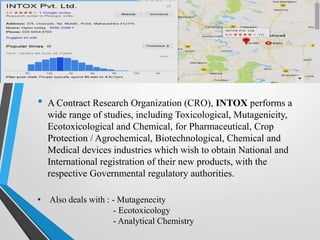
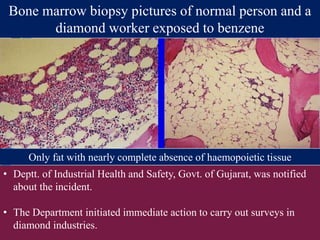
Toxicology is the study of poisons and their effects. It includes the study of adverse drug effects and detection, prevention, and treatment of poisonings. Toxicology has several branches including toxicogenomics, chemical toxicology, environmental toxicology, medical toxicology, forensic toxicology, and aquatic toxicology. Toxicovigilance actively monitors toxic risks in communities and evaluates measures to reduce them through surveillance of poisoning cases, investigations, and risk communication. Examples of toxicovigilance activities include identifying 1,2-dichloroethane exposures in agarbatti workers and benzene exposures in diamond workers.

![Swiss physician Paracelsus (1493-1541)
credited with being -
“The Father of Modern Toxicology”[1]
“All substances are poisons: there is none
which is not a poison. The right dose
differentiates a poison from a remedy”
1. Toxicology Education Foundation. (2017). History of Toxicology. [online] Available at:
http://toxedfoundation.org/history-of-toxicology/ [Accessed 30 Jul. 2017].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-2-320.jpg)
![Toxicology
• Study of poisonous effect of drugs and other chemicals
(household, environmental pollutant, industrial,
agricultural, homicidal) with emphasis on detection,
prevention and treatment of poisonings.
• It also includes the study of adverse effects of drugs,
since the same substance can be a drug or a poison,
depending on the dose[2].
• Poison - any substance, including any drug, that has the
capacity to harm a living organism[3].
2. K.D.Tripathi, Essentials of medical pharmacology, Introduction, Routes of drug administration.
3. Goodman & Gilman's The Pharmacological Basis of Therapeutics > Section I. General Principles > Chapter 4. Drug Toxicity and
Poisoning.](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-3-320.jpg)
![Branches
1. Toxicogenomics – applies molecular profiling approaches to the
study of toxicology
2. Chemical toxicology – deals with the mechanisms and structures
of chemical agents and their toxic effects[4].
3. Environmental toxicology - also known as entox, is a
multidisciplinary field of science concerned with the study of the
harmful effects of various chemical, biological and physical agents
on living organisms[5].
4. silverstars, b. (2017). Toxicology and Its Types. [online] Biotecharticles.com. Available at: http://www.biotecharticles.com/Toxicology-
Article/Toxicology-and-Its-Types-16.html [Accessed 27 Jul. 2017].
5. xwwsSfu.ca. (2017). About the MET Program - Department of Biological Sciences - Simon Fraser University. [online] Available at:
http://www.sfu.ca/biology/graduate/about-met.html [Accessed 27 Jul. 2017].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-4-320.jpg)
![4.) Medical toxicology - medical subspecialty focusing on the
diagnosis, management and prevention of poisoning and other
adverse health effects due to medications, occupational and
environmental toxins, and biological agents[6].
5.) Forensic toxicology - deals with the medico-legal aspects of the
harmful effects of chemicals on human beings.
6.) Aquatic toxicology – studies the effects of manufactured
chemicals and natural materials on aquatic organisms[4].
4. silverstars, b. (2017). Toxicology and Its Types. [online] Biotecharticles.com. Available at:
http://www.biotecharticles.com/Toxicology-Article/Toxicology-and-Its-Types-16.html [Accessed 27 Jul. 2017].
6. Acmt.net. (2017). ACMT - Introduction to Medical Toxicology. [online] Available at: http://www.acmt.net/overview.html [Accessed
27 Jul. 2017].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-5-320.jpg)




