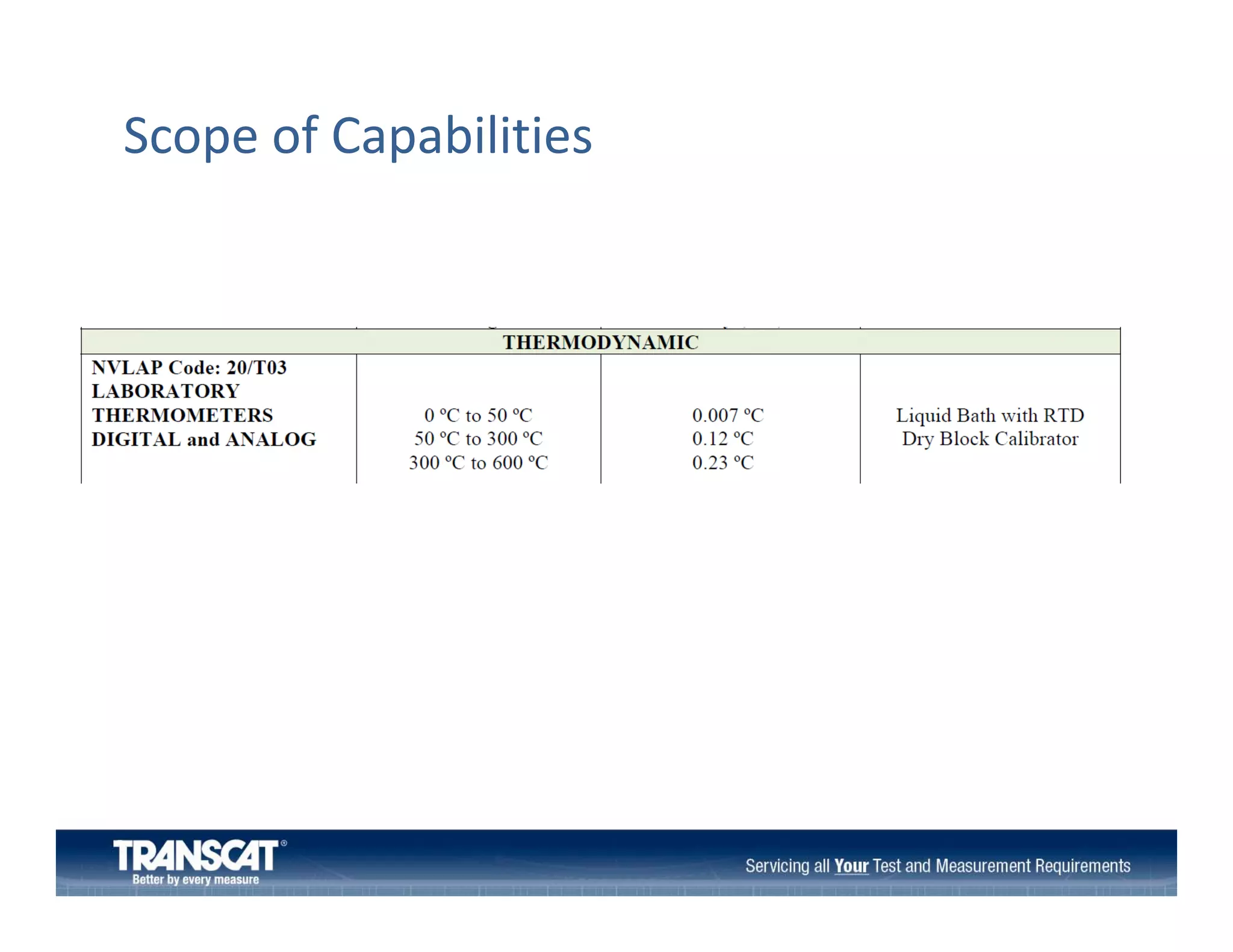
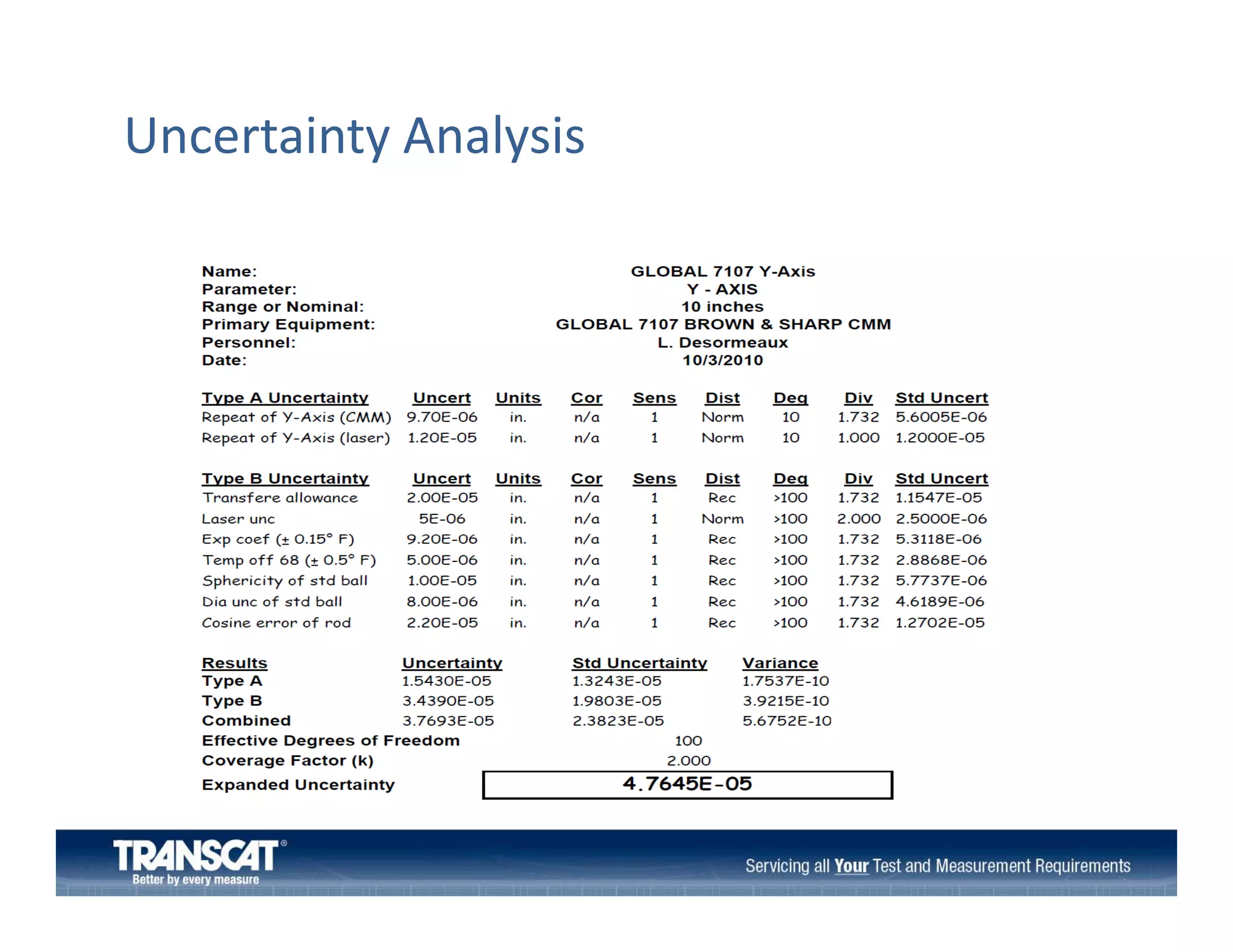
The document outlines the preparation process for calibration laboratory audits, detailing various standards and requirements, particularly ISO/IEC 17025. It emphasizes the importance of competent personnel, quality systems, measurement traceability, and proper handling of measurement and test equipment. Additionally, the document includes guidance on conducting audits, do's and don'ts, and the significance of developing a clear scope of capabilities.