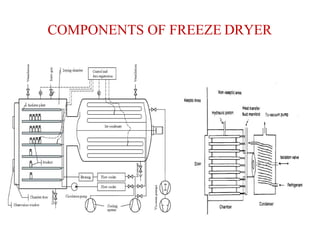
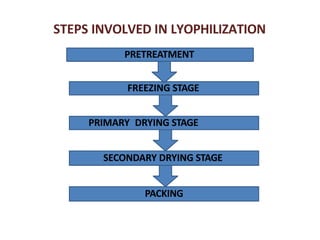
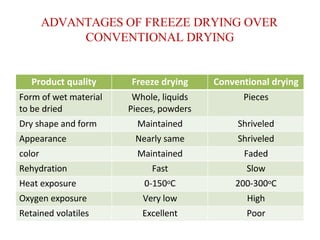
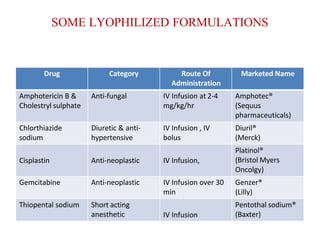
Lyophilization, also known as freeze drying, is a process used to remove water from materials while preserving their structure. It involves freezing the material and then reducing pressure to allow the frozen water to sublimate from the solid to gas phase. The process is carried out below the triple point of water to enable sublimation of ice. Freeze drying has applications in the food and pharmaceutical industries as it can preserve thermolabile compounds and materials by removing water while retaining quality.


























![ Define the following terms:
[Lyophilization, etc]
Respond to the following questions:
Give a detailed account of ………………
Explain in details the process of …………..
Describe in details with examples the…………
With examples, illustrate the pharmaceutical applications of ……………](https://image.slidesharecdn.com/15-lyophilization-200630111026/85/15-lyophilization-31-320.jpg)
