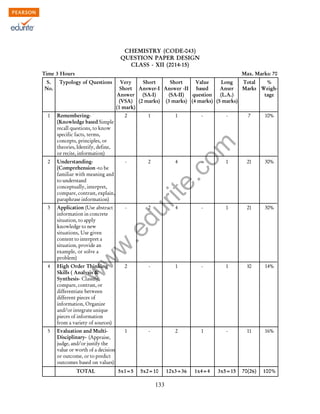
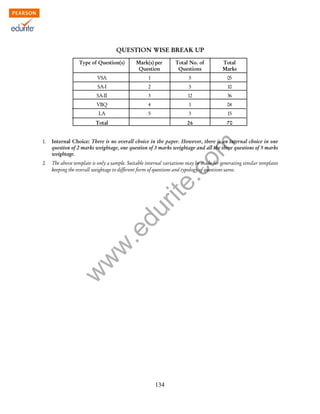
This document provides details about the syllabus and exam structure for Class XII Chemistry in the academic year 2014-2015. It is divided into 16 units covering topics in inorganic, organic, physical and analytical chemistry. The theory exam will be of 70 marks with 3 hours duration. Practical exams will involve 30 marks for experiments covering surface chemistry, chemical kinetics, thermochemistry, electrochemistry, chromatography, preparation of compounds and qualitative analysis. The document also provides a sample question paper format dividing the questions into different cognitive levels and typologies.