogeneous mixtures and homogeneous mixtures. A homogeneous mixture is characterized by having a single phase while a heterogeneous mixture has more than one phase.
- 12. • Heterogeneous mixtures: sand and water, iron fillings and sand • Homogeneous mixtures: air, mineral water, orange juice
- 13. CLASSIFY THE FOLLOWING MIXTURES AS HOMOGENEOUS OR HETEROGENEOUS. • sand and oil • sugar and water • sand and alcohol • fruit salad • orange juice • rubbing alcohol
- 16. PERIODIC TABLE OF ELEMENTS • Dmitri Mendeleev • A table that shows the elements arranged systematically
- 17. • Ferrous Sulphate • Monosodium Glutamate
- 33. Element Symbol Latin Name Antimony Sb Stibium Copper Cu Cuprum Gold Au Aurum Iron Fe Ferrum Lead Pb Plumbum Mercury Hg Hydragyrum Potassium K Kalium Silver Ag Argentum Sodium Na Natrium Tin Sn Stannum Tungsten W Wolfram
- 34. ACID AND BASE
- 35. INDICATOR • A dye that changes into a specific color depending on whether it is placed on acidic or basic solution. LITMUS • A dye taken from the lichen plant. It turns red in acidic mixtures and blue in basic mixtures.
- 36. IMPORTANCE OF PH
- 37. • pH and the Human Body • If the pH in the body is too basic, you will hyperventilate • If the blood becomes to basic, you will breathe slowly
- 38. • Changes in pH affect the growth of microorganisms , which cause food spoilage. • Most bacteria grows best at or near pH 7. To prevent this, pickling is an effective food preservation method because it lowers pH. • Use of pH in Food Processing and Fruit Preservation
- 39. • Control of pH in Soil • Some plants grow in acidic soil while others prefer basic soil. • Useful plants in the Philippines that grow in acidic soil • banana, kaimito, durian, pineapple, soy bean, coffee, eggplant, squash, kamote and rice. • Useful plants in the Philippines that grow in basic soil • grapes and pechay, • Useful plants in the Philippines that grow in neutral soil • orange, peanut, watermelon, beans, cabbage, tomato, corn, garlic and onion.
- 40. • The average pH of rain is 5.6. This slightly acidic pH is due to the presence of carbon dioxide in the air. • Rain with a pH below 5.6 is called acid rain. The acidic pollutants in the air come from the burning of fuels used in power plants, factories and vehicles produce gasses which are acidic. pH of Rainwater
- 41. • Most personal care products have pH kept at a specific levels to avoid harmful effects on the body. • pH 12 will dissolve hair Maintaining pH of Personal Care Products
- 42. SAFETY IN HANDLING ACIDS AND BASES
- 43. HYDROCHLORIC ACID (MURIATIC ACID) • Used in construction to remove excess mortar from bricks and in the home to remove hardened deposits from toilet bowls. • Concentrated solutions of HCl (about 38%) cause severe burns , but dilute solutions can be used safely in the home.
- 44. CAUTION FOR MURIATIC ACID • Harmful or fatal if swallowed. • Strong irritant to eye, skin, and mucous membrane. • Do not take internally • Avoid contact with eyes, nose an • Keep tightly sealed. • Keep out of reach of children.
- 45. SODIUM HYDROXIDE (LYE OR LIQUID SOSA) • Used to open clogged kitchen and toilet pipes, sinks and drains. • WARNING • Avoid contact with any part of the body • Causes severe eyes and skin damage and burns • Store in cool dry place and locked cabinet • Harmful or fatal if swallowed
- 46. NEUTRAL • Mixtures that are not acidic or basic
- 47. USES OF NEUTRALIZATION • Treating indigestion • Using toothpaste to avoid tooth decay • Treating soil • Treating factory waste