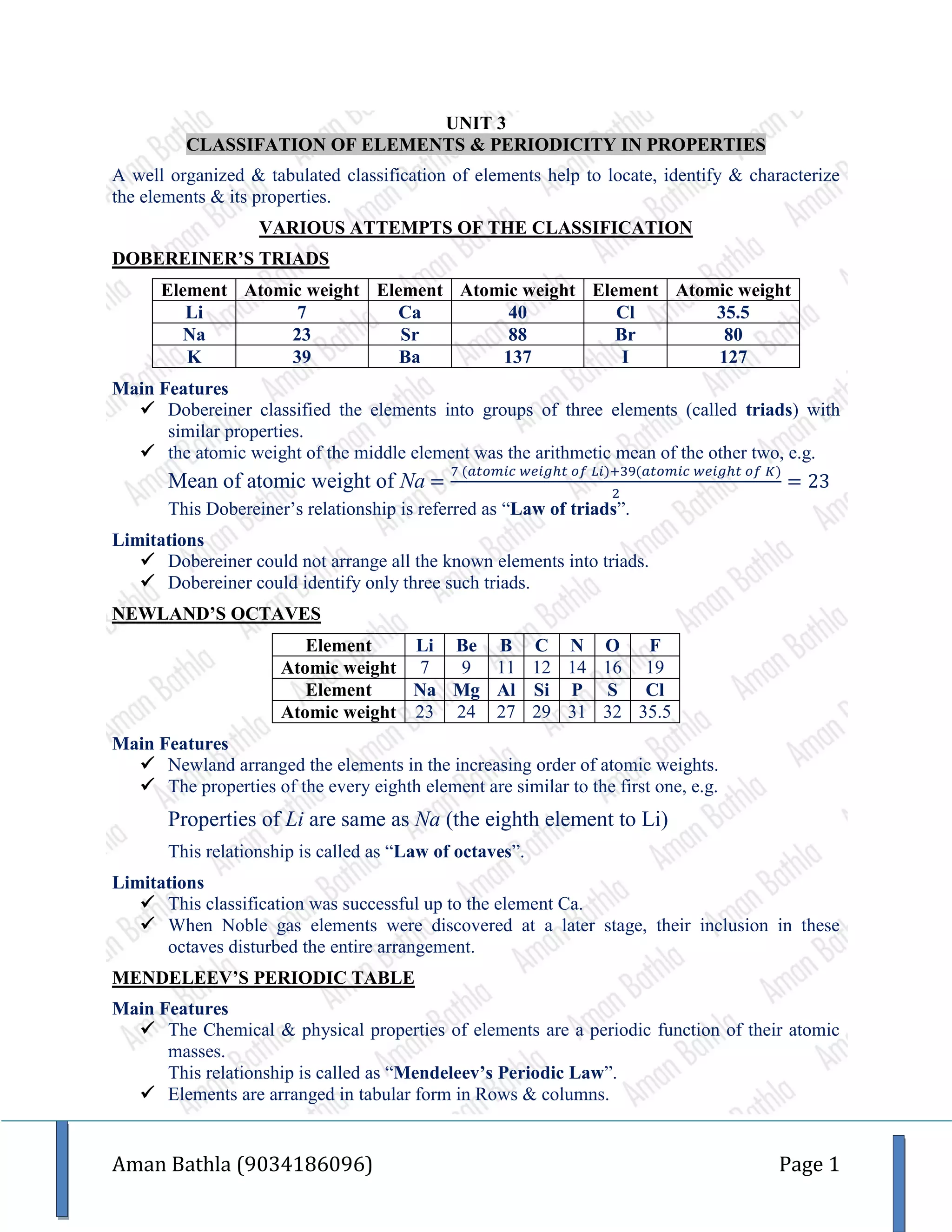
The document describes the classification of elements and periodicity in properties, covering various classification methods including Dobereiner's triads, Newland's octaves, and Mendeleev's periodic table, along with their limitations. It highlights the modern periodic table which is based on atomic numbers and outlines trends such as atomic radii, ionization enthalpy, and electron gain enthalpy across periods and groups. Additionally, it categorizes elements into metals, non-metals, and metalloids, while explaining periodic trends and the IUPAC nomenclature for elements with atomic numbers above 100.


![Aman Bathla (9034186096) Page 5
e.g. The position of an element 1𝑠2
2𝑠2
2𝑝6
3𝑠2
3𝑝6
4𝑠2
3𝑑7
Principal quantum number for the valance shell is 4, so the element belong to Period No. 4
Last electron has been filled in 3d sub-shell, so the element belongs to d-block.
For d-block, group no. equal to 2 + 7=9
2. Locating the position of an element in the periodic table by Atomic number.
Period No.
No. of Elements
Present
Atomic Number
range
First 2 1-2
Second 8 3 - 10
Third 8 11 - 18
Fourth 18 19 - 36
Fifth 18 37 – 54
Sixth 32 55 – 86*
Seventh 32 87 – 118*
* The elements of atomic number range 58-71 belong to f-block have group no. 3rd
are called
LANTHANOIDS.
* The elements of atomic number range 90-103 belong to f-block have group no. 3rd
are called
ACTIANOIDS.
For the given atomic number, find the Period number by the atomic number range.
For group number, count the no. from the extreme left atomic number as first in the
atomic number range (you can count from the extreme right atomic number also). The no.
at which that atomic number exists is the group number of that element.
Remember: The element with atomic number 2 has group number 18.
The element with atomic number 5 to 10 or 13 to 18 has group number
equal to the number we have counted from extreme left atomic number plus 10.
3. Find the electronic configuration of an element in the periodic table by the position.
[ 𝑋] 𝑛𝑙 𝑥
Firstly find the position of the element.
The period to which the element belongs indicates the value of principal quantum number
(n) of the valance shell.
The block to which the elements belong indicates the value of l.
The noble gas before the element of which the electronic configuration to be find
indicates [X].
The x can be identified by the group number as group has same outer most shell
configuration.
Group No. Elect. Conf. Group No. Elect. Conf. Group No. Elect. Conf.
1 ns1
7 ns2
nd5
13 ns2
np1
2 ns2
8 ns2
nd6
14 ns2
np2
3 ns2
nd1
9 ns2
nd7
15 ns2
np3
4 ns2
nd2
10 ns2
nd8
16 ns2
np4
5 ns2
nd3
11 ns1
nd10
17 ns2
np5
6 ns1
nd5
12 ns2
nd10
18 ns2
np6](https://image.slidesharecdn.com/chem11unit3-180614074616/85/Classification-of-elements-and-periodicity-in-properties-5-320.jpg)






