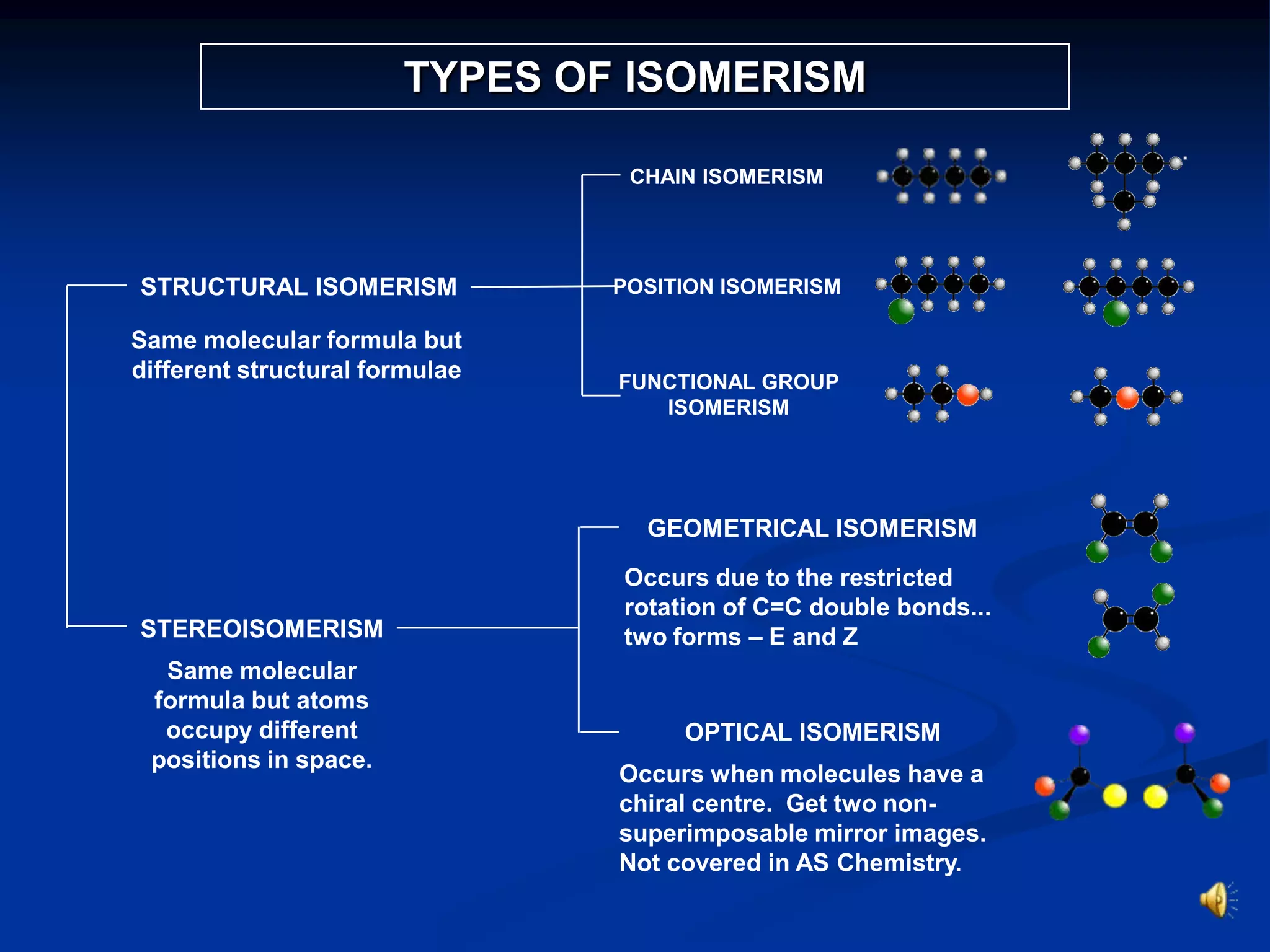
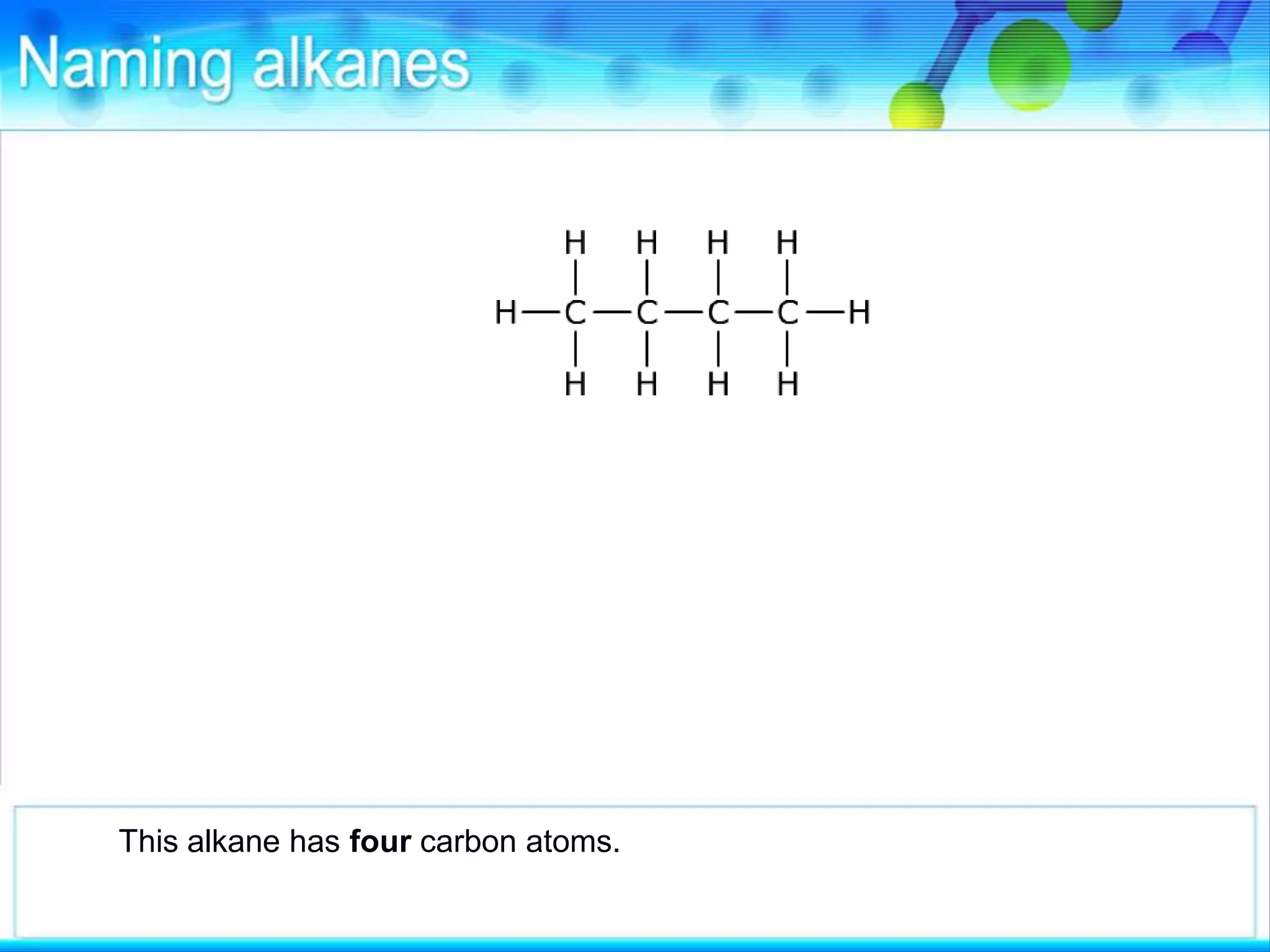
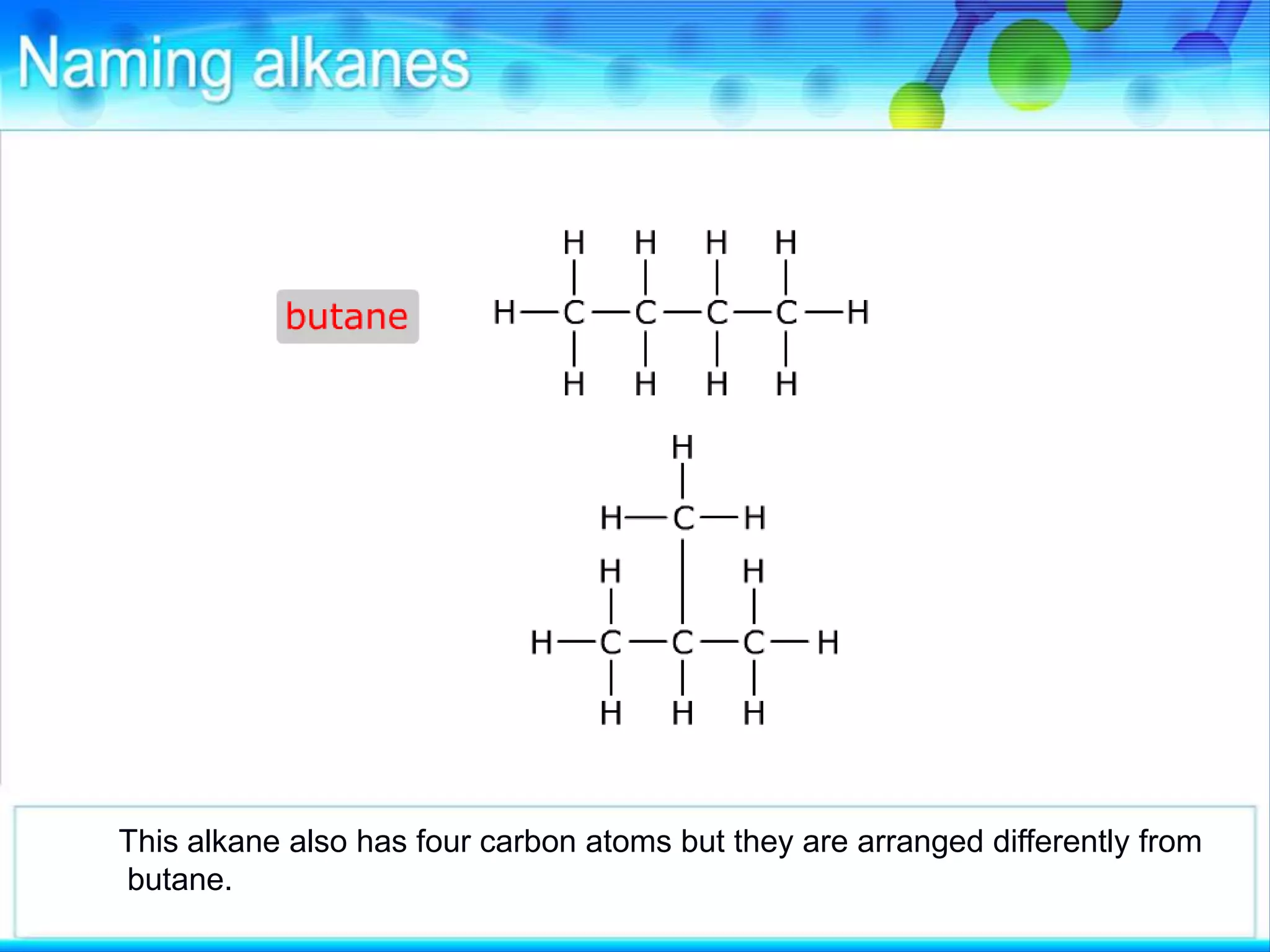
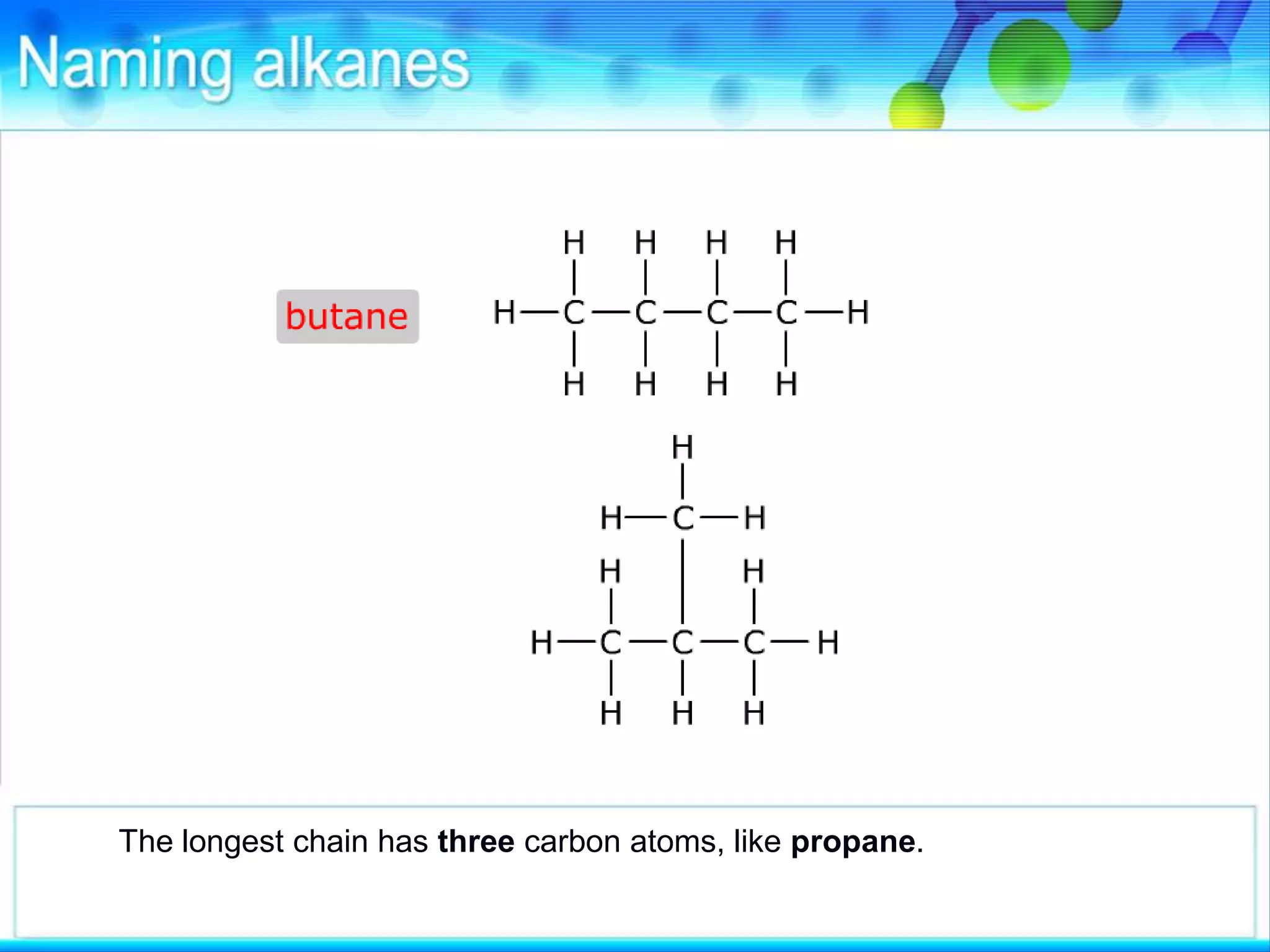
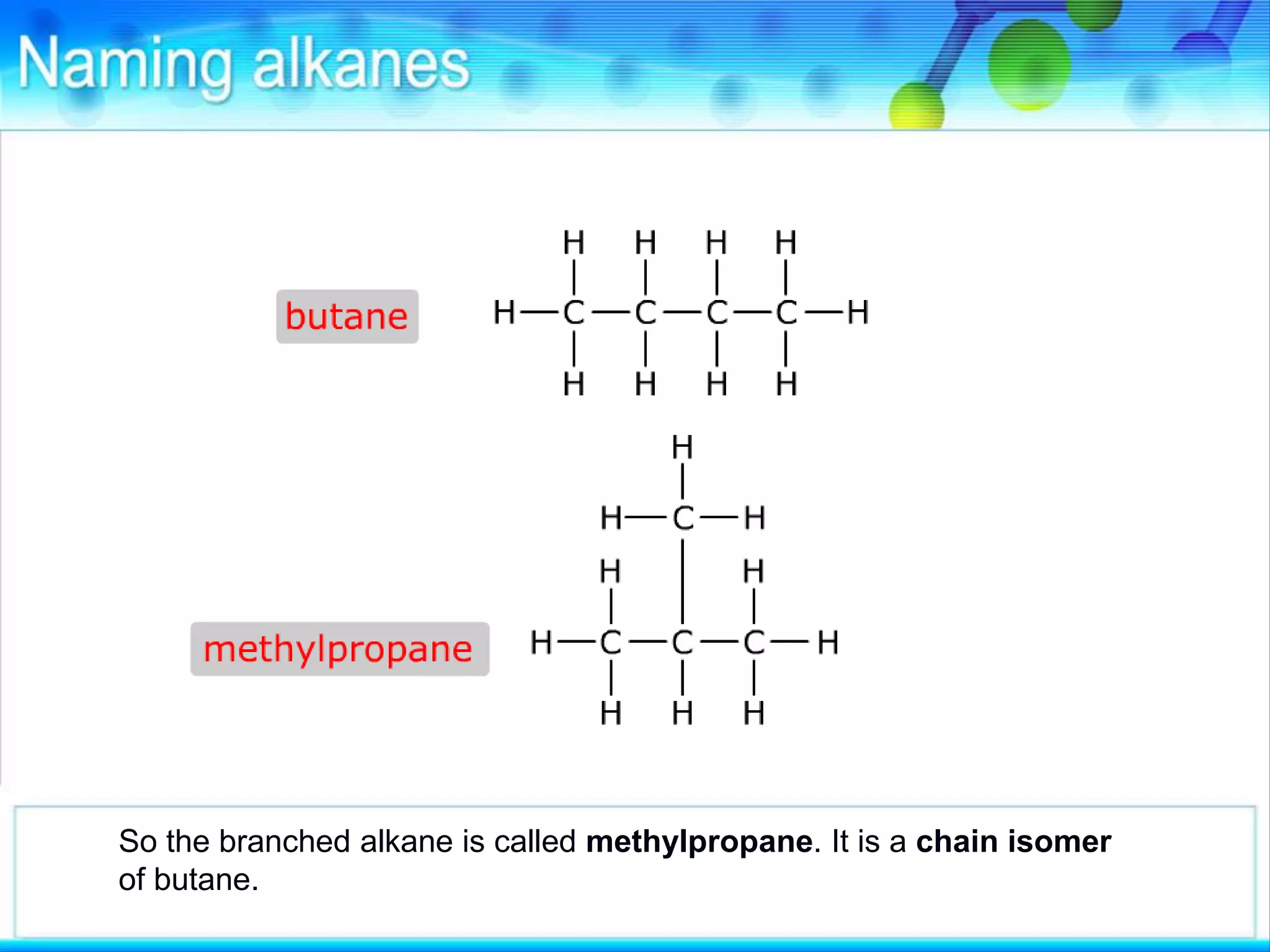
