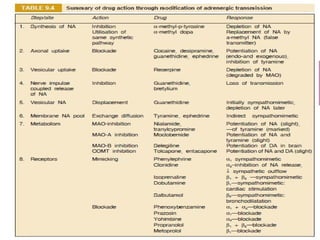
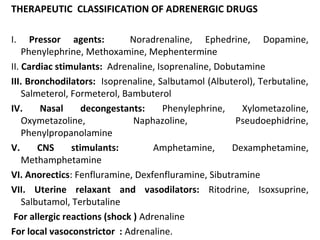
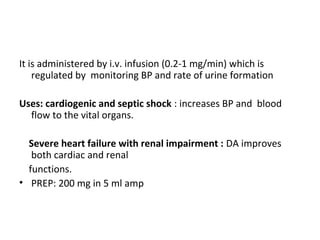
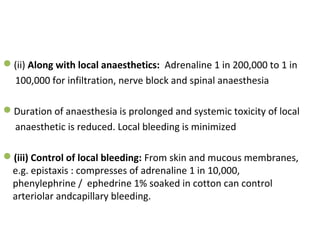
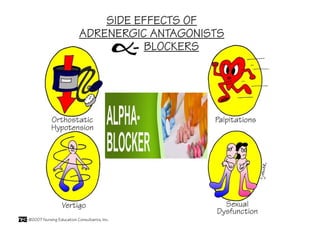
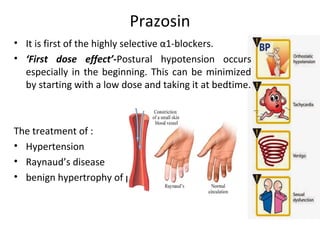
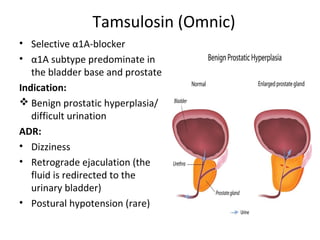
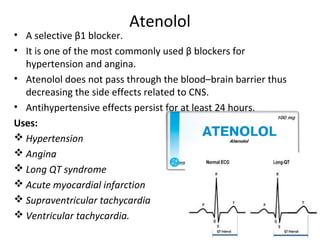
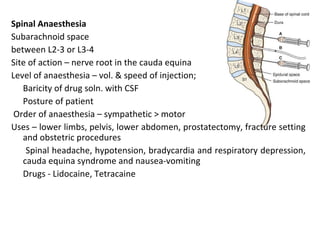
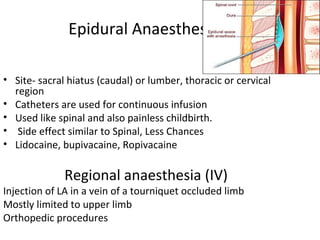
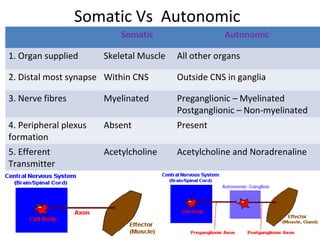
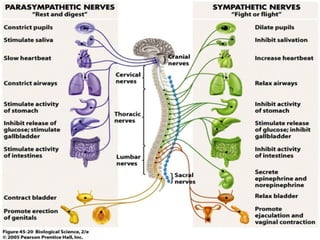
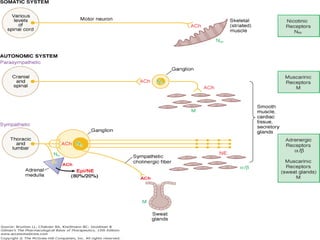
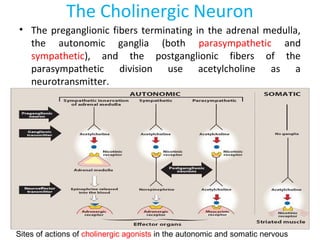
The document details the classifications and actions of drugs affecting the autonomic nervous system, distinguishing between cholinergic and adrenergic drugs based on their neurotransmitter affinities. It describes the structure and function of muscarinic and nicotinic receptors, the various cholinergic agonists, and the mechanism of action of anticholinesterases. Additionally, it outlines therapeutic applications and adverse effects of these drugs, emphasizing their role in conditions like glaucoma and myasthenia gravis.

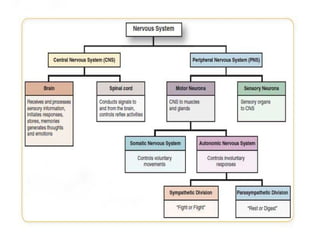
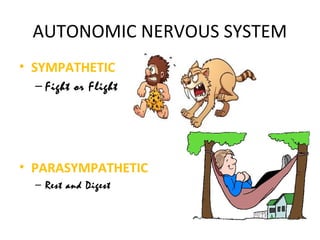











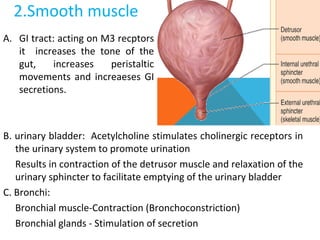
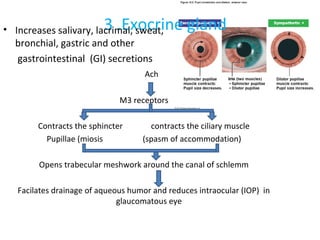







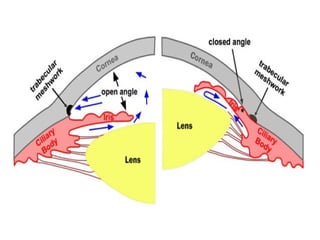

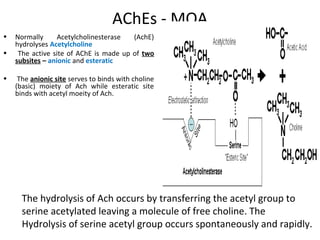
































































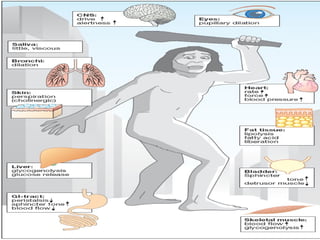






















![6. GIT: In isolated preparations of gut, relaxation occurs through
activation of both α2 and β2 receptors
• peristalsis is reduced and sphincters are constricted (α1)
7. BLADDER: Detrusor is relaxed (β2) and trigone is constricted (α1):
both actions can cause difficulty in micturition.
8. UTERUS: Non pregnant-Contraction[α1 > β2]
Pregnant-Relaxation[β2]](https://image.slidesharecdn.com/drugsactingonpns2-170203074854/85/Drugs-acting-on-PNS-132-320.jpg)