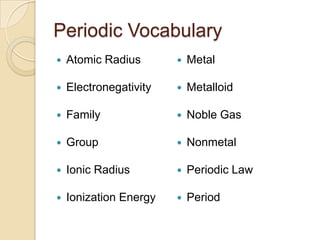
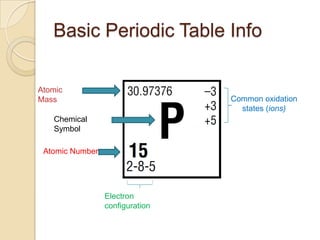
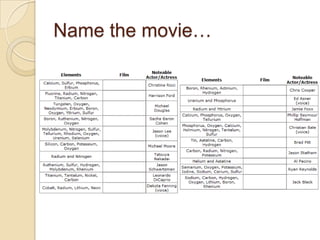
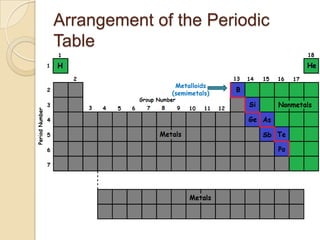
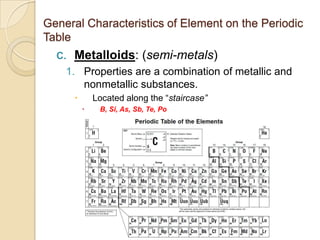
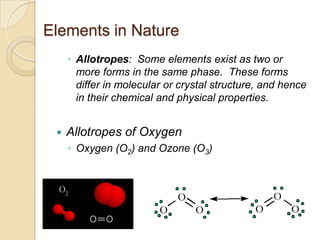
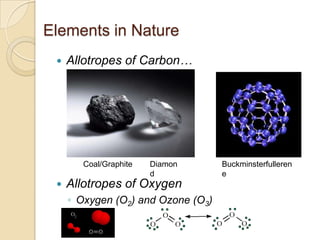
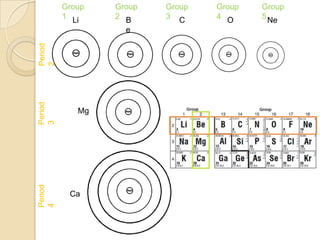
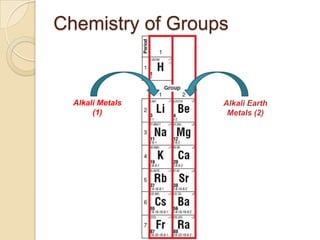
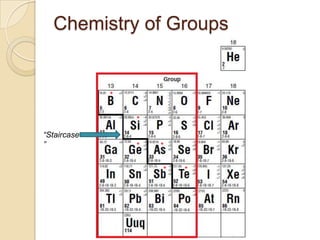
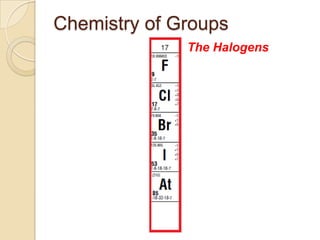
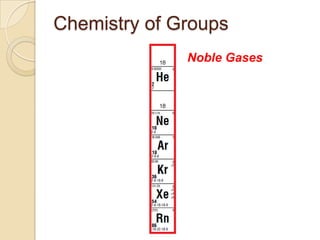
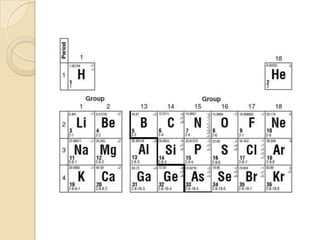
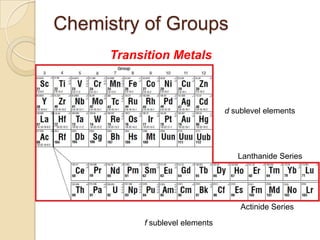
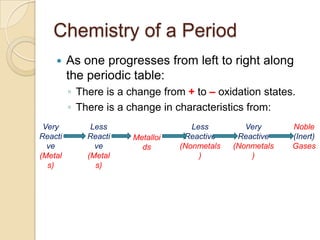
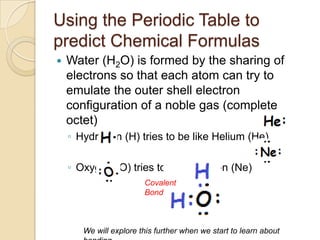
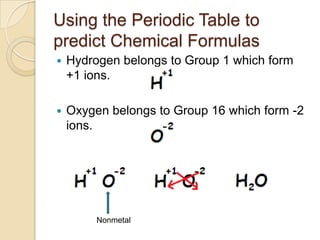
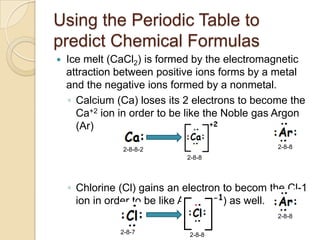
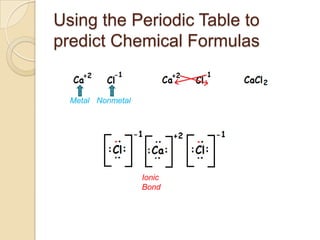
The document discusses the periodic table and periodic trends in properties of elements. It defines key periodic table terms like groups, periods, atomic radius, ionization energy, and provides examples of how properties change within periods and groups. It also discusses Mendeleev's original periodic table and the modern periodic law.