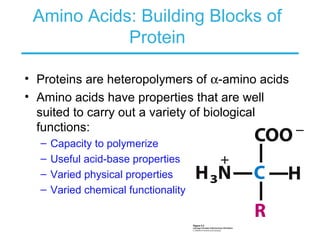
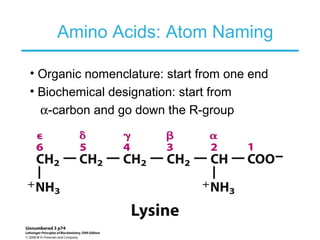
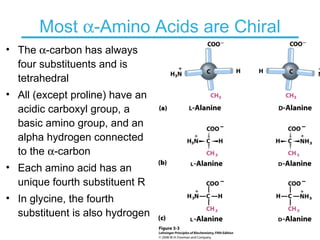
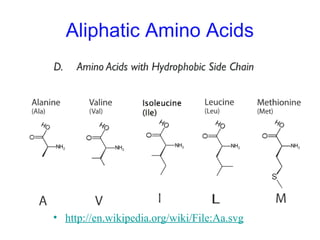
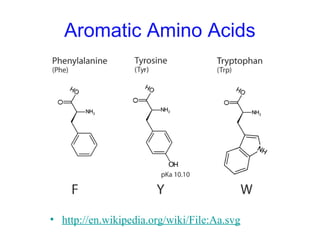
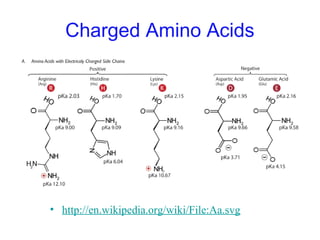
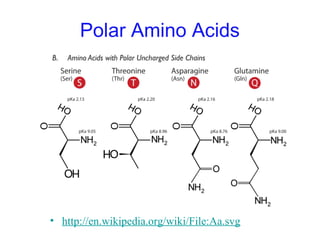
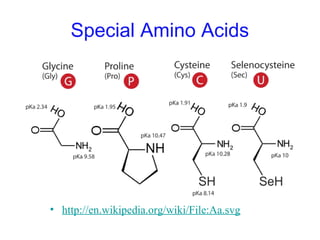
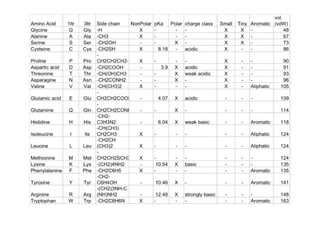
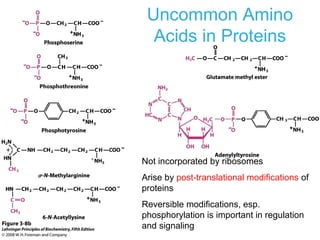
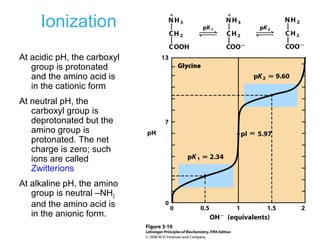
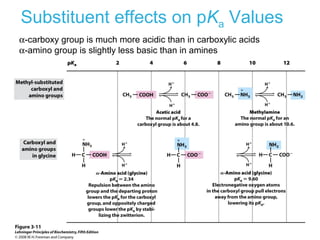
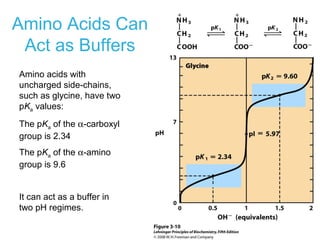
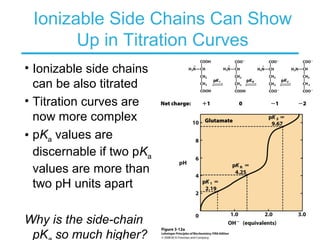
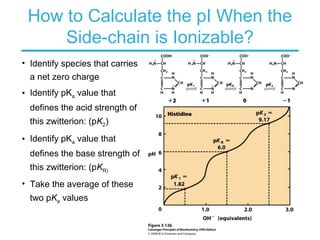
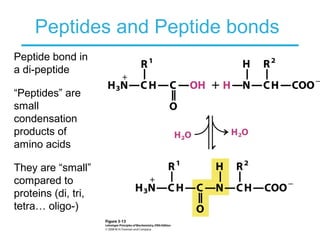
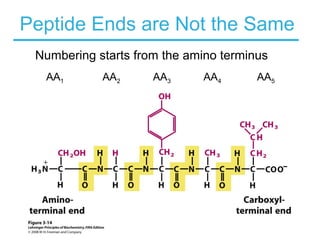
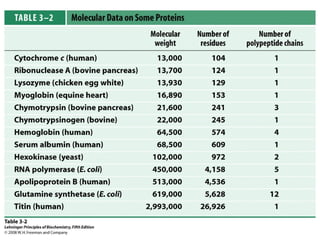
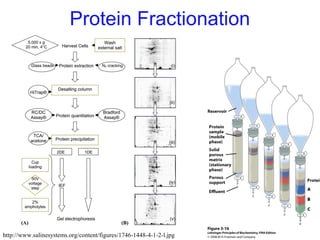
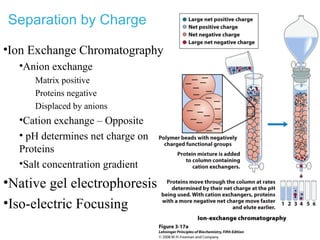
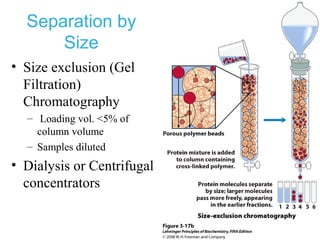
