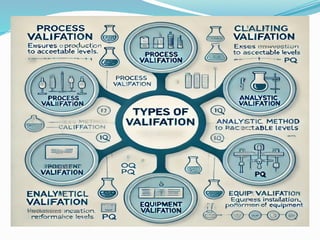
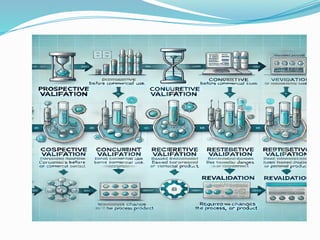
Validation in the pharmaceutical industry is the documented process of ensuring that procedures and systems meet expected results consistently, crucial for regulatory compliance with bodies like the FDA and EMA. It includes stages such as planning, qualification, and process validation, ensuring product safety, quality, and efficacy while also adapting to technological changes and evolving regulations. Continuous monitoring and revalidation are vital for maintaining product quality and compliance, enhancing operational efficiency, and mitigating risk.