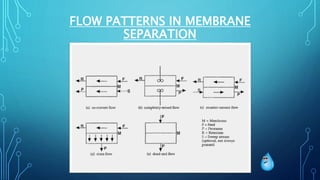
The document provides an overview of membrane separation processes, highlighting their significance in various industries such as food, pharmaceuticals, and biotechnology. It discusses types of membrane processes, their advantages and disadvantages, and applications in industrial settings, including purification and concentration of valuable products. Additionally, it covers the characteristics of synthetic membranes and various cleaning methods for maintaining membrane performance.