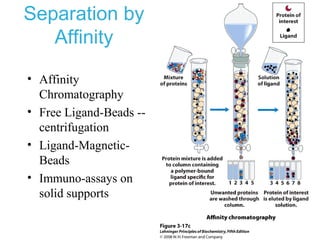
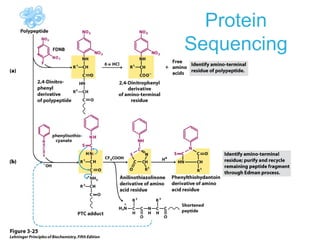
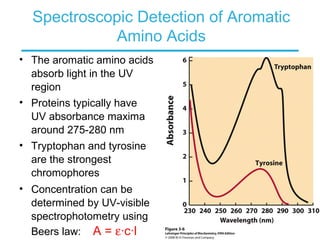
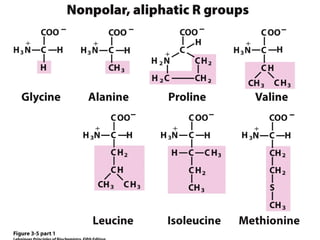
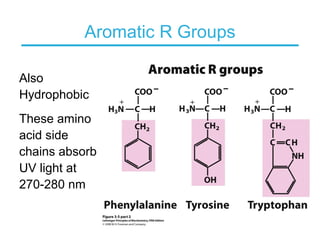
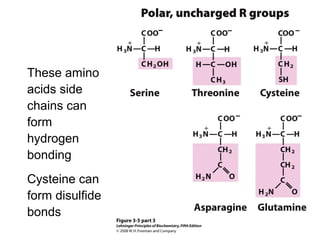
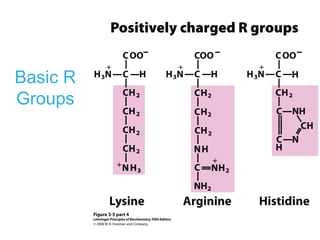
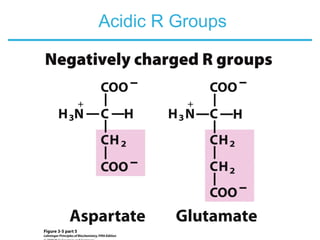
This chapter discusses amino acids, peptides, and proteins. It covers the structure and properties of amino acids, how they combine to form peptides and proteins, and the various functions of proteins, including catalysis, transport, structure, and motion. Methods for analyzing, separating, and purifying proteins are also described, such as ion exchange chromatography, size exclusion chromatography, electrophoresis, and protein sequencing. Finally, the chapter classifies common amino acids based on the properties of their variable side chains.