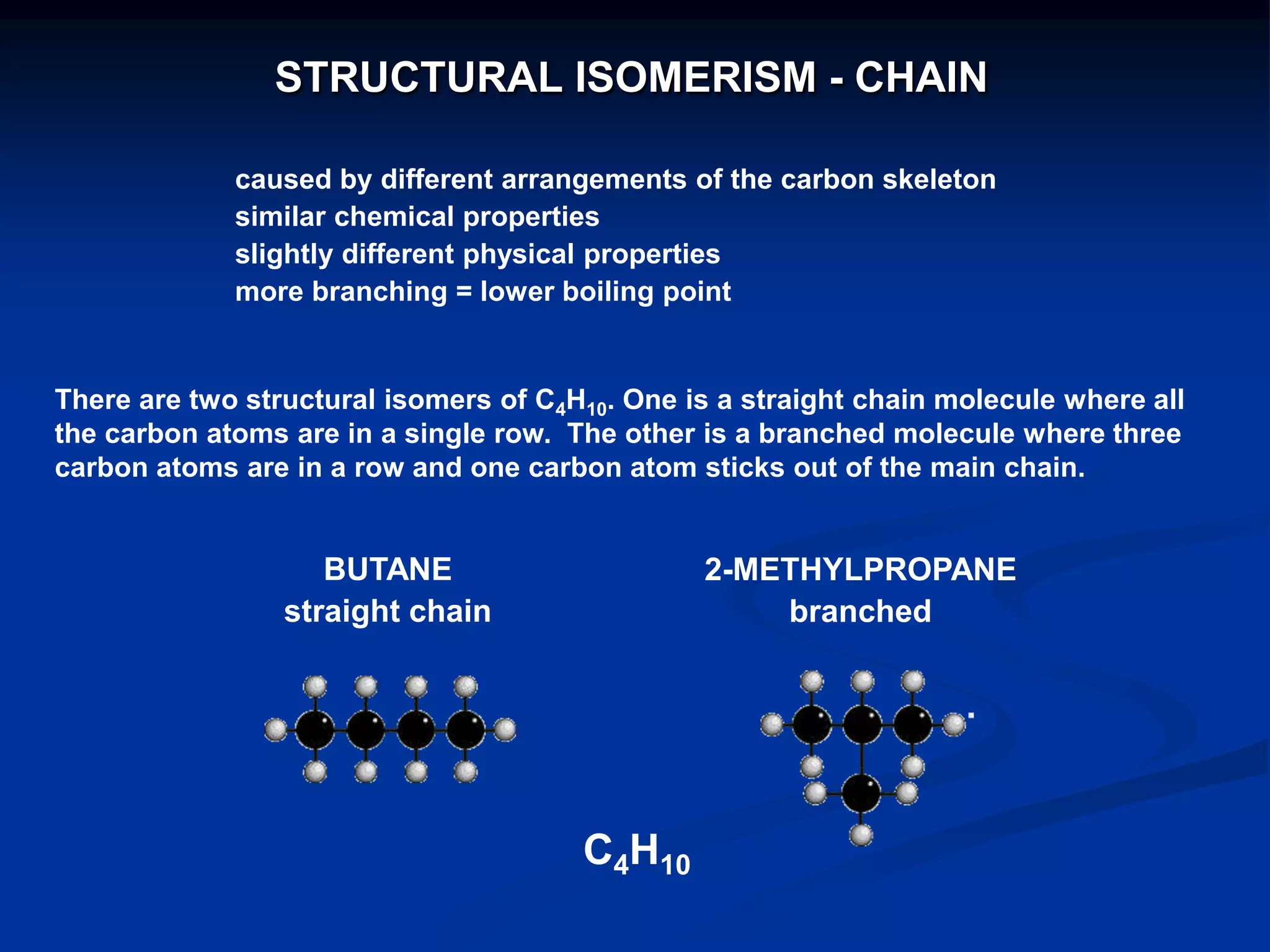
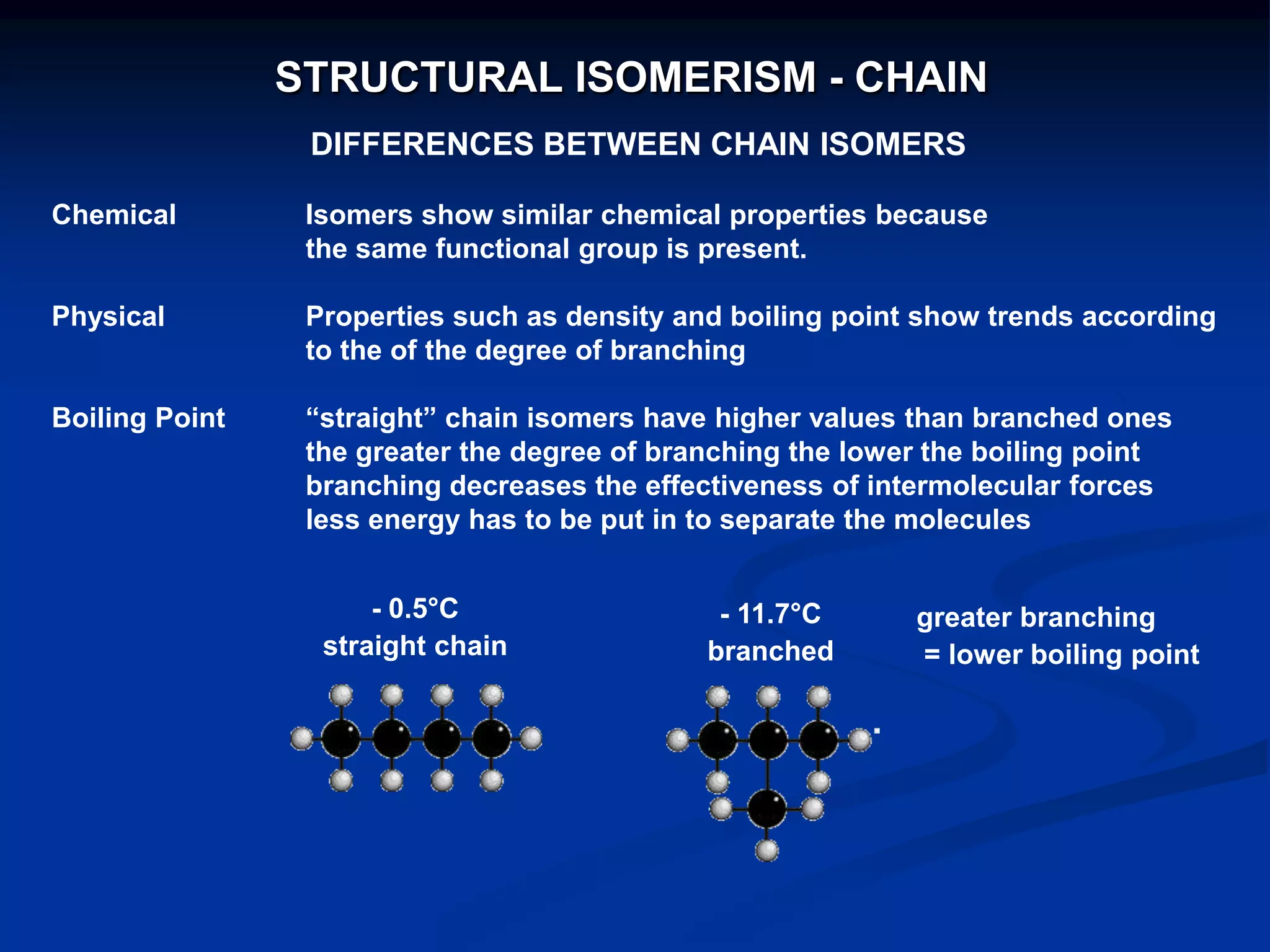
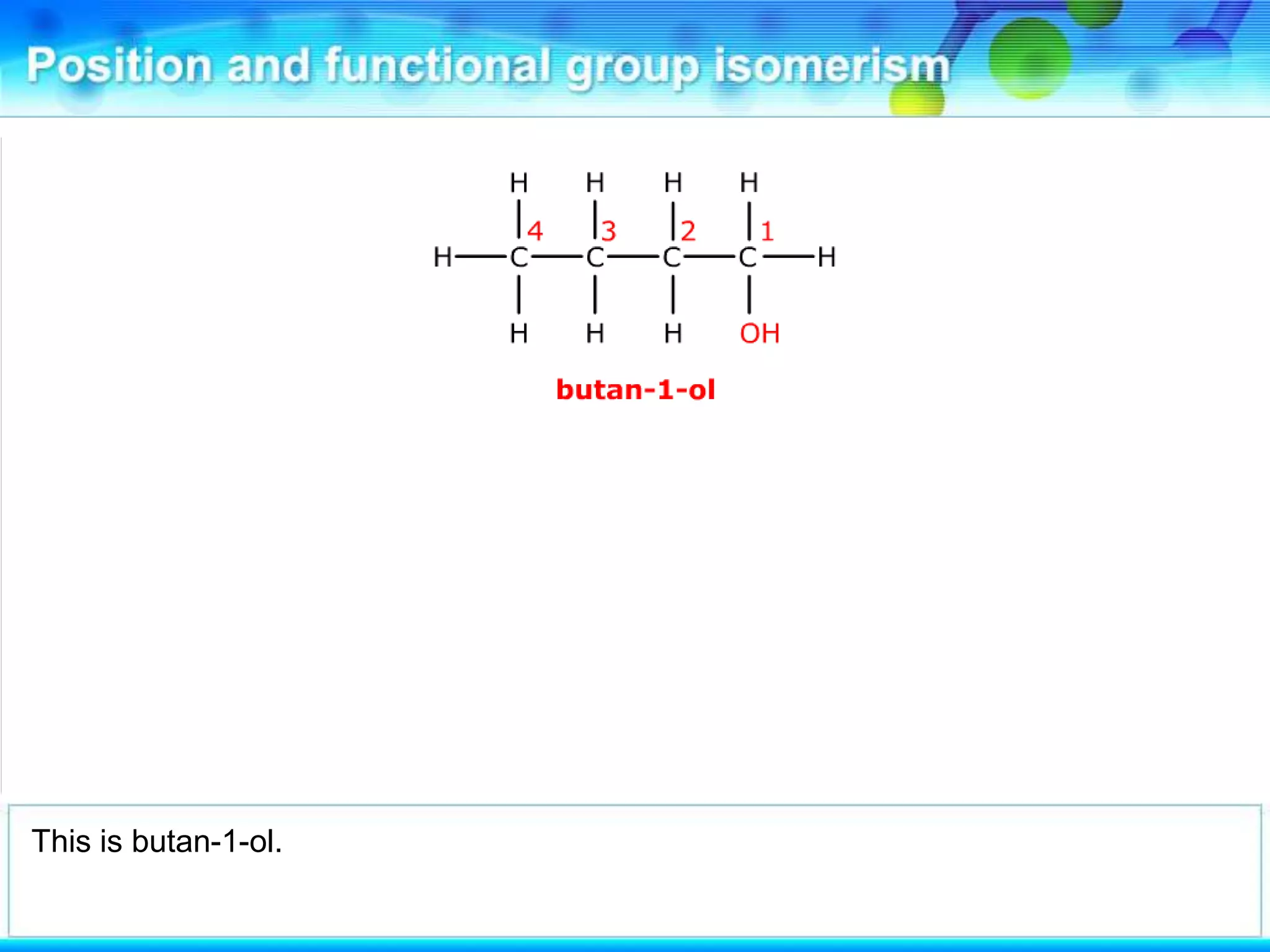
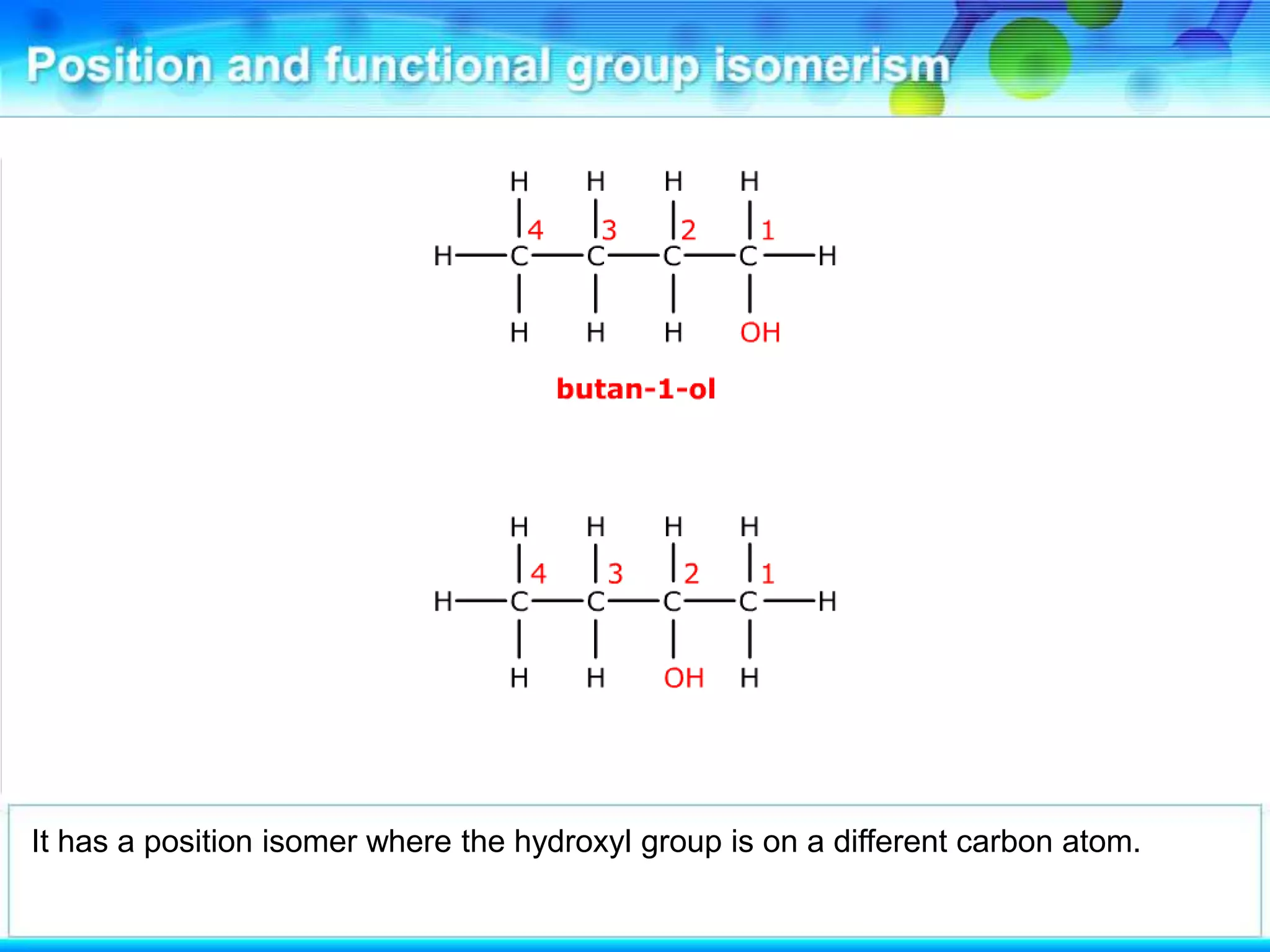
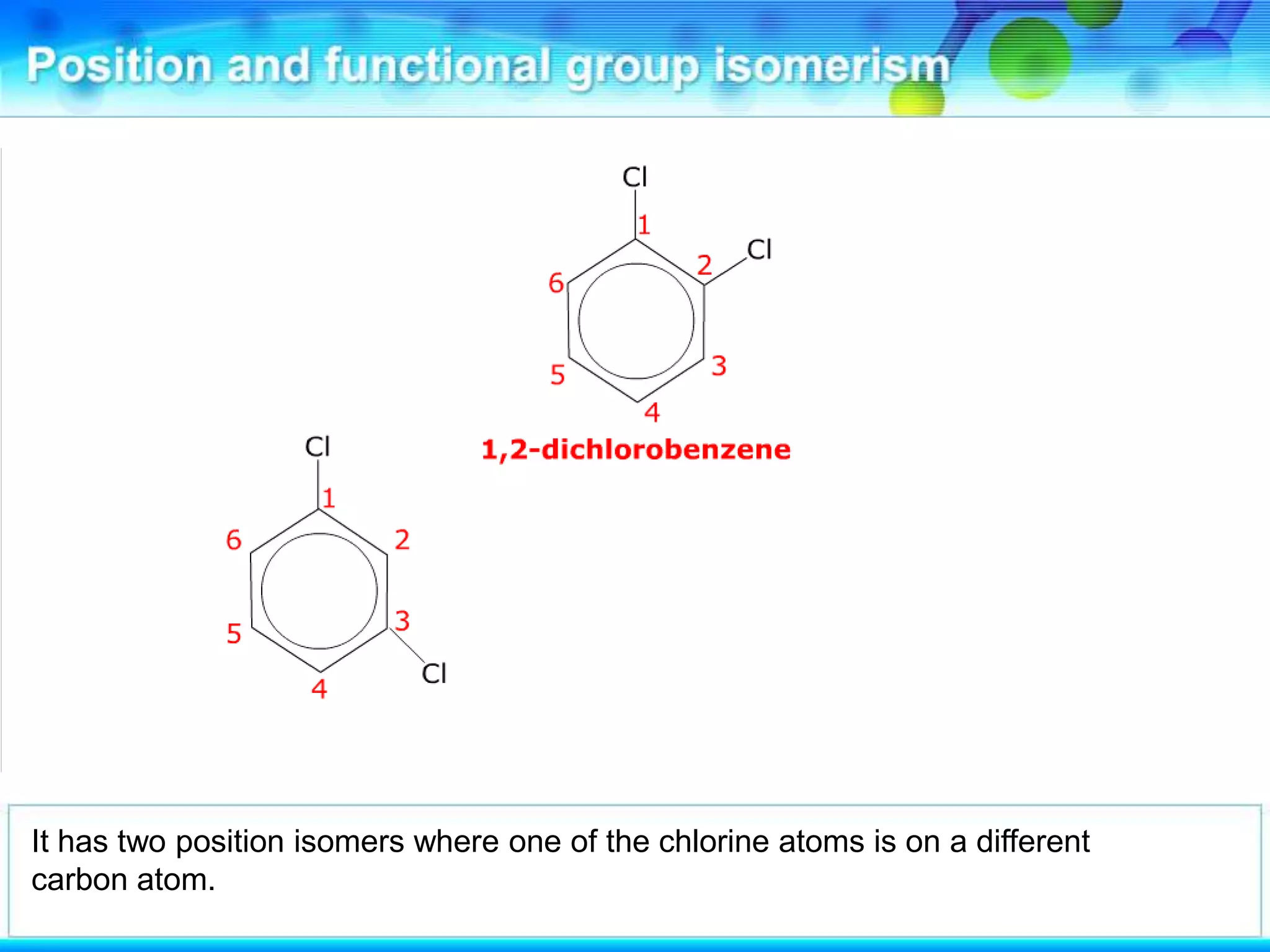
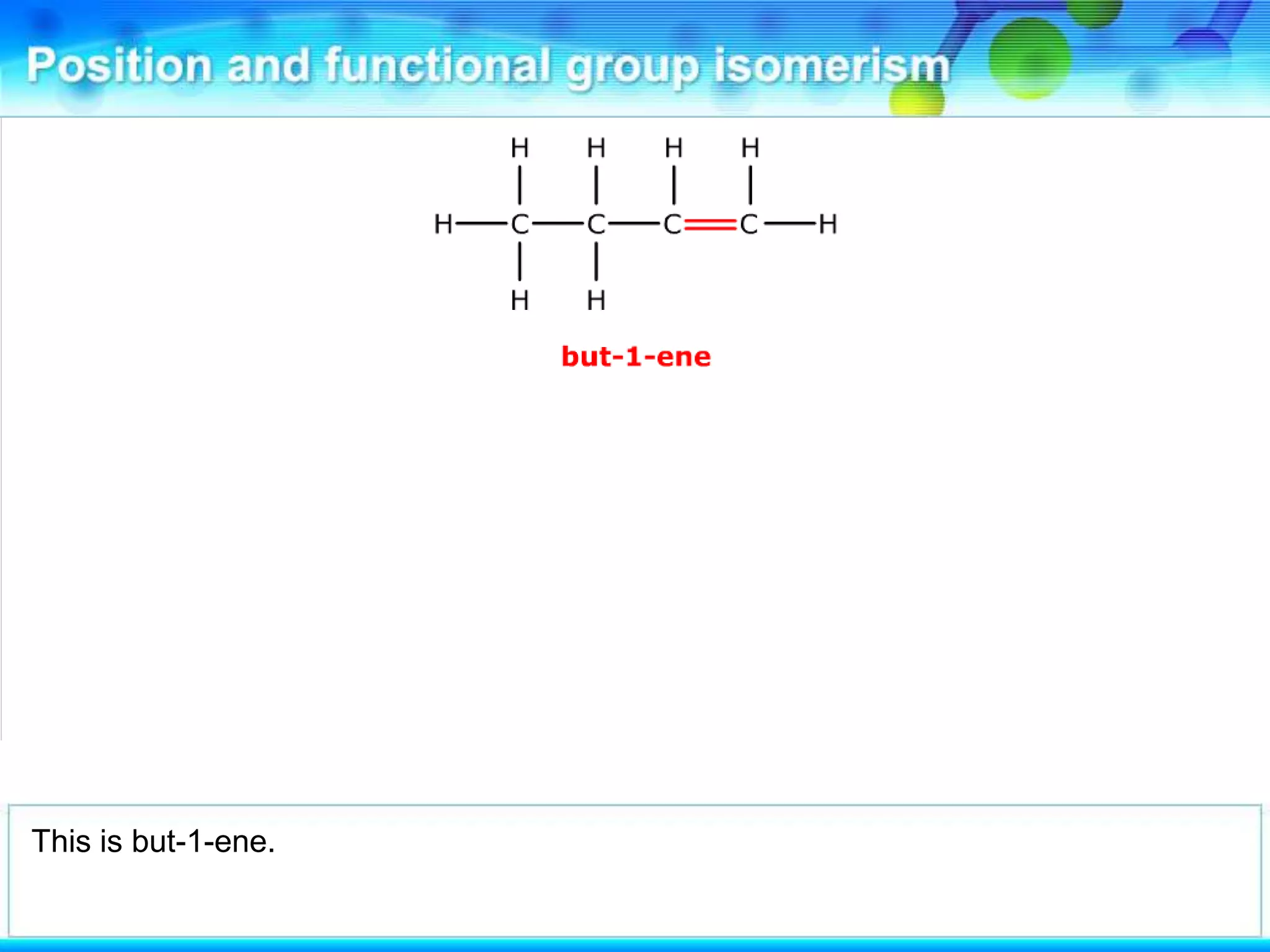
