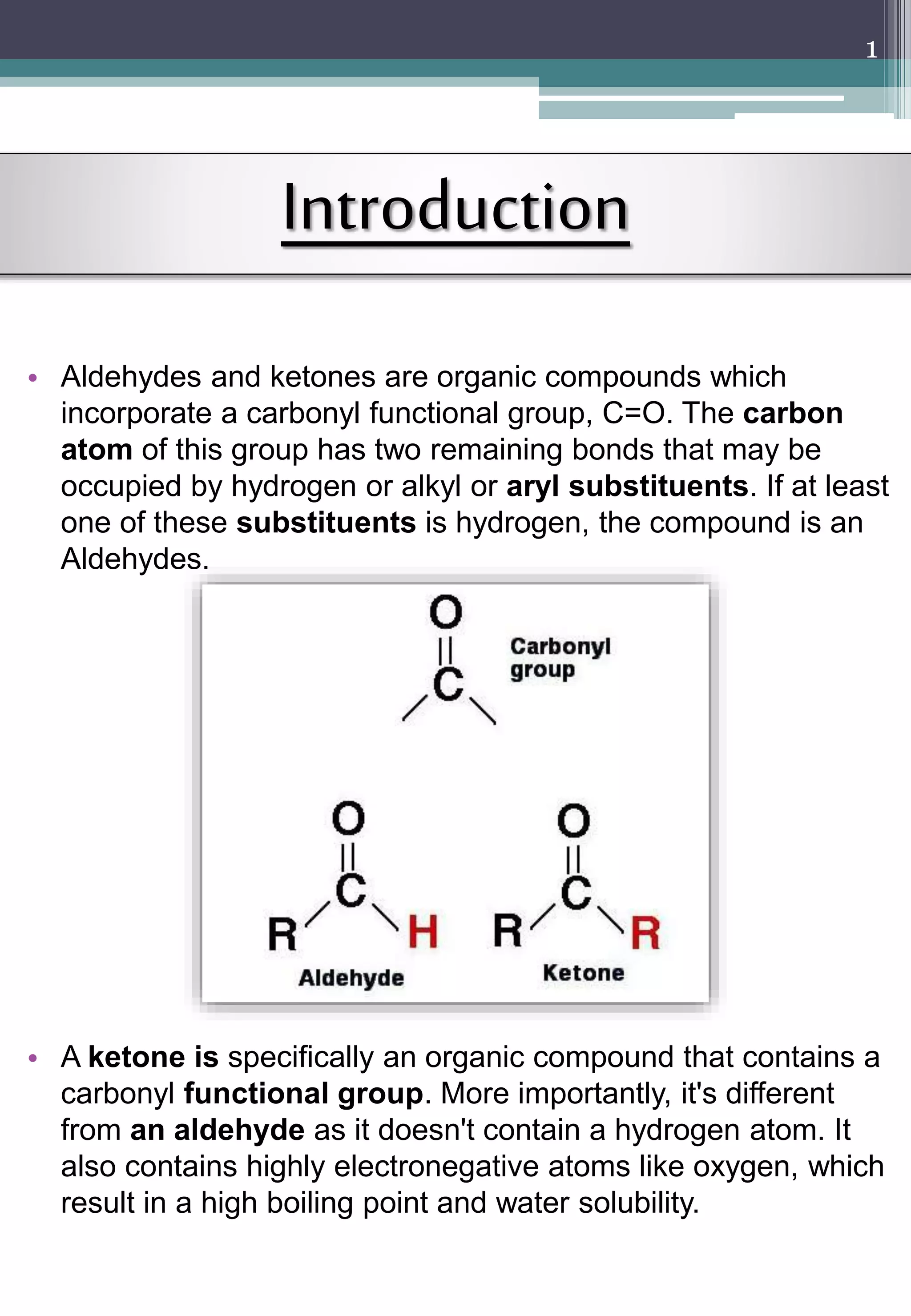
This document is a chemistry project on aldehydes, ketones, and carboxylic acids. It includes a certificate verifying completion, acknowledgements, index, and 5 sections discussing topics like nomenclature, preparation methods, reactions, and inductive effects. The project was assigned by a teacher and completed by a 12th grade student to fulfill an academic requirement. It provides an overview of key concepts regarding these functional groups in an educational format.