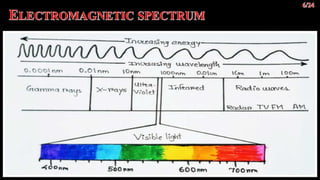
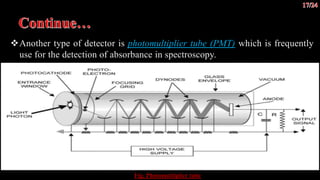
The document discusses UV-visible spectroscopy, a technique that uses electromagnetic radiation to analyze the atomic and molecular structure of matter by measuring light absorption at specific wavelengths. It explains the components of a UV-visible spectrophotometer, the principles of Beer’s and Lambert’s laws, and highlights the applications, advantages, and limitations of this method across various scientific fields. UV-visible spectroscopy is notable for its ability to analyze small sample sizes and its widespread use in industries such as pharmaceuticals, forensics, and biochemistry.