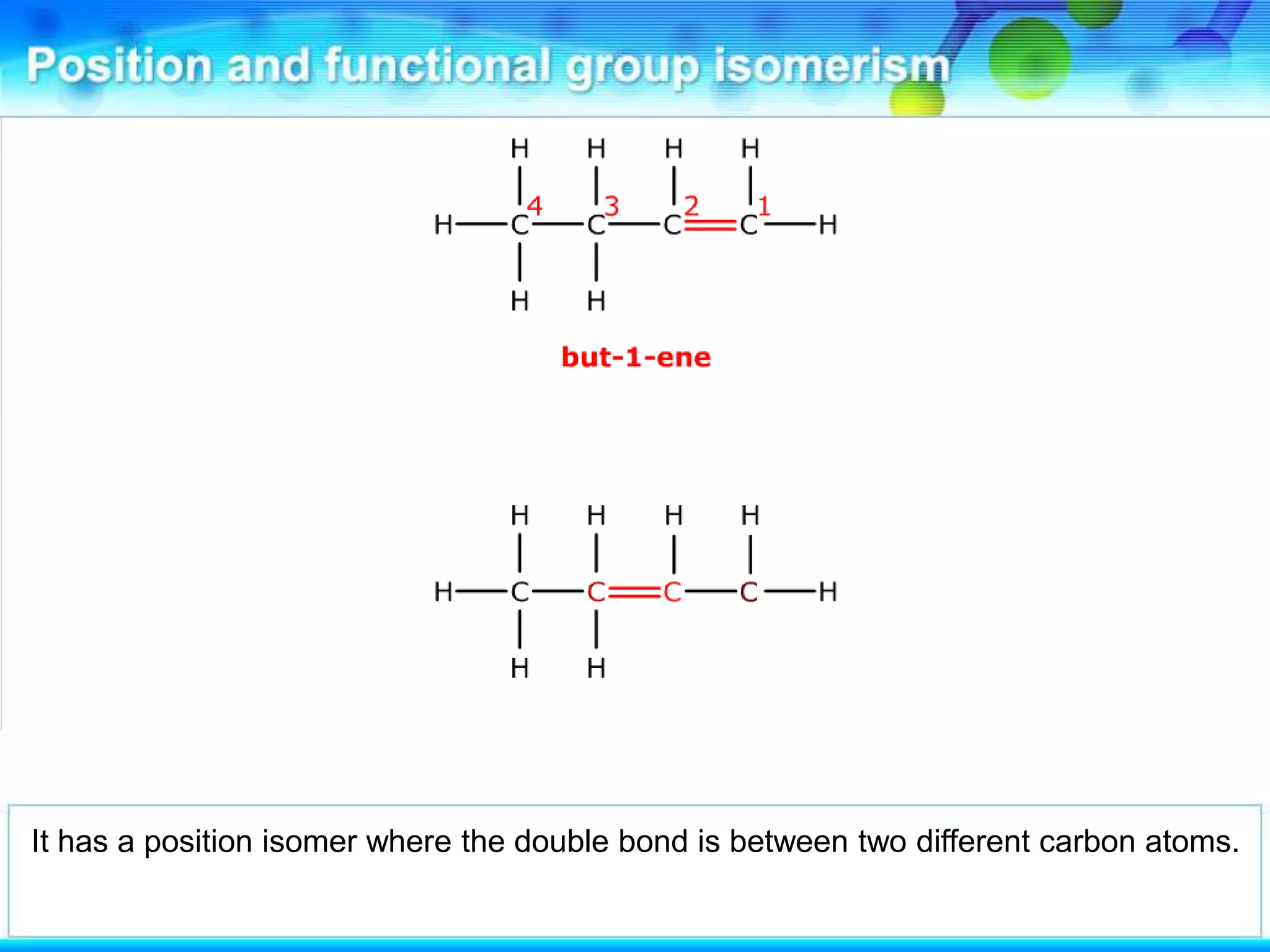
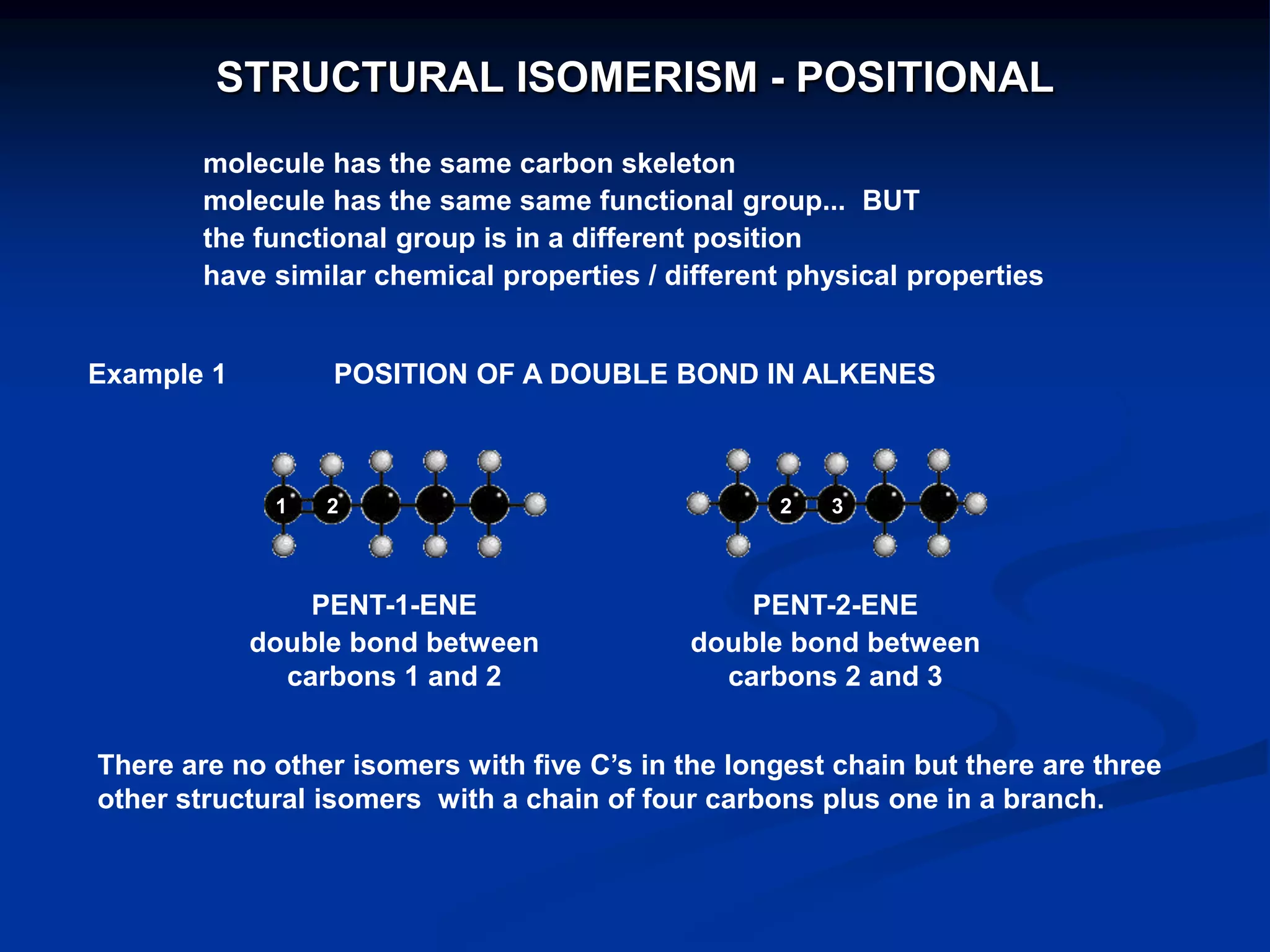
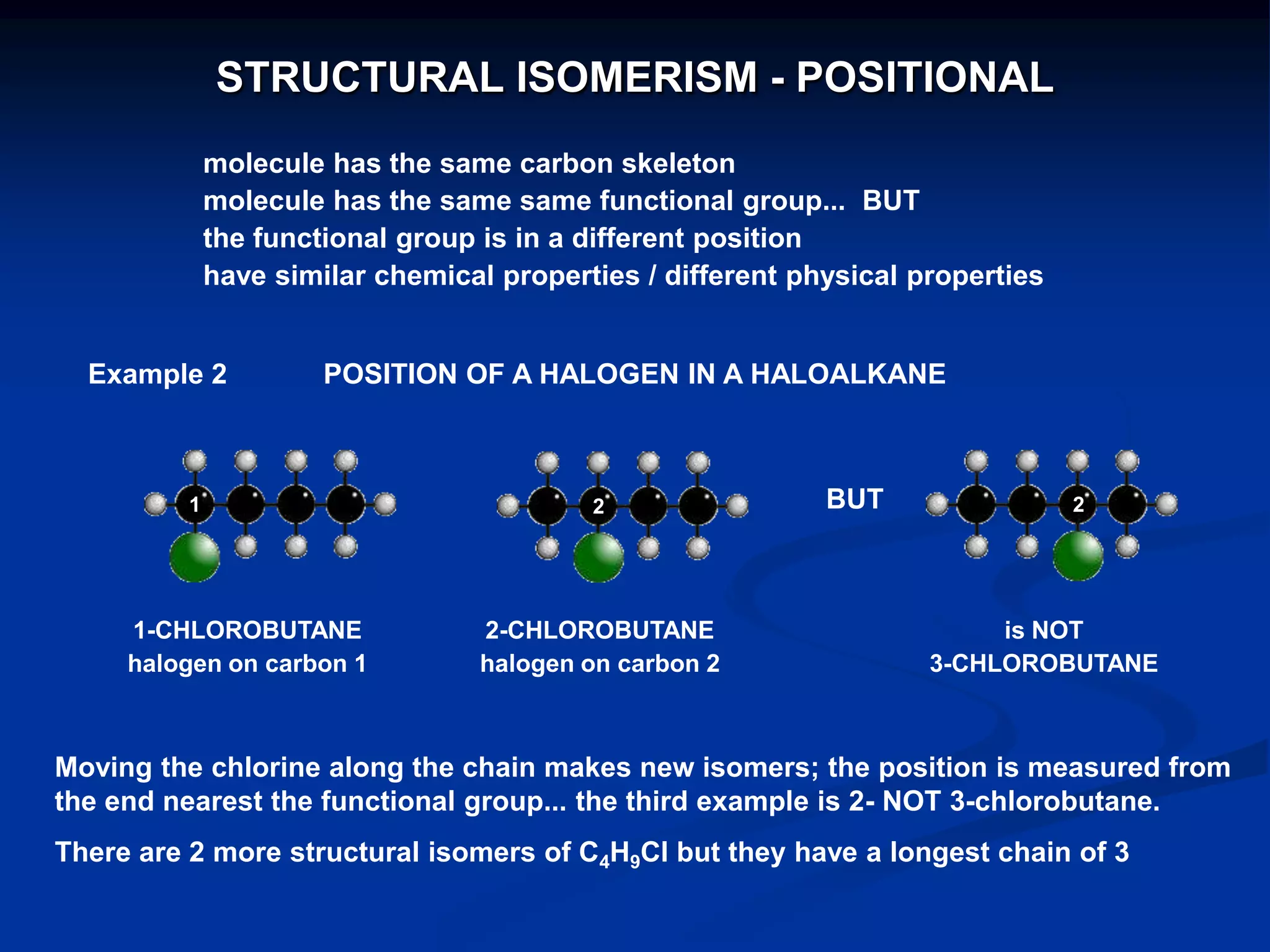
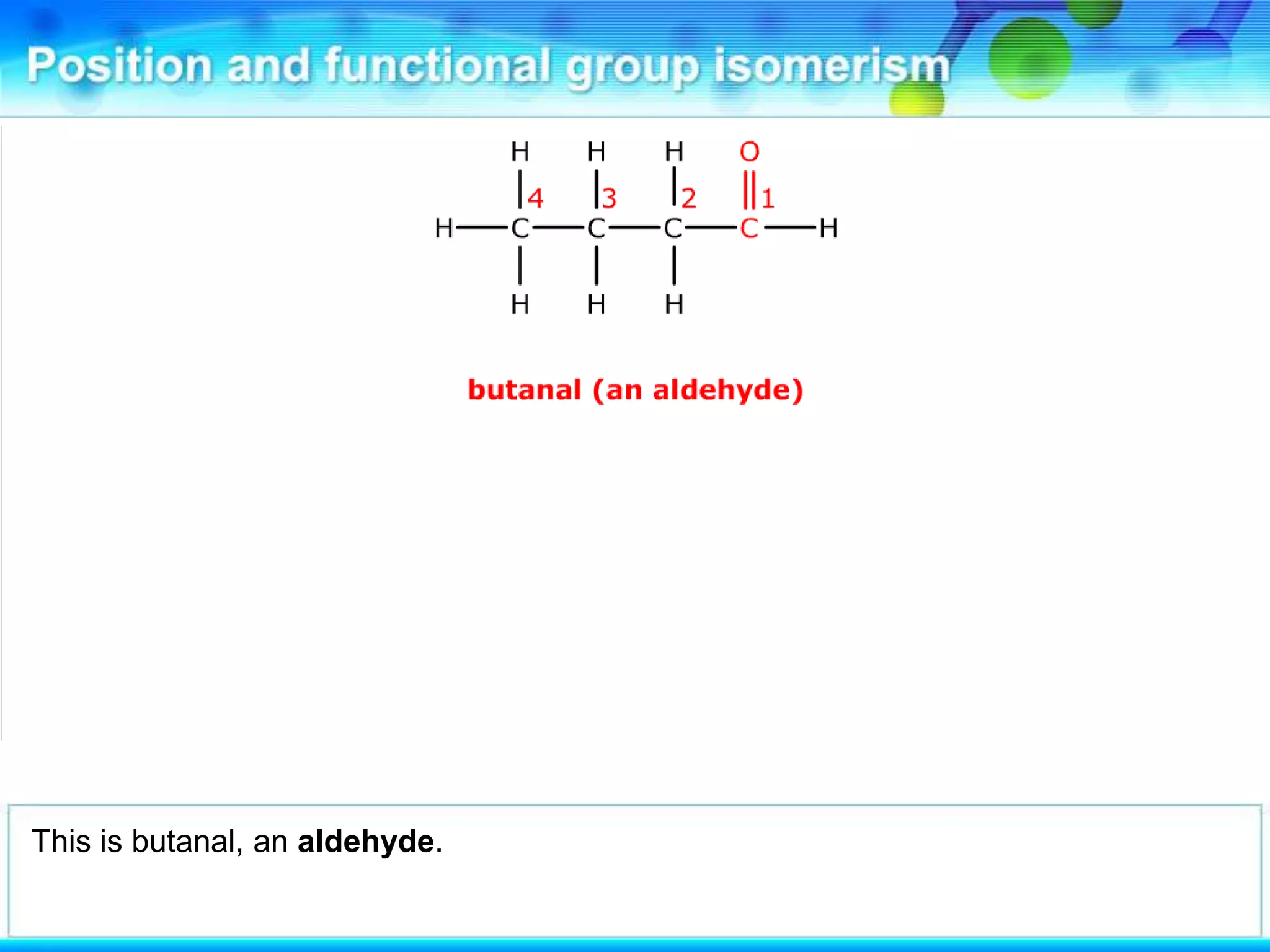
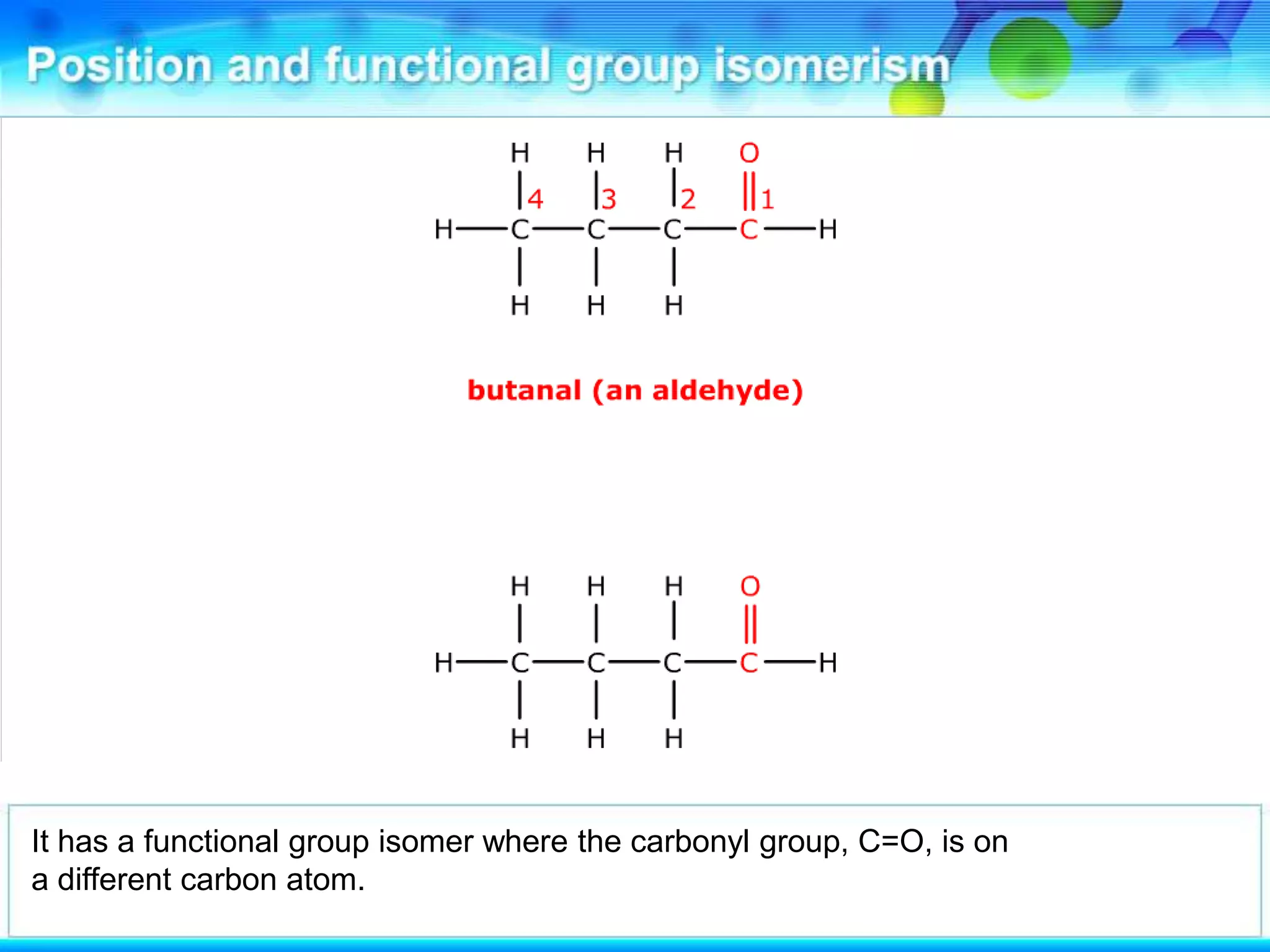
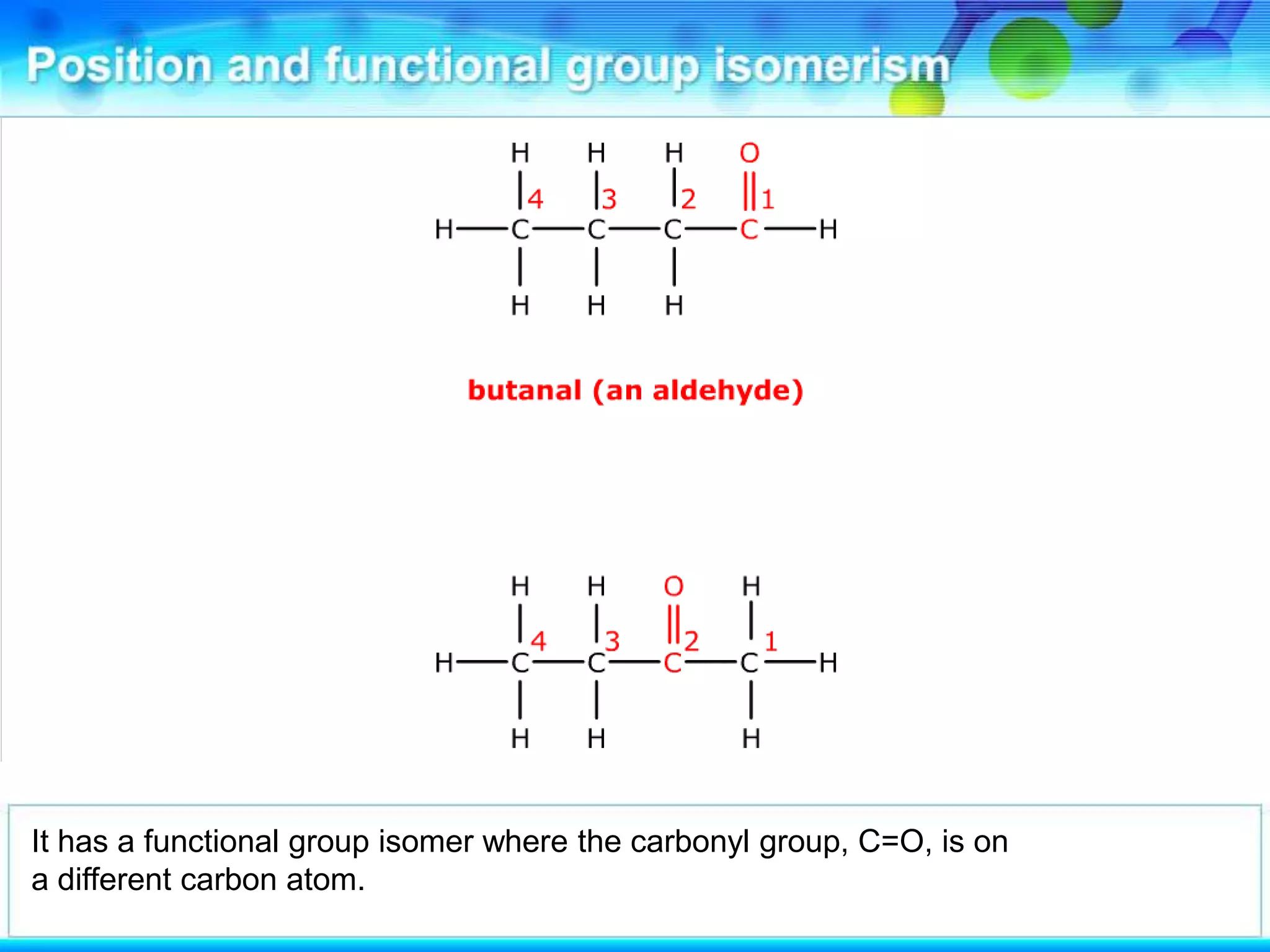
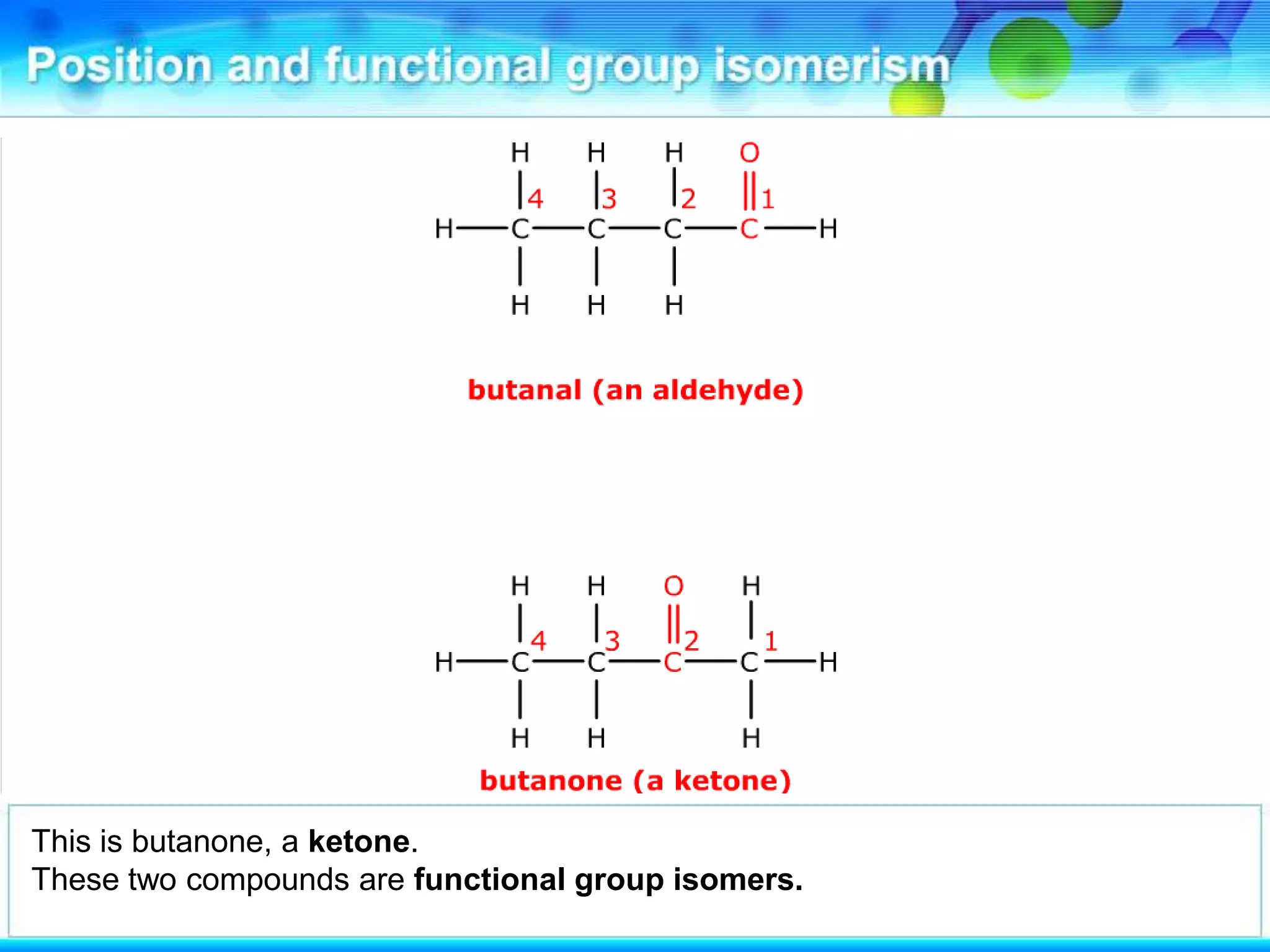
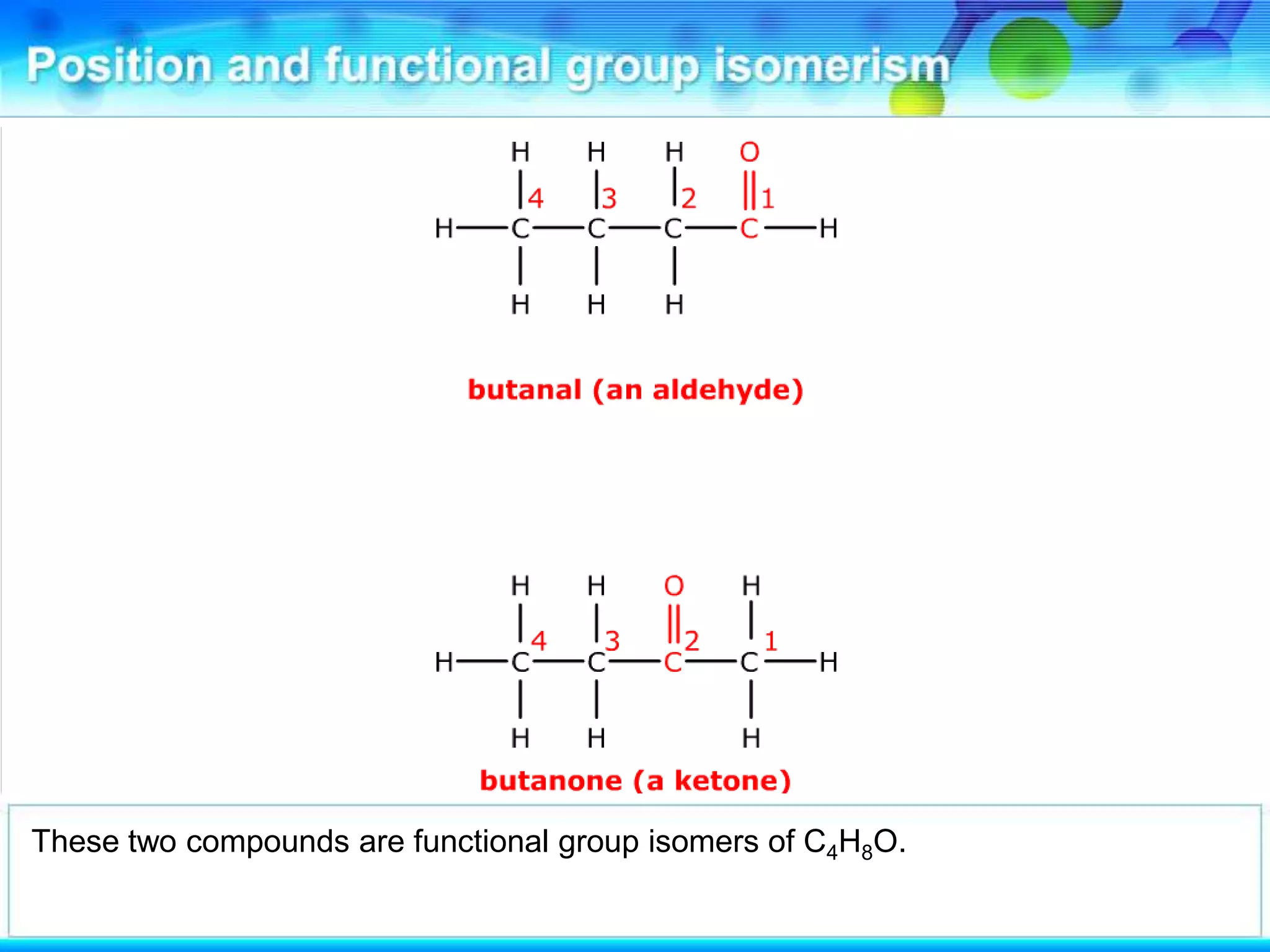
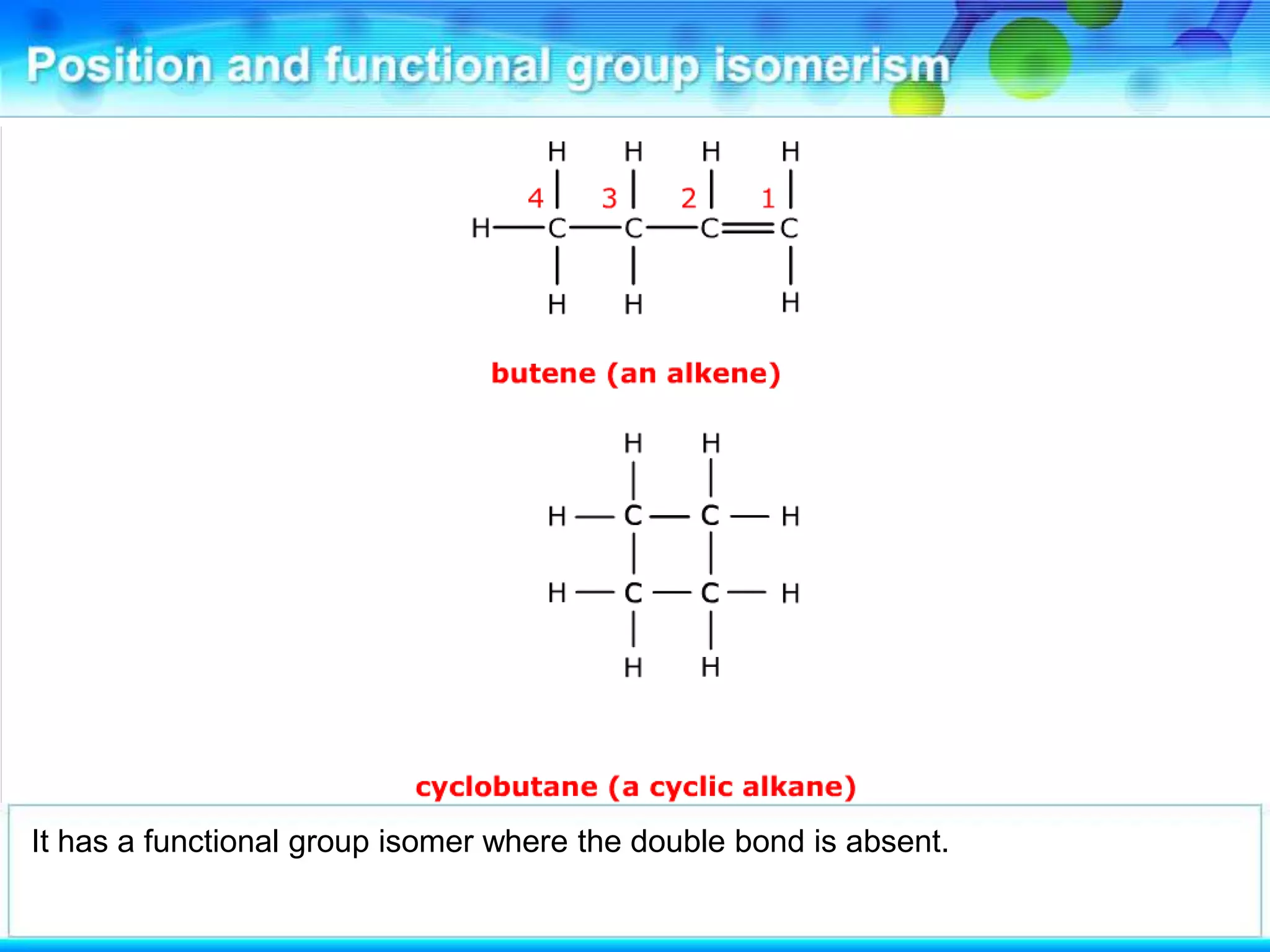
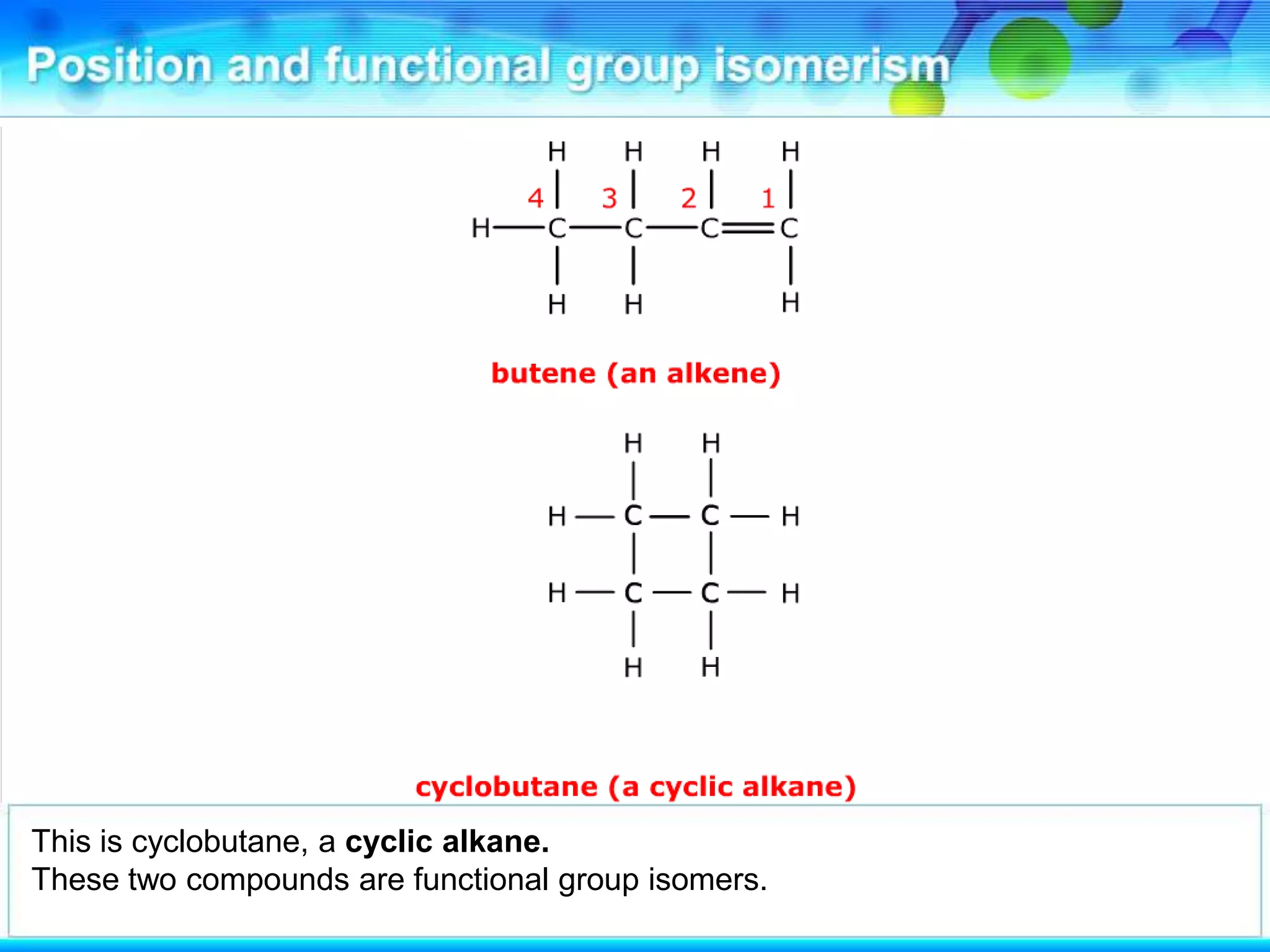
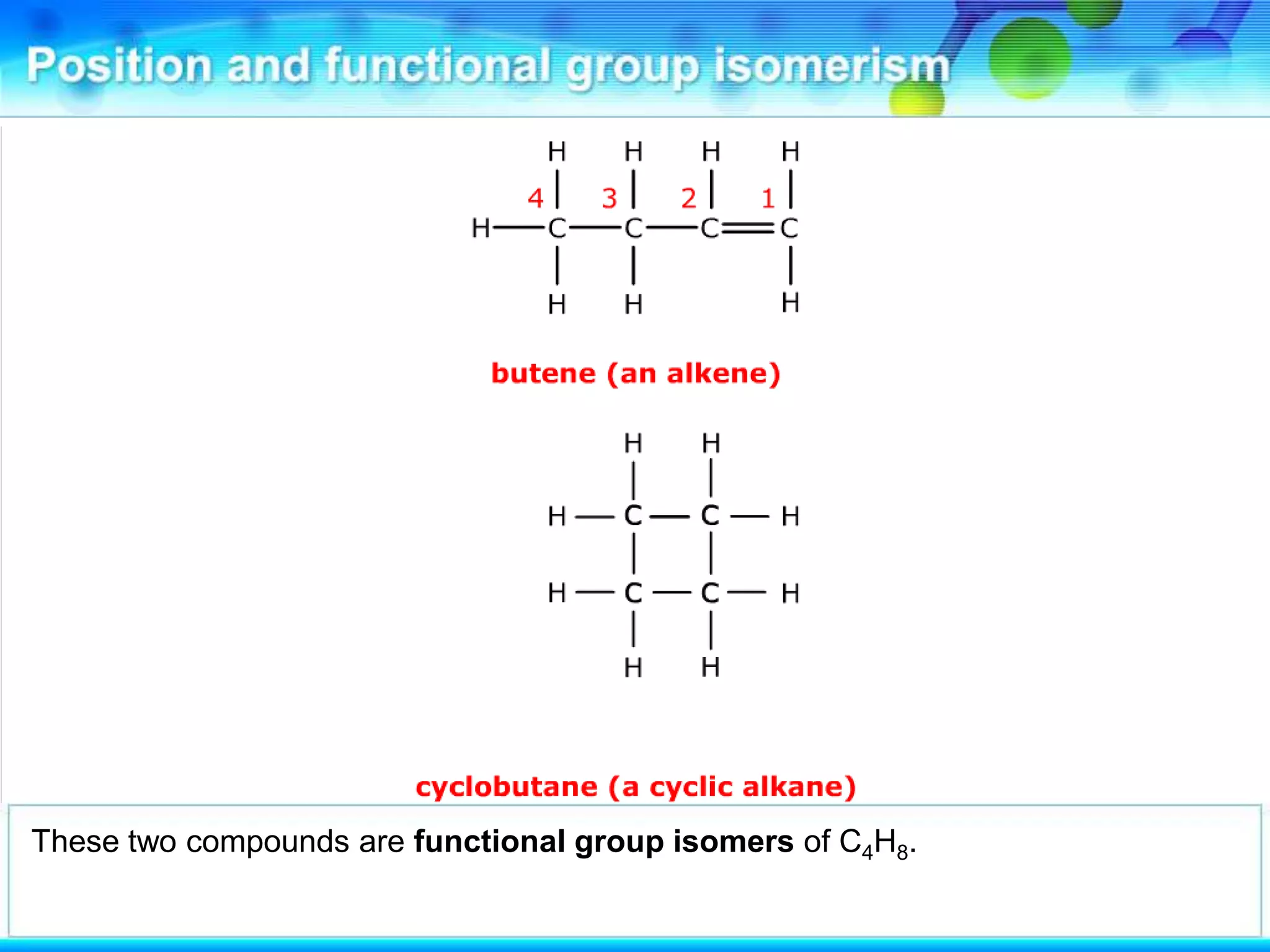
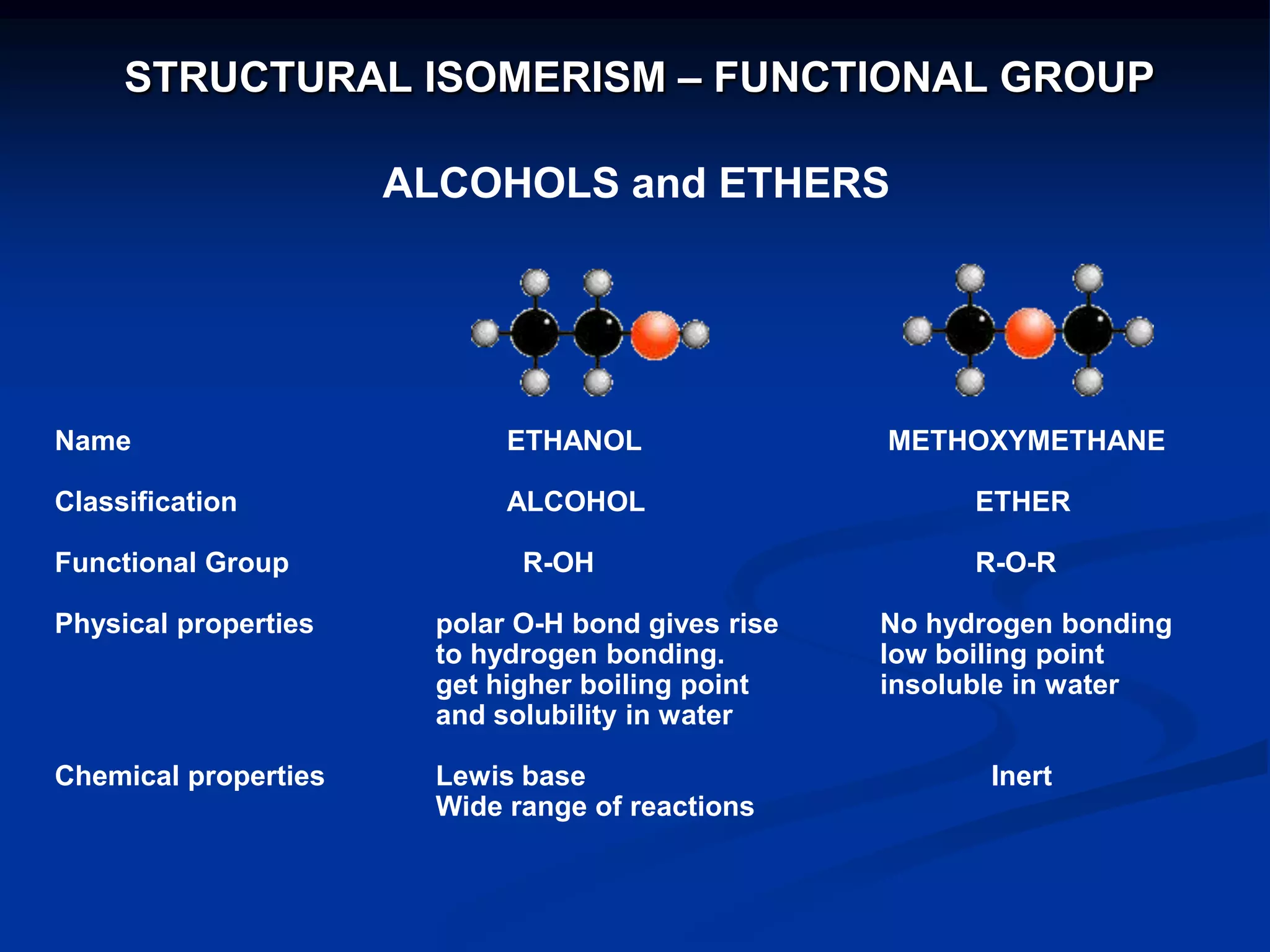
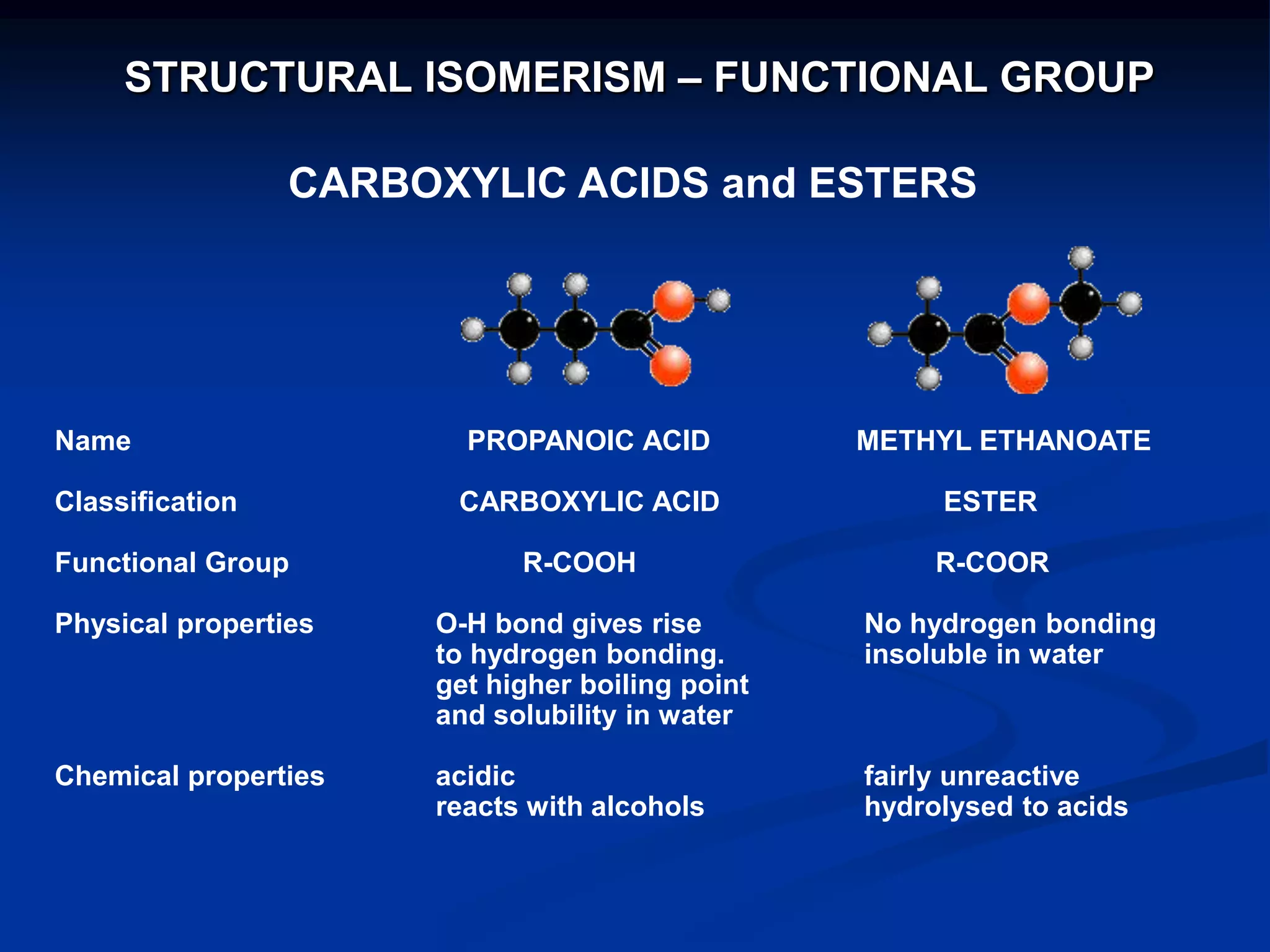
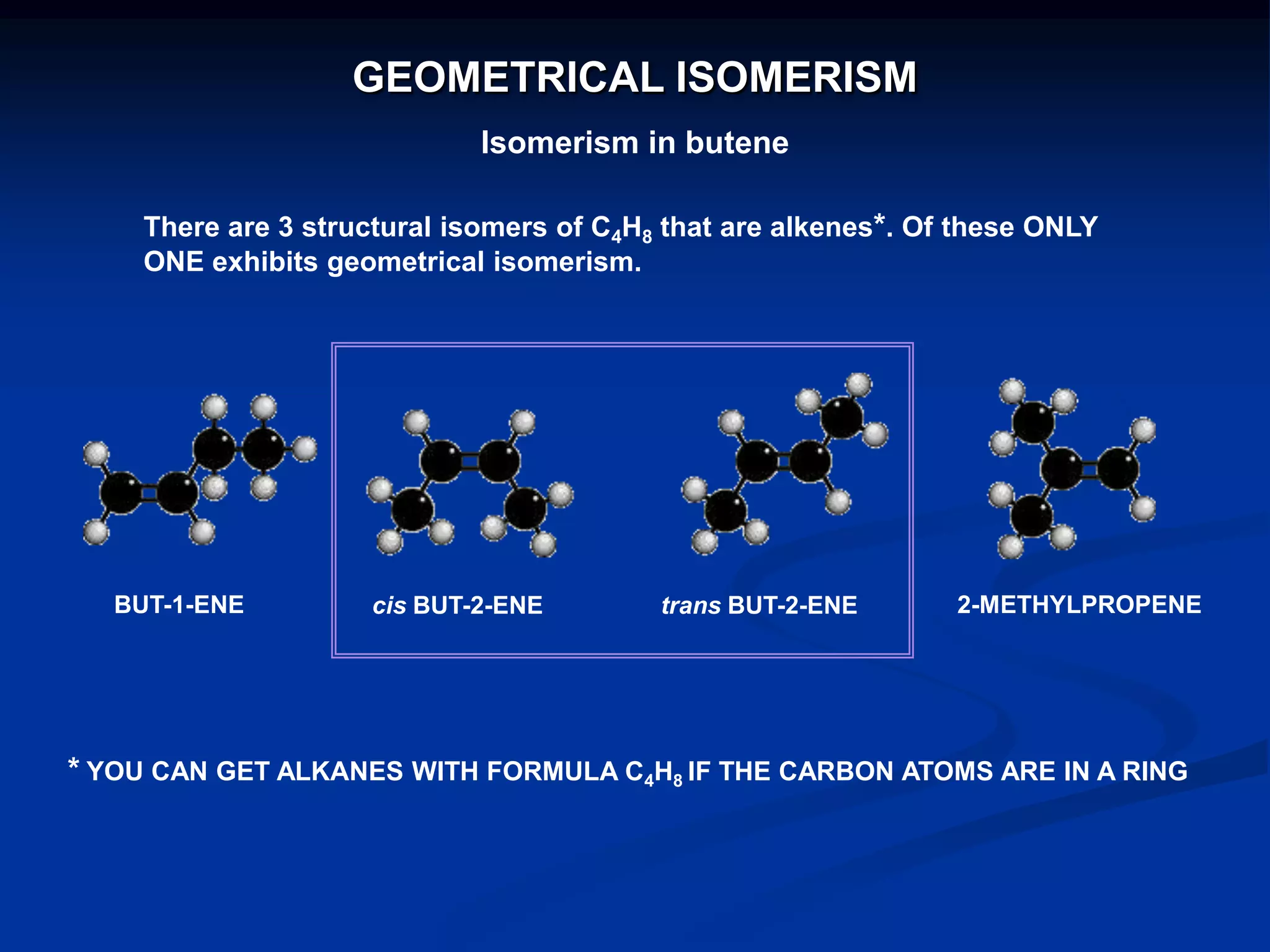
There are several types of isomerism that can occur when compounds have the same molecular formula:
- Structural isomerism occurs when compounds have the same molecular formula but different structural formulas due to variations in how atoms are connected or arranged in the carbon skeleton. This includes chain and positional isomers.
- Stereoisomerism occurs when compounds have the same molecular formula and structural formula but different spatial arrangements of atoms. Geometrical isomerism, a type of stereoisomerism, can occur in alkenes and results in cis-trans isomers due to restricted rotation around carbon-carbon double bonds.
- Functional group isomerism occurs when compounds have the same molecular formula but different