Report
Share
Download to read offline
Recommended
More Related Content
What's hot
What's hot (20)
Chemical bonding and different types of interactions

Chemical bonding and different types of interactions
Similar to Lesson 3 - chemical bonding
Similar to Lesson 3 - chemical bonding (20)
Chemical bonding xi , dr.mona srivastava , founder masterchemclasses

Chemical bonding xi , dr.mona srivastava , founder masterchemclasses
Conditions for Formation of Ionic and Covalent Bonds

Conditions for Formation of Ionic and Covalent Bonds
Recently uploaded
https://app.box.com/s/m9ehjx5owsaox9ykvb9qg3csa8a0jnoxTỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...Nguyen Thanh Tu Collection
Recently uploaded (20)
dusjagr & nano talk on open tools for agriculture research and learning

dusjagr & nano talk on open tools for agriculture research and learning
21st_Century_Skills_Framework_Final_Presentation_2.pptx

21st_Century_Skills_Framework_Final_Presentation_2.pptx
Introduction to TechSoup’s Digital Marketing Services and Use Cases

Introduction to TechSoup’s Digital Marketing Services and Use Cases
TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...
When Quality Assurance Meets Innovation in Higher Education - Report launch w...

When Quality Assurance Meets Innovation in Higher Education - Report launch w...
HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx

HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx
NO1 Top Black Magic Specialist In Lahore Black magic In Pakistan Kala Ilam Ex...

NO1 Top Black Magic Specialist In Lahore Black magic In Pakistan Kala Ilam Ex...
Including Mental Health Support in Project Delivery, 14 May.pdf

Including Mental Health Support in Project Delivery, 14 May.pdf
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Lesson 3 - chemical bonding
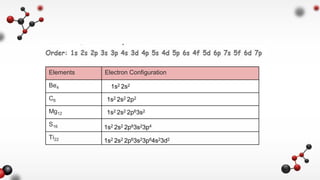
- 1. Elements Electron Configuration Be4 C6 Mg12 S16 Ti22 1s2 2s2 1s2 2s2 2p2 1s2 2s2 2p63s2 1s2 2s2 2p63s23p4 1s2 2s2 2p63s23p64s23d2
- 2. 1. P 15 2. Co 27 3. Ru 44 4. Kr 36 5. Sr 38
- 4. 02. What are the types of Chemical Bond? 03. How are ionic and covalent compounds different? 01. What is a Chemical bond? 04. Practice exercise
- 5. What is a chemical bond? 01. CHEMICAL BOND
- 6. CHEMICAL BOND --- is a force that holds of two or more atoms together and makes them function as a unit. -- is the physical phenomenon of chemical substances being held together by the attraction of atoms to each other through sharing, as well as exchanging of electrons or electrostatic force.
- 7. What causes this forces? -- bonds are formed when constituent atoms come close enough together such that the outer electrons of one atom are attracted to the positive nuclear charge of its neighbor.
- 8. 02. TYPES OF CHEMICAL BOND What are the different types of chemical bond and how are each type formed?
- 9. The type of bonding is determined by how the outermost electrons of an atom, the so-called valence electrons interact with neighboring atoms.
- 10. The position of an element in the periodic table determines the type of bonds it makes.
- 12. IONIC BOND -- results from the transfer of electrons from one element to another Types of Chemical Bond COVALENT BOND -- results from the sharing of electrons between two atoms
- 13. IONIC BOND -- form between a metal and nonmetal. Types of Chemical Bond COVALENT BOND -- formed when two nonmetals combine; also formed when a metalloid bond to a nonmetal.
- 14. IONIC BOND When sodium and chlorine react to form sodium chloride, electrons are transferred from the sodium atoms to the chlorine atoms to form Na- and Cl- ions,, which the aggregate to form solid sodium chloride.
- 15. Calcium Chloride CaCl2 Ca would have a charge of 2+ since it lost two electrons. Each Cl ion would have a charge of 1- since each gained an electron. A subscript “2” is used to show that two ions were used in the bond
- 16. Magnesium Chloride MgCl2 Mg would have a charge of 2+ since it lost two electrons. Each Cl ion would have a charge of 1- since each gained an electron. A subscript “2” is used to show that two ions were used in the bond