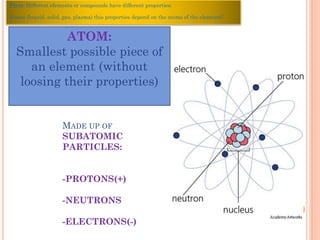
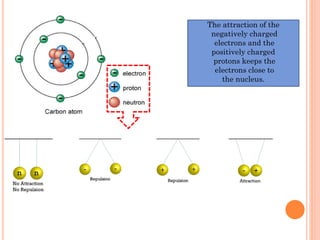
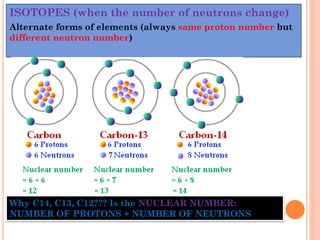
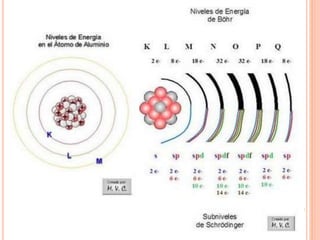
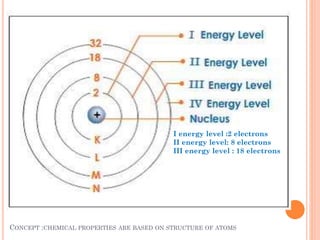
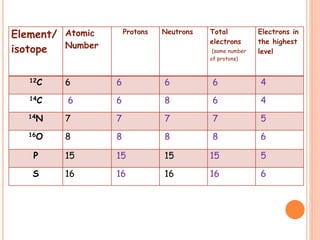
The document discusses the structure of atoms and the properties of elements. It states that atoms are made up of subatomic particles, including protons, neutrons, and electrons. The nucleus contains protons and neutrons, while electrons orbit the nucleus. The number of protons determines the element and is called the atomic number. Atoms can have different numbers of neutrons, forming isotopes of an element. The attraction between protons and electrons keeps the atom electrically neutral. The structure of atoms determines how elements react chemically.