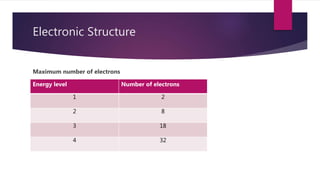
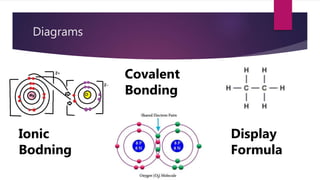
This document discusses electronic structure and bonding. It describes how electrons fill different shells and orbitals based on principal quantum number. Maximum electrons per shell is 2n2. Ionic bonding occurs via electrostatic attraction between cations and anions to form an ionic lattice. Covalent bonding involves orbital overlap between atoms to form shared pairs of electrons and molecules. Bond strength and properties depend on whether bonding is mainly ionic or covalent.