Solubility melting point of organic practical
•
3 likes•1,118 views
Familiarity with the solubility and melting points of some common organic solids is one of the basic aspect of practical organic chemistry classes at high school as well as undergraduate level.
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Estimation of sodium bicarbonate and carbonate in mixture

Estimation of sodium bicarbonate and carbonate in mixture
Lect. 8. application of nernst distribution law copy - copy

Lect. 8. application of nernst distribution law copy - copy
Determination of alkalinity of a given mixture of hydroxide and carbonate 

Determination of alkalinity of a given mixture of hydroxide and carbonate
Detection and confirmation test for functional group

Detection and confirmation test for functional group
Report 1 prepare and standardize a 0.1 M NaOH solutions

Report 1 prepare and standardize a 0.1 M NaOH solutions
Similar to Solubility melting point of organic practical
Similar to Solubility melting point of organic practical (20)
Phenols: methods of preparation, chemical reaction

Phenols: methods of preparation, chemical reaction
non aqueous titration, pharmaceutical and cosmetic analysis,Pca seminar

non aqueous titration, pharmaceutical and cosmetic analysis,Pca seminar
Determining Intermolecular Forces-Solubility for Functional Groups Fun.docx

Determining Intermolecular Forces-Solubility for Functional Groups Fun.docx
Acid base concepts Acid Base concepts; Arrhenius, Lowery-Bronsted, Lewis. So...

Acid base concepts Acid Base concepts; Arrhenius, Lowery-Bronsted, Lewis. So...
Recently uploaded
Recently uploaded (20)
GEOLOGICAL FIELD REPORT On Kaptai Rangamati Road-Cut Section.pdf

GEOLOGICAL FIELD REPORT On Kaptai Rangamati Road-Cut Section.pdf
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
Gliese 12 b: A Temperate Earth-sized Planet at 12 pc Ideal for Atmospheric Tr...

Gliese 12 b: A Temperate Earth-sized Planet at 12 pc Ideal for Atmospheric Tr...
In silico drugs analogue design: novobiocin analogues.pptx

In silico drugs analogue design: novobiocin analogues.pptx
National Biodiversity protection initiatives and Convention on Biological Di...

National Biodiversity protection initiatives and Convention on Biological Di...
The importance of continents, oceans and plate tectonics for the evolution of...

The importance of continents, oceans and plate tectonics for the evolution of...
Topography and sediments of the floor of the Bay of Bengal

Topography and sediments of the floor of the Bay of Bengal
THE IMPORTANCE OF MARTIAN ATMOSPHERE SAMPLE RETURN.

THE IMPORTANCE OF MARTIAN ATMOSPHERE SAMPLE RETURN.
Gliese 12 b, a temperate Earth-sized planet at 12 parsecs discovered with TES...

Gliese 12 b, a temperate Earth-sized planet at 12 parsecs discovered with TES...
biotech-regenration of plants, pharmaceutical applications.pptx

biotech-regenration of plants, pharmaceutical applications.pptx
Solubility melting point of organic practical
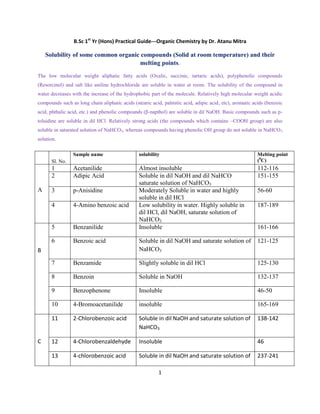
- 1. 1 B.Sc 1st Yr (Hons) Practical Guide---Organic Chemistry by Dr. Atanu Mitra Solubility of some common organic compounds (Solid at room temperature) and their melting points. The low molecular weight aliphatic fatty acids (Oxalic, succinic, tartaric acids), polyphenolic compounds (Resorcinol) and salt like aniline hydrochloride are soluble in water at room. The solubility of the compound in water decreases with the increase of the hydrophobic part of the molecule. Relatively high molecular weight acidic compounds such as long chain aliphatic acids (stearic acid, palmitic acid, adipic acid, etc), aromatic acids (benzoic acid, phthalic acid, etc.) and phenolic compounds (β-napthol) are soluble in dil NaOH. Basic compounds such as p- toluidine are soluble in dil HCl. Relatively strong acids (the compounds which contains –COOH group) are also soluble in saturated solution of NaHCO3, whereas compounds having phenolic OH group do not soluble in NaHCO3 solution. Sl. No. Sample name solubility Melting point (0 C) A 1 Acetanilide Almost insoluble 112-116 2 Adipic Acid Soluble in dil NaOH and dil NaHCO saturate solution of NaHCO3 151-155 3 p-Anisidine Moderately Soluble in water and highly soluble in dil HCl 56-60 4 4-Amino benzoic acid Low solubility in water. Highly soluble in dil HCl, dil NaOH, saturate solution of NaHCO3 187-189 B 5 Benzanilide Insoluble 161-166 6 Benzoic acid Soluble in dil NaOH and saturate solution of NaHCO3 121-125 7 Benzamide Slightly soluble in dil HCl 125-130 8 Benzoin Soluble in NaOH 132-137 9 Benzophenone Insoluble 46-50 10 4-Bromoacetanilide insoluble 165-169 C 11 2-Chlorobenzoic acid Soluble in dil NaOH and saturate solution of NaHCO3 138-142 12 4-Chlorobenzaldehyde Insoluble 46 13 4-chlorobenzoic acid Soluble in dil NaOH and saturate solution of 237-241
- 2. 2 NaHCO3 14 4-Chloroaniline Soluble in dil HCl 67-72 15 Cinnamic acid Soluble in dil NaOH and saturate solution of NaHCO3 132-135 N 16 2-Napthol(-napthol) Soluble in dil NaOH 121-123 17 4-Nitrophenol Soluble in water (due to presence of polar group and also intermolecular hydrogen bonding is possible). 111-116 18 3-Nitro benzoic acid Soluble in water, but solubility is low. Highly soluble in dil NaOH and saturate solution of NaHCO3 139-142 19 4-Nitro benzoic acid Soluble in water, but solubility is low. Highly soluble in dil NaOH and saturate solution of NaHCO3 237-242 20 4-Nitroaniline Low solubility in water. Soluble in dil HCl 146-150 21 3-Nitro aniline Soluble in dil HCl 109-114 22 2-Nitro aniline Soluble in dil HCl 70-74 23 4-Nitroacetanilide Insoluble 213-216 O 24 Oxallic acid Soluble in water 186-191 P 25 Phthalic acid soluble in dil NaOH and saturate solution of NaHCO3 205-210 (it decomposes) 26 Phenylacetic acid soluble in dil NaOH and saturate solution of NaHCO3 76-79 R 27 Resorcinol Soluble in water 110-113 S 28 Succinic acid Soluble in water 184-189 29 Salicylic acid Soluble in dil NaOH and saturate solution of 159
- 3. 3 NaHCO3 30 Salicylamide Soluble in dil HCl and dil NaOH 139-144 T 31 p-Toluidine Soluble in dil HCl 42-46 (low melting, careful)
