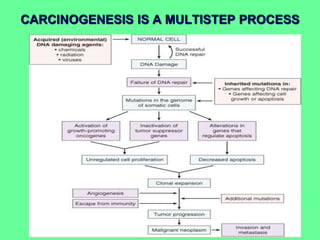
Cancer develops through a multi-step process involving genetic mutations that disrupt the normal cell cycle and allow uncontrolled cell growth. The cell cycle is regulated by various proto-oncogenes and tumor suppressor genes. Mutations in these genes, such as activating mutations in proto-oncogenes or loss of function mutations in tumor suppressor genes, can cause cells to ignore growth controls and proliferate unchecked. This can eventually lead to the development of malignant tumors. Common tumor suppressor genes include RB1, TP53, BRCA1, and BRCA2, which are involved in processes like cell cycle regulation and DNA repair. Mutations in these genes increase cancer risk.