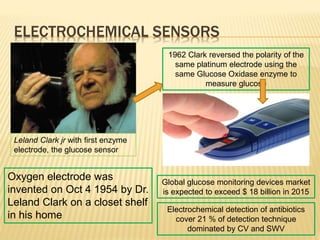
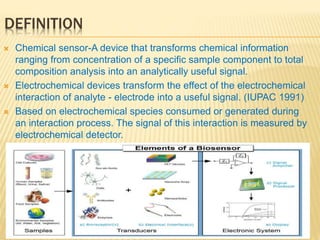
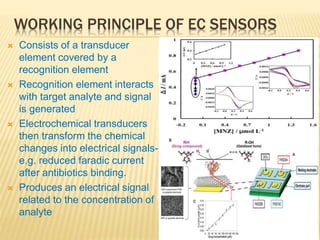
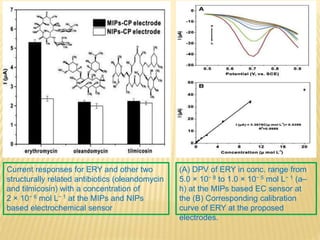
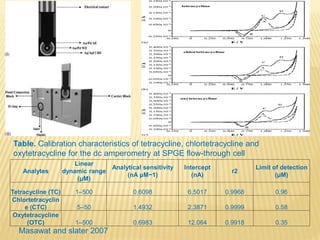
This document discusses electrochemical sensors for detecting antibiotic residues in food. It begins with an introduction on the increasing global use of antibiotics and development of antibiotic resistance. It then discusses the working principles of electrochemical sensors and how they can be used to detect antibiotics. Specifically, it describes how electrochemical sensors use recognition elements like enzymes, antibodies, aptamers, and molecularly imprinted polymers to detect antibiotics. It also discusses using different electrode systems and materials like carbon nanotubes, nanoparticles, and graphene to improve detection. The document aims to provide an overview of developing electrochemical sensor techniques for antibiotic residue detection in food.