Embed presentation
Downloaded 59 times





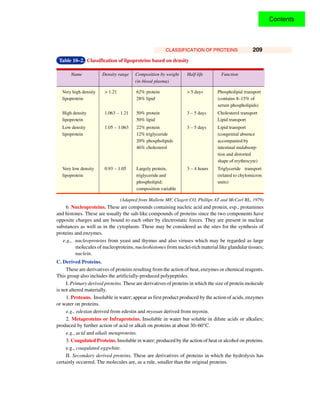




This document discusses different ways of classifying proteins. It describes 4 main classification systems: 1) Based on the source as either animal or plant proteins, 2) Based on shape as either globular or fibrous, 3) Based on composition and solubility into simple, conjugated and derived proteins, and 4) Based on biological function. It provides examples of different protein types that fall under each classification system.









