atomic structure chemistry tsh .pptx
•Download as PPTX, PDF•
0 likes•6 views
atomic structure
Report
Share
Report
Share
Recommended
More Related Content
Similar to atomic structure chemistry tsh .pptx
Similar to atomic structure chemistry tsh .pptx (20)
More from thesciencehubprg
More from thesciencehubprg (20)
Recently uploaded
https://app.box.com/s/71kthbth9ww0fyjrppmh1p2gasinqj5zĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...

ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...Nguyen Thanh Tu Collection
https://app.box.com/s/h5mhqoyabotgw05s0df0ltw3e39pgnmyTỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...Nguyen Thanh Tu Collection
Recently uploaded (20)
Personalisation of Education by AI and Big Data - Lourdes Guàrdia

Personalisation of Education by AI and Big Data - Lourdes Guàrdia
Including Mental Health Support in Project Delivery, 14 May.pdf

Including Mental Health Support in Project Delivery, 14 May.pdf
ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...

ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...
Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room

Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room
Basic Civil Engineering notes on Transportation Engineering & Modes of Transport

Basic Civil Engineering notes on Transportation Engineering & Modes of Transport
Analyzing and resolving a communication crisis in Dhaka textiles LTD.pptx

Analyzing and resolving a communication crisis in Dhaka textiles LTD.pptx
The Liver & Gallbladder (Anatomy & Physiology).pptx

The Liver & Gallbladder (Anatomy & Physiology).pptx
TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...
Spring gala 2024 photo slideshow - Celebrating School-Community Partnerships

Spring gala 2024 photo slideshow - Celebrating School-Community Partnerships
Observing-Correct-Grammar-in-Making-Definitions.pptx

Observing-Correct-Grammar-in-Making-Definitions.pptx
Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...

Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...
DEMONSTRATION LESSON IN ENGLISH 4 MATATAG CURRICULUM

DEMONSTRATION LESSON IN ENGLISH 4 MATATAG CURRICULUM
atomic structure chemistry tsh .pptx
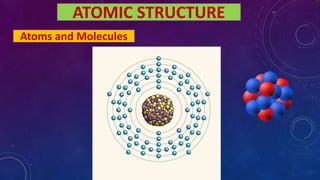
- 1. ATOMIC STRUCTURE Atoms and Molecules
- 3. ATOMIC STRUCTURE DOLTONS ATOMIC THEORY 1)EACH ELEMENT is composed of extremely small particles called atoms. 2)All atoms of a given element are identical. 3)Atoms of different elements possess different properties including atomic masses. 4)Atoms are neither created nor destroyed in chemical reaction. 5)Atoms of elements take part to form molecules.
- 5. ATOMIC STRUCTURE Nucleons and mass number
- 10. ATOMIC STRUCTURE Thomsons atomic model 1) An atom consists of a positively charged sphere and the electrons are embedded in it. 2) The negative and positive charges are equal in magnitude . So the atom as a whole is electrically neutral.
- 11. ATOMIC STRUCTURE Alpha Particle Scattering Experiment
- 12. ATOMIC STRUCTURE
- 13. ATOMIC STRUCTURE
- 14. ATOMIC STRUCTURE
- 15. ATOMIC STRUCTURE
- 17. ATOMIC STRUCTURE
- 19. ATOMIC STRUCTURE