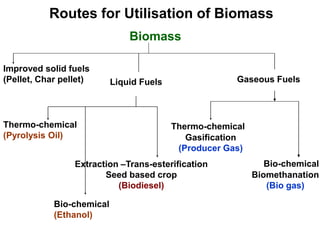
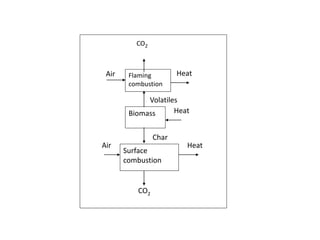
The document discusses effective utilization of biomass through various processing and conversion techniques. It describes that biomass can be processed physically through activities like dewatering, drying and sizing or chemically by converting it into char, liquids or gaseous fuels. The key conversion techniques discussed are thermochemical processes like combustion, gasification and pyrolysis and biochemical processes like biogasification and fermentation. Specific thermochemical gasification techniques using different gasifying agents are also explained.




























![EQUIVALENCE RATIO: [weight of oxidant/weight of dry fuel ]
----------------------------------------------
[stoichiometric oxidant /fuel ]
ER ≥ 1 ---- combustion
ER < 1 ---- gasification/ pyrolysis
ER = 0 ---- pyrolysis
SPECIFIC GASIFICATION RATE: Amount of gas generated per unit area
per unit time
Or amount of biomass gasified per unit area per
unit time
Higher for down draft gasifiers in order to achieve good cracking of tar](https://image.slidesharecdn.com/biomassutilization-230922074648-a81f1eea/85/Biomass-Utilization-ppt-31-320.jpg)