![Toxicovigilance
• Active process of identifying and evaluating the toxic
risks existing in a community, and evaluating the measures
taken to reduce or eliminate them[7].
7. Who.int. (2017). WHO | Toxicovigilance. [online] Available at:
http://www.who.int/ipcs/poisons/centre/toxicovigilance/en/ [Accessed 24 Jul. 2017]](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-10-320.jpg)
![How it started
In the early 1950s, some clinical and
hospital structures developed actions,
mainly devoted to providing information
on poisoning to medical people.
This was the beginning of Poison
Information Centres in Europe &
North America.
Soon after, The World Federation of
Associations of Clinical Toxicology
Centres & Poison Control Centres was
founded in 1975,in Lyon[8].
The National Poisons Information Centre
(NPIC) was established in the Department
of Pharmacology at AIIMS in 1995.](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-11-320.jpg)


![Surveillance
Capturing all the poisoning cases from intensive and acute medical care units,
general accidental and emergency departments, and at causality of all the public
healthcare hospitals.
Getting poisoning cases from local and oversea media reports plays a main role
for a better understanding of poisoning risk in the population.
Use of computerized laboratory data as a detection support tool of toxic reactions
in hospital plays a major step for easy identification of risk factors and improves
patient medical care.
Categories of information include the patient, the caller, the exposure, the
substance, clinical toxicity, treatment, and medical outcome.
Example: the toxic exposure surveillance system (TESS) database was initiated in
1985, and provides a baseline of more than 36.2 million cases through 2003[9].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-14-320.jpg)
![Investigations
• Identification and investigation should start immediately upon
acknowledge of notification of poisoning incidents of public
health implication and consequences.
• For investigation, efforts should be made from both the
government departments and other healthcare services for
implementation of effective control measures.
• The Government laboratory supplies a comprehensive analytical
service to the Department of Health in the testing of drugs and
poisons to alleviate the investigation of poisoning incidents.
• Investigation of the phenomenon of reported clinical adverse
reactions should be initiated by the competent authority[9].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-15-320.jpg)

![Risk Communication
• Press release will be issued as appropriate on current health issues
and concerns.
• Health education materials in the form of electronic publications
and fact sheets are uploaded onto the community health profile
website to keep the public and health professionals informed of the
latest development.
• Important messages are disseminated to the public through the
distribution of pamphlets, fact sheets, posters, and health talks.
• Public announcements on poisoning cases are made as appropriate
to raise the public’s alertness to possible poisoning risks[9].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-18-320.jpg)







![C. Ambiguous label information
resulting in overdose
• The National Poisons Information Service in the United
Kingdom identified a high frequency of enquiries about
accidental, ten-times overdoses of an antidote, N-
acetylcysteine, used for paracetamol (acetaminophen)
poisoning.
• This was notified to the manufacturer. Further investigation
suggested that a contributory factor in the overdoses was the
way in which the content of N-acetylcysteine was expressed
on the ampoule labels: the labelling information could have
been understood to mean that the ampoule contained 200mg
in total rather than 200 mg/mL (2g in total).
• The manufacturers subsequently changed the label so that
the content of the N-acetylcysteine was stated more
explicitly (Mant et al (1984) British Medical Journal, 289
(6439):217-219)[7].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-27-320.jpg)
![• The Swiss Toxicological Information Service (STIS)
observed a steep increase in the number of enquiries
about people suffering respiratory problems after
using sprays for waterproofing leather and textiles.
• Examination of enquiry data revealed that three
brands were implicated. STIS informed the Swiss
Federal Office of Public Health, which ordered the
withdrawal of the products from the market and
alerted the public.
• Further investigations suggested that a reformulation
of the products was responsible for causing lung
damage[7].
D. Reformulation of a waterproofing spray,
resulting in lung damage in users](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-28-320.jpg)
![• The national poisons centre in Morocco (Centre
Antipoison du Maroc) regularly analyses its enquiries
to identify trends and problems.
• An analysis of enