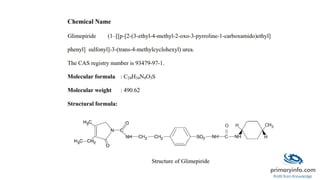
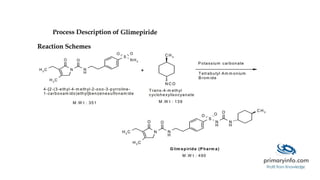
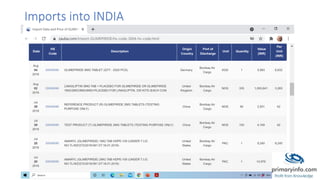
Glimepiride is a third-generation sulfonylurea antidiabetic drug used to treat type 2 diabetes. It has a low solubility and high permeability. Studies have shown glimepiride is effective at reducing fasting plasma glucose and HbA1c levels when taken once daily. Common side effects include dizziness and headaches, but no single side effect occurred in more than 2% of patients. Due to its erratic absorption in diabetic patients, developing it as a floating drug delivery system may help overcome this issue.