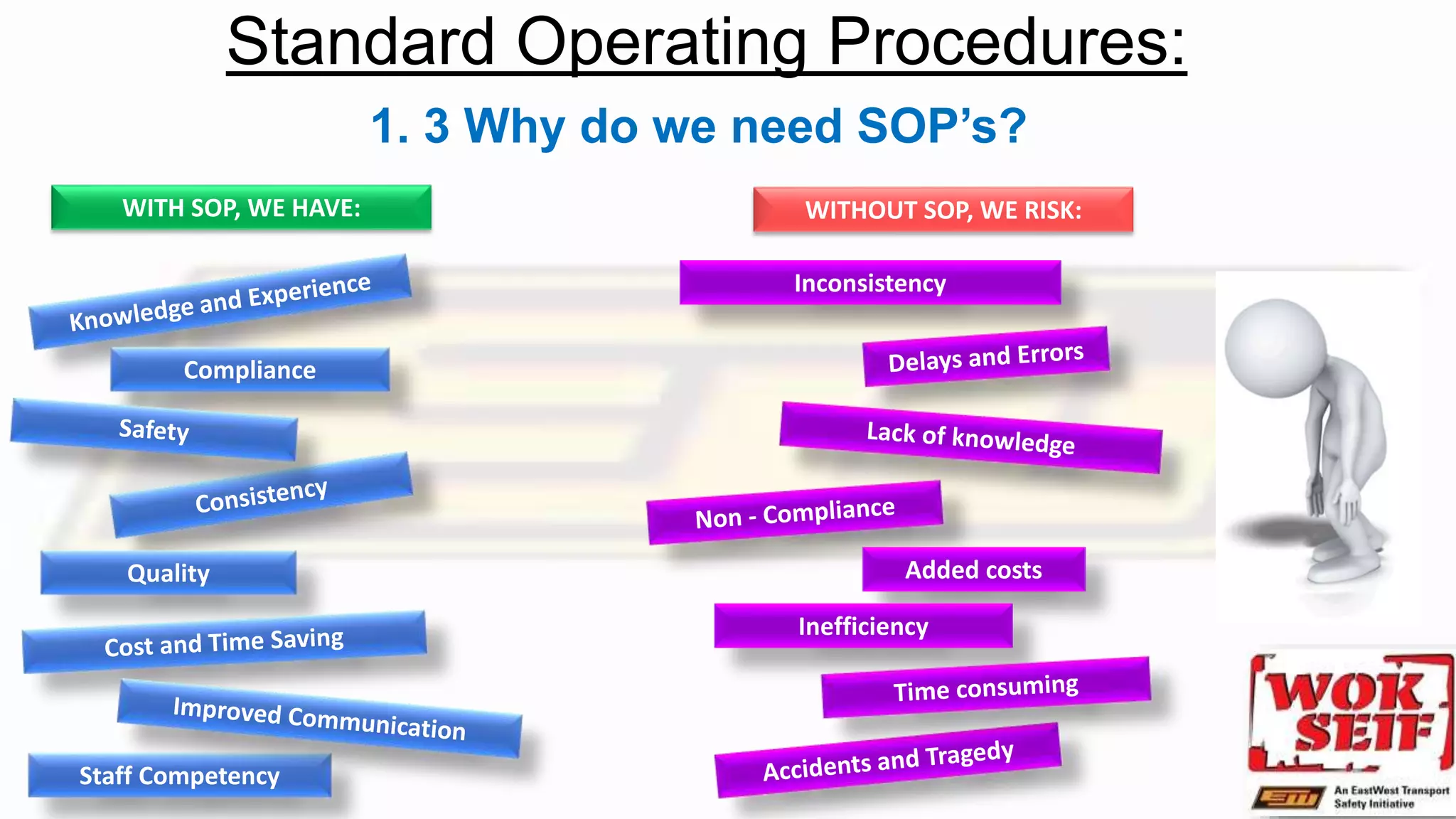
The document outlines a learning session focused on Standard Operating Procedures (SOPs), detailing objectives such as writing, updating, and applying SOPs within a company. It emphasizes the importance of SOPs in maintaining staff competency and efficiency while guiding new staff during induction. Additionally, the document highlights the steps for creating, applying, and reviewing SOPs to align with organizational changes and improvements.