Covalent bonds
•Download as PPTX, PDF•
2 likes•706 views
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot (20)
Viewers also liked
Viewers also liked (13)
Elevate PowerPoint Deck Introduction - by @itseugenec

Elevate PowerPoint Deck Introduction - by @itseugenec
One Point Per Slide – Why It’s Important and How to Do It

One Point Per Slide – Why It’s Important and How to Do It
5 Ways To Surprise Your Audience (and keep their attention)

5 Ways To Surprise Your Audience (and keep their attention)
Similar to Covalent bonds
Similar to Covalent bonds (20)
More from mralfordscience
More from mralfordscience (10)
Recently uploaded
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Recently uploaded (20)
BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...

BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...

IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Measures of Central Tendency: Mean, Median and Mode

Measures of Central Tendency: Mean, Median and Mode
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi

Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi
social pharmacy d-pharm 1st year by Pragati K. Mahajan

social pharmacy d-pharm 1st year by Pragati K. Mahajan
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Covalent bonds
- 2. Covalent bonds form between non-metal atoms that SHARE electrons. Doing so helps them achieve a stable outer electron shell. Covalent bonds are stronger than ionic bonds. Each atom pulls at the others’ electrons.
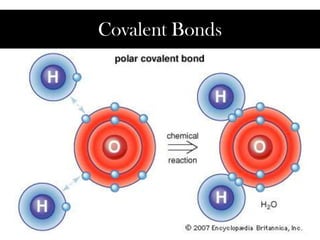
- 4. Polar covalent bonds occur when the two atoms are different. The atoms with more protons has a stronger pull on the electrons, so that atoms becomes partially negative. As a result, the atom with a weaker pull becomes partially positive.
- 7. Naming Covalent Compounds Prefix • Mono- 1 • Di- 2 • Tri- 3 • Tetra- 4 • Penta- 5 • Hexa- 6 • Hepta- 7 • Octa- 8 • Nona- 9 • Deca- 10 • First element in the formula is named first using prefix • Second element follows using # prefix AND suffix ‘-ide’ • Mono is not used for the first element • Example • N2O – Dinitrogen Monoxide • N2O5 – Dinitrogen Pentoxide