1) The document describes the canonical partition function for an ideal monatomic gas. It shows that the partition function can be broken into translational, electronic, and nuclear contributions which are independent.
2) Key relationships are developed between the partition function and thermodynamic properties like internal energy, entropy, heat capacity, and more. Knowing the partition function provides expressions for properties like the Helmholtz free energy.
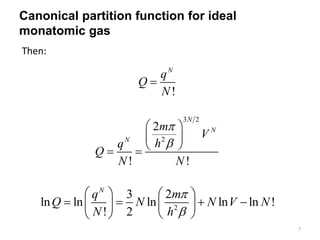
3) For an ideal monatomic gas, the translational partition function is derived and shown to be proportional to volume to the power of 3/2. The electronic and nuclear contributions are accounted for but not explicitly derived.













































![Energy fluctuations in the canonical ensemble
47
d = [E/(3NkT/2)]-1](https://image.slidesharecdn.com/canonicalpartitionfunctionparameters-230105161441-4851ec75/85/Canonical-Partition-Function-Parameters-ppt-47-320.jpg)



