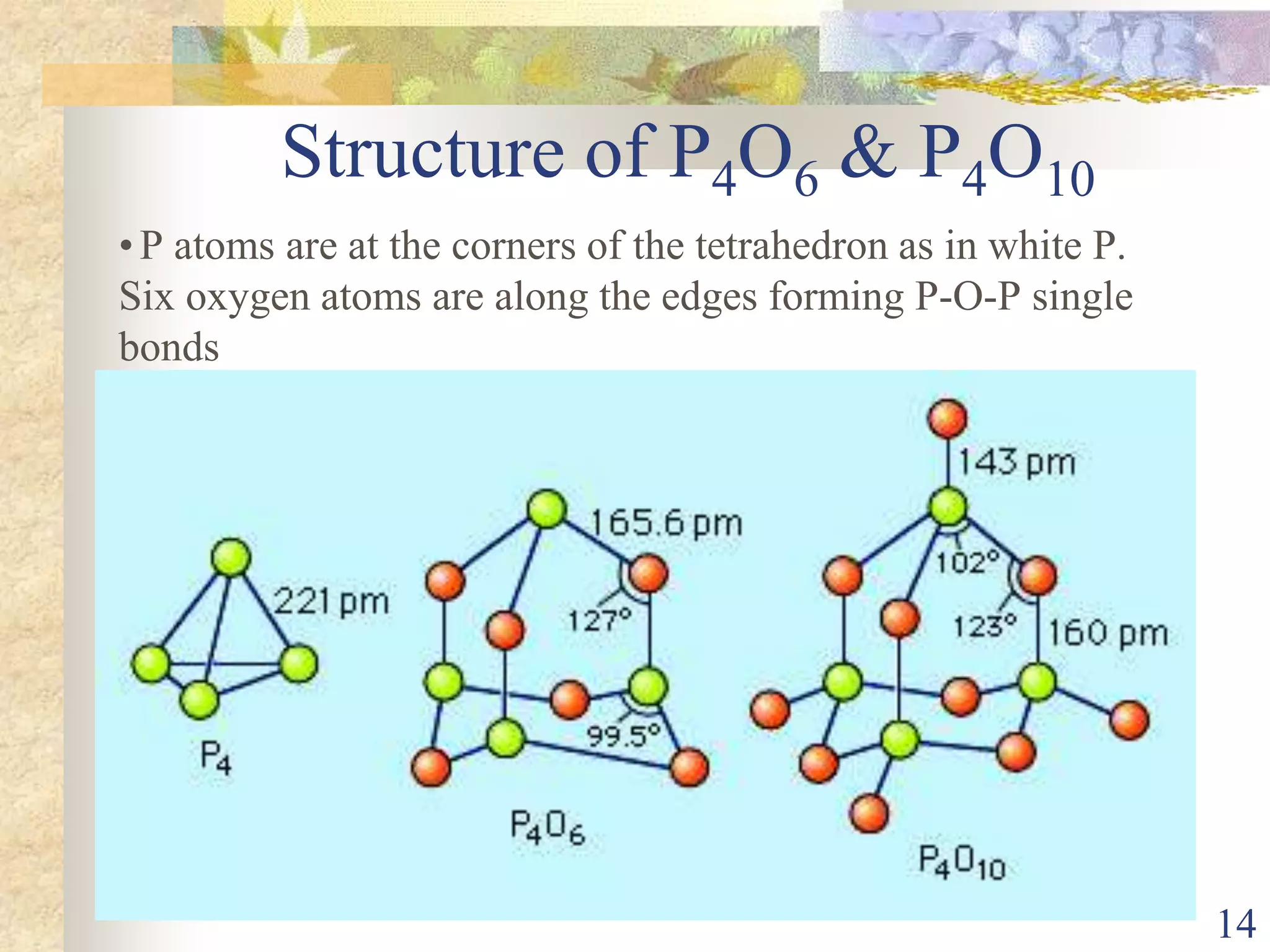
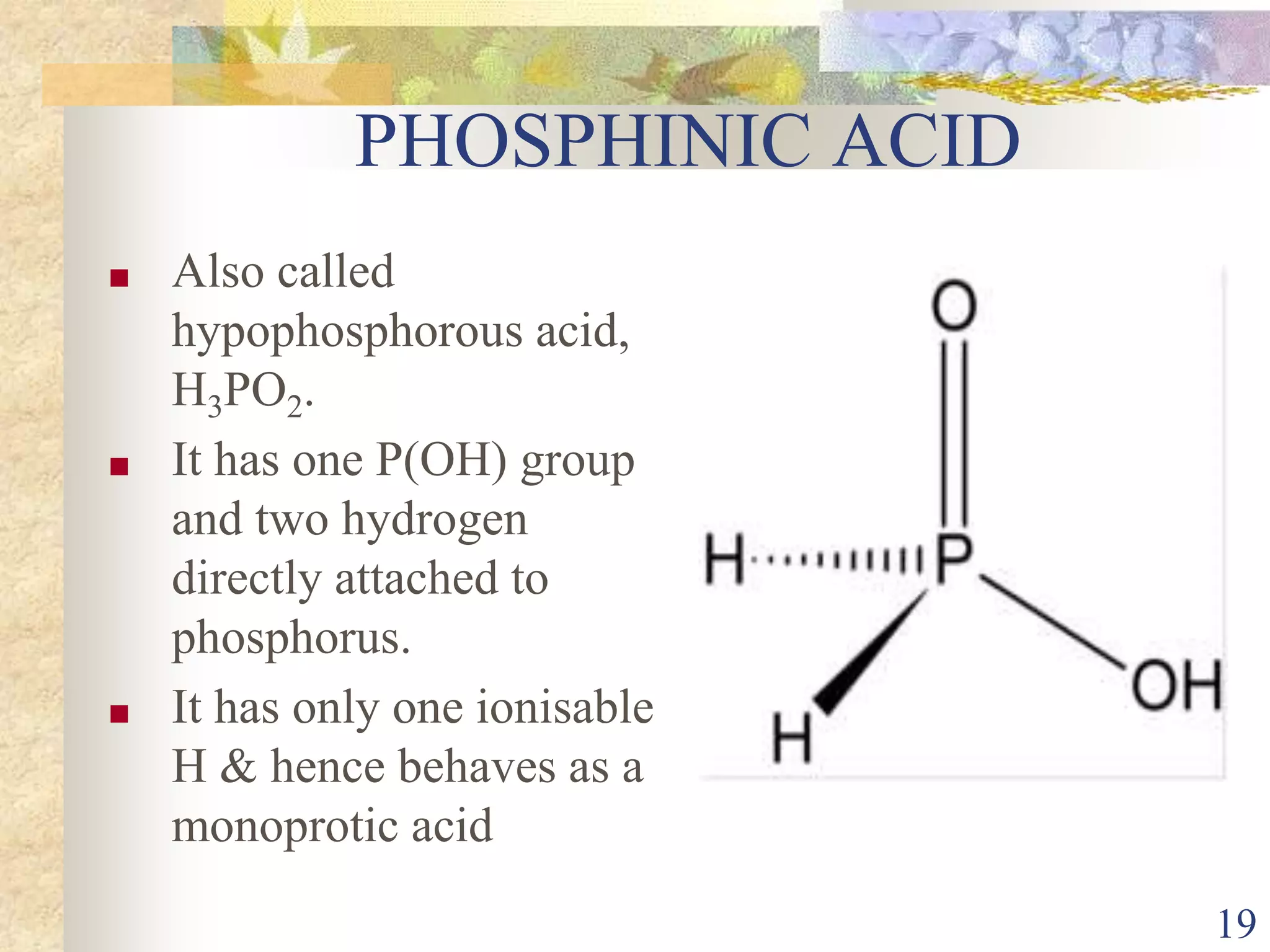
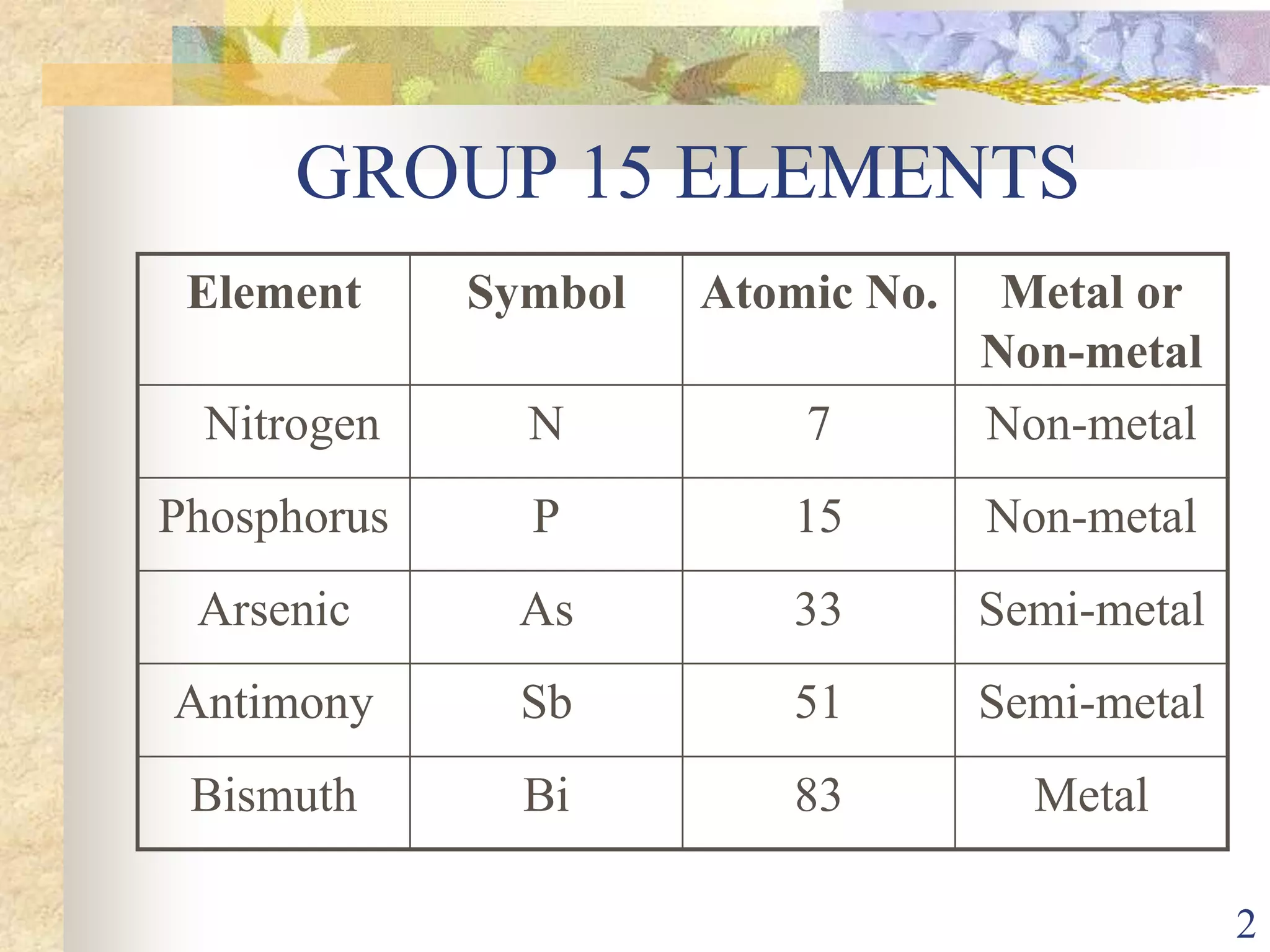
The document presents an overview of Group 15 elements, detailing their properties, oxidation states, chemical reactivity, hydrides, halides, oxides, and various allotropes of phosphorus. It covers the characteristics of these elements, such as electronic configurations, stability trends, and their uses in fertilizers and pharmaceuticals. Additionally, it discusses specific acids formed by phosphorus compounds and their respective structures.










![•PCl5 is molecular in the gas and liquid phases
but in the solid state exists as [PCl4]+[PCl6]-
containing tetra and hexa-coordinated
phosphorus species.
•PCl5 is made by the reaction of PCl3 with Cl2 in
CCl4:- PCl3 + Cl2 → PCl5
•PCl5 fumes in air, it reacts with water to give
initially POCl3; if water is excess, the product is
H3PO4. PCl5 + H2O → POCl3 + 2HCl
POCl3 + 3H2O → H3PO4 + 3HCl
12](https://image.slidesharecdn.com/pblockelementgroup15elements-230206065658-37154e6e/75/P-block-element-GROUP-15-ELEMENTS-pptx-12-2048.jpg)