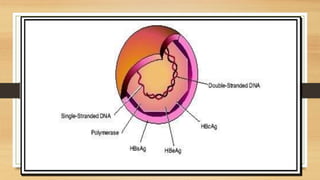
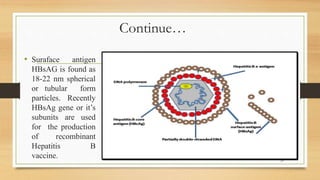
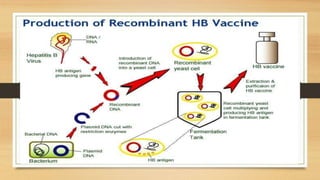
The document discusses applications of recombinant DNA technology, focusing on important recombinant proteins and their uses. It provides details on the production of human insulin, interferons, and hepatitis B vaccine through recombinant DNA techniques. Human insulin was the first therapeutic protein produced via recombinant DNA, and is made by inserting the human insulin gene into E. coli bacteria. Interferons are produced recombinantly in yeast cells, which can properly glycosylate the proteins. The hepatitis B vaccine is made from antigenic proteins of the hepatitis B virus produced recombinantly, potentially through genetic engineering of banana plants.