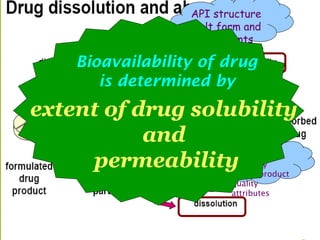
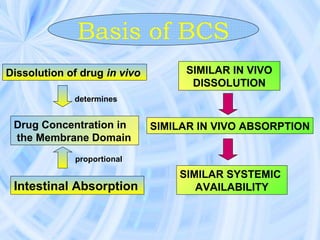
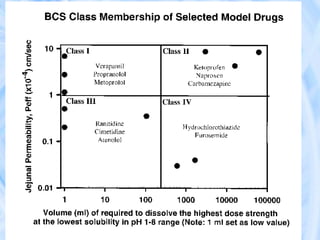
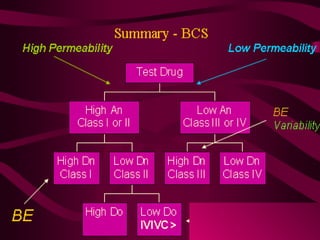
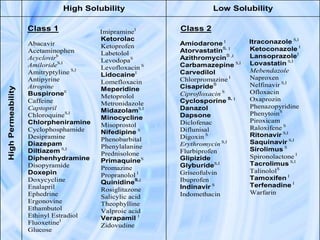
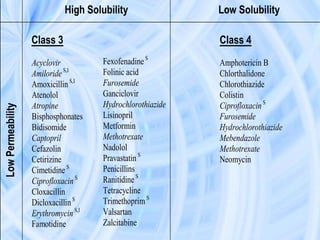
The document provides an overview of the Biopharmaceutics Classification System (BCS), which classifies drug substances based on their aqueous solubility and intestinal permeability. The BCS aims to predict a drug's absorption based on these characteristics. It defines four classes of drugs. Class 1 drugs are highly soluble and permeable, while Class 4 drugs are poorly soluble and permeable. The BCS guidance provided by the FDA can help determine if in vivo bioequivalence studies are needed for new drug products. It provides a framework for biowaivers and for developing formulations to improve solubility or permeability depending on a drug's class.