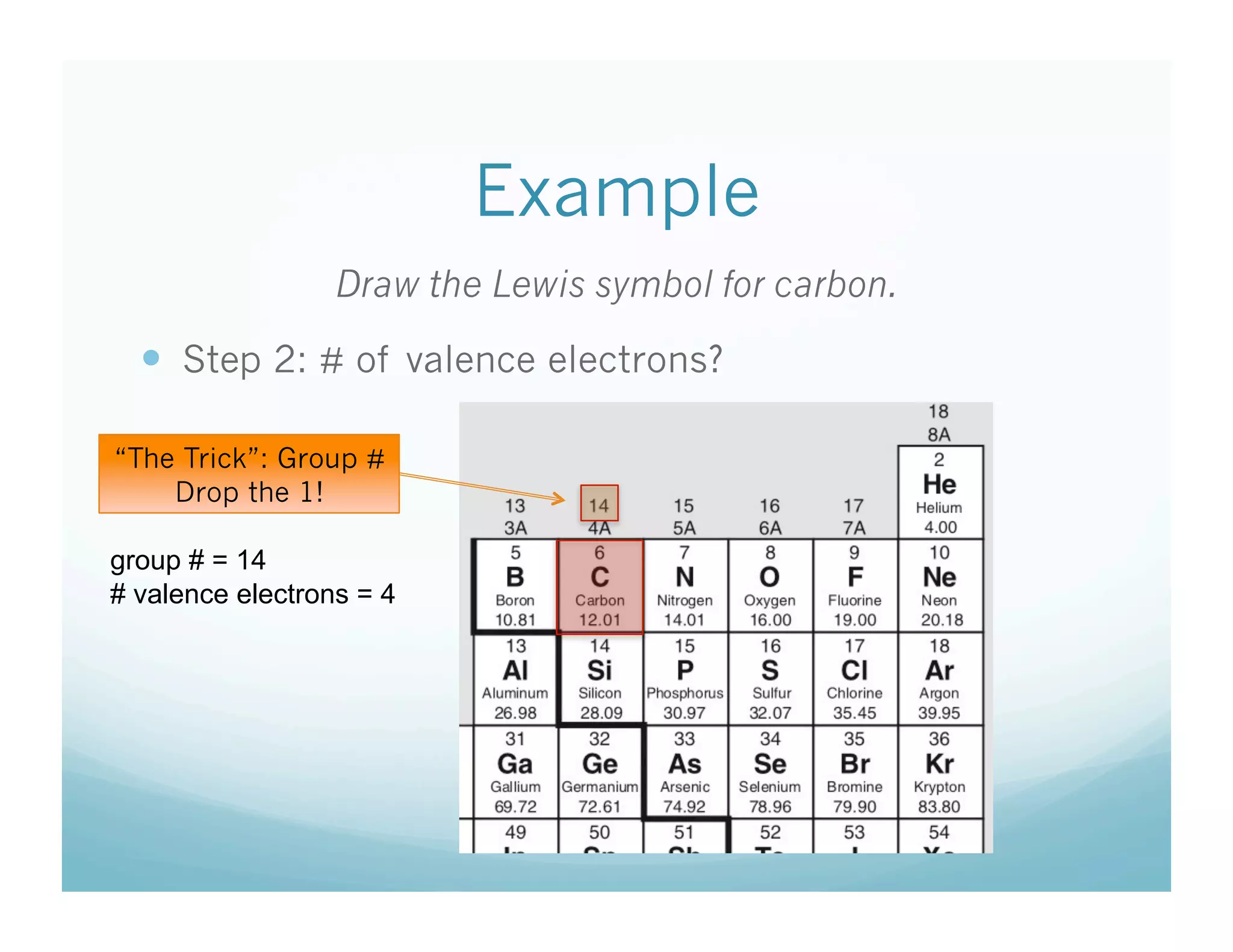
The document provides instructions for students on how to draw Lewis symbols to represent atoms in covalent bonding. It explains that Lewis symbols show the element's symbol in the center and dots around the outside to represent the number of valence electrons, which is determined by the group number on the periodic table. Examples are given to demonstrate how to draw the Lewis symbols for various elements like hydrogen, carbon, sulfur, and others by following the three step process of writing the symbol, determining valence electrons, and placing dots around the outside.