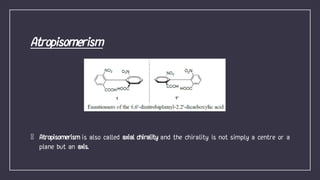
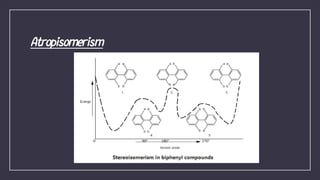
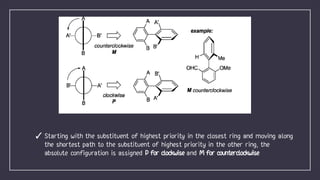
Atropisomerism results from restricted rotation about a single bond, as in biphenyl compounds where large substituents on either side of the central bond cause steric hindrance. This restricted rotation in biphenyls substituted with different groups leads to the formation of chiral atropisomers as enantiomeric pairs. The biphenyl rings are perpendicular to minimize clashes between the ortho substituents, and rotation about the biphenyl bond through the pivotal bond is restricted, demonstrating axial chirality along the biphenyl axis. The stability of atropisomers is determined by factors including the steric demand of substituents near the axis, any bridges present, and mechanisms allowing isomerization beyond physical rotation.