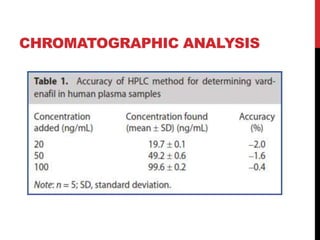
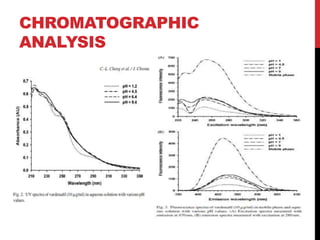
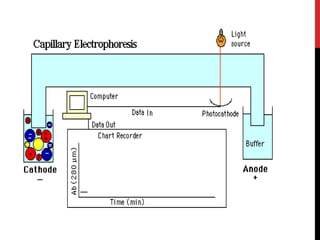
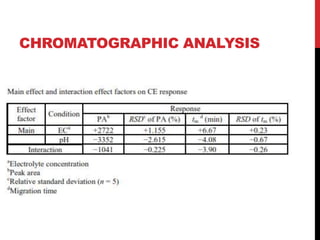
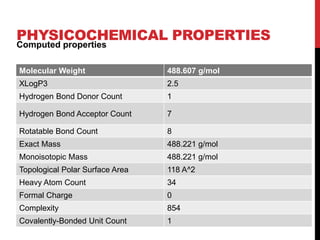
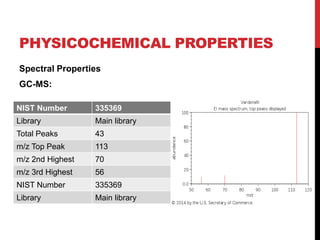
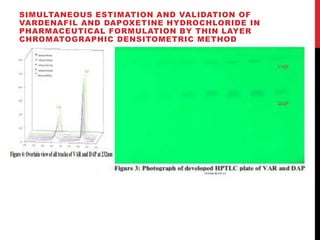
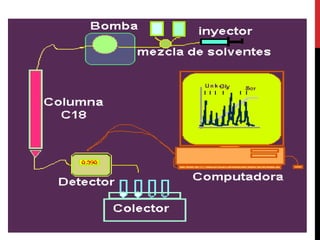
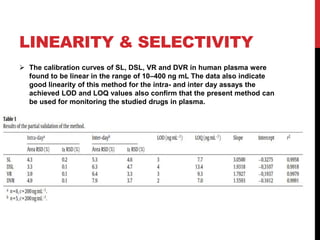
Vardenafil is a PDE5 inhibitor used to treat erectile dysfunction. This document discusses several studies that developed and validated analytical methods for quantifying vardenafil concentration. One study used thin layer chromatography with densitometric detection to simultaneously estimate vardenafil and dapoxetine in pharmaceutical formulations. Another used HPLC with amperometric detection employing a boron-doped diamond electrode to determine vardenafil and related metabolites in plasma. A third developed a HPLC-chemiluminescence method to quantify trace levels of vardenafil in dietary supplements.




















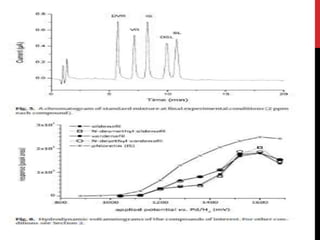

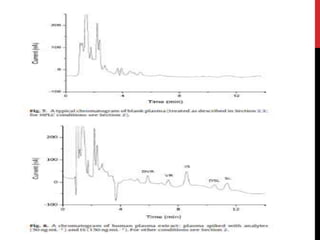





![CHROMATOGRAPHIC ANALYSIS
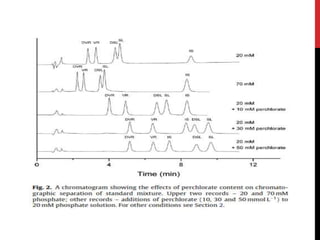
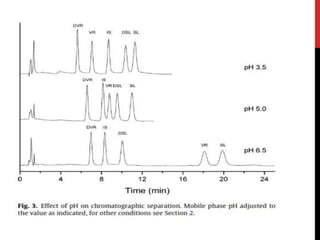
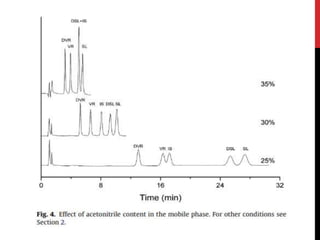
• For HPLC-CL analysis, the mobile phase should be, not only suitable for
vardenafil separation in complex matrix but also compatible with CL
detection.
• Tested buffers :
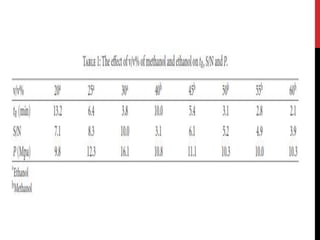
1. Acetonitrile-acidic aqueous buffer [1, 4, 11, 19]
2. methanol-acidic aqueous buffer [8, 20]
• acetonitrile brought about very high CL background. This high
background went against the sensitive vardenafil detection
• methanol and ethanol were used, relatively low CL backgrounds and
smooth baselines could be obtained. Thus, methanol and ethanol were
selected.](https://image.slidesharecdn.com/newmicrosoftpowerpointpresentation2autosaved-170325092443/85/vardinafil-methods-of-analysis-37-320.jpg)