Liquid menbranes
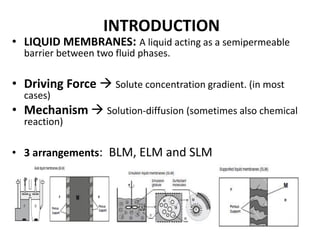
- 1. INTRODUCTION • LIQUID MEMBRANES: A liquid acting as a semipermeable barrier between two fluid phases. • Driving Force Solute concentration gradient. (in most cases) • Mechanism Solution-diffusion (sometimes also chemical reaction) • 3 arrangements: BLM, ELM and SLM
- 2. Classification of liquid membranes according to transport mechanisms
- 3. TRANSFER EQUATIONS Difusion and chemical reaction Common steps • Solution-diffusion (with or without chemical reactions) is a commonly accepted mechanism for the transport of a solute in liquid membrane. • Rates of chemical changes and/or rates of diffusion may control all liquid membrane transport kinetics. • Analysis of mechanisms and kinetics of the chemical and diffusion steps of the overall LM transport system is needed to find the rate-controlling ones. • Diffusion steps in aqueous feed. • Diffusion of the complex solute- carrier in the LM phase. • Partitions between aqueous feed and organic LM phases at feed -LM interface and between LM and aqueous strip phases at LM-strip interface. • Kinetics of chemical interactions with formation of solute-carrier complex and destruction of complex.
- 4. Diffusion transport regime Steady state No steady state• The diffusion flux Js (M, g/cm2s) of the species is defined as the amount of matter passing perpendicularly through the unit area during the unit time • In a steady-state permeation experiment, the flux of a species S through a membrane of thickness h is related to the concentration gradient through Fick’s first law: • When steady state cannot be assumed, the concentration change with time must be considered.
- 5. Diffusion transport regime SLM diffusion coeficient (Dm) Bulk diffusion coefficient (Db) • The diffusional process through a SLM is affected by the porosity and tortuosity of the polymeric support. • Dm has to be corrected for the membrane characteristics to obtain the bulk diffusion coefficient Db. • The bulk diffusion coefficient Db is derived by Stokes-Einstein relationship: and the Wilke-Chung relation:
- 6. Diffusion transport regime Lag-time experiment Permeability experiments • The time required for the complex to diffuse across the membrane from the feed phase to the receiving phase, assuming dilute conditions. • Db=τDlag • At time t=0, a carrier which is substituted with a chromophoric group is added to the feed phase ([cf]=0). The carrier diffuses through the membrane and the increase of concentration in the receiving phase ([cr]t) is monitored by UV/Vis spectroscopy. • Db=Dmτ/ϵ
- 7. Chemical reactions’ kinetics regime transport • When one or more of the chemical reactions are sufficiently slow in comparison with the rate of diffusion to and away from the interfaces. • Two series of chemical reactions mechanisms and their kinetics have to be analyzed: 1. Solute uptake at the aqueous feed phase-organic LM interface or partition and chemical interactions with solvent exchange and formation of solute-carrier complex 2. Solute release with chemical interactions between LM and aqueous strip phases at LM-strip interface with destruction of the complex
- 8. Chemical reactions’ kinetics regime transport Determination of activation energy 1. Irreversible first-order reactions: 2. Reversible first-order reactions: 3. Series of first-order reactions • Activation energy of transport gives information about the rate-limiting step in the transport process. • Ea values below 20 kJ/mol are accepted as indicative of pure diffusion-limited transport. At activation energies above 40 kJ/mol, chemical reactions do play a role in the transport.
- 9. Mixed transport regime • When both chemical reactions and film diffusion processes occur at rates that are comparable, the solvent extraction kinetics are said to take place in a mixed diffusional-kinetic regime. • Unless simplifying assumptions can be used, frequently the differential equations have no analytical solutions, and boundary conditions have to be determined by specific experiments.
- 10. Emulsion Liquid Membrane Design 1. Membrane Film model: film with constant thickness. a) Uniform flat sheet model: (planar geometry). Considers resistance as a sum of resistances through phases. b) Spherical shell model: mass transfer rate directly related to DC across the film. 2. Advancing Front Model: irreversibility of stripping reaction. Mass flux: Flux of solute arriving at Rf: Rate of reagent being consumed: Mass balance for solute in Ep: Mass balance for solute in Rf: 3. Reversible Reaction Model: reversible stripping reaction (No reaction front). Mass Balance for solute concentration in membrane portion and external phase:
- 11. Bulk Liquid Membrane Design 1. Bulk Organic Hybrid Liquid Membranes(BOHLM): Uncharged (hydrophobic) symmetric membranes as barriers. • Overall permeability coefficients on KF/E and KE/R: • Overall mass balance: • Mass transfer coeficient: 2. Bulk Aqueous Hybrid Liquid Membranes: Charged (hydrophilic)symmetric membranes as barriers. • Overall permeability coefficients on KF/E and KE/R: • Model equation: • Overall mass balance:
- 12. Supported Liquid Membrane Design The membrane phase is held by capillary forces in the pores of a microporous polymeric or inorganic film. The inmobilized liquid is the membrane phase and the film serves as a support for the membrane, which separates the feed phase from the strip phase. • Diving force: difference in concentration • Mass transfer takes place due to difference in chemical potential: Variation of Chem.pot: • Flux: where the diffusion coefficient is:
- 13. EQUIPMENT Chemical Composition of Liquid Membranes • Diluents Main components (immiscible with other phases) • Surfactants Specially important in ELM • Carriers / Tranporters Only for Facilitated Transport
- 14. EQUIPMENT Equipment for Emulsion LM (ELM) • Emulsion: colloidal suspension of a liquid in another immiscible liquid.
- 15. EQUIPMENT Equipment for Bulk LM (BLM) • Layered LM • U-tube • Bulk Hybrid LM Supports (solid membranes) • Organic (BHOLM) or Aqueous (BHALM) • Configurations: FS, Sp, Tub, HF
- 16. EQUIPMENT Equipment for Supported LM (SLM) • Support microporous solid membrane • LM inside the pores • Configurations: • FS, Sp, Tub, HF
- 17. APPLICATIONS Applications of Emulsion LM (ELM) • Metal ion separations • Separation of biomolecules • Enzyme immobilization (not for separation processes) (Most of them on laboratory scale)
- 18. Supported Liquid Membranes’ Applications a) Analytical Applications
- 19. b) Applications on the Biotechnological and environmental sciences: c) Separation of stereoisomers:particularly enantiomers
- 20. Bulk Liquid Membranes’ Applications • Metal separation • Wastewater treatment • Biotechnologies • Drugs recovery-separation • Organic compounds separation • Gas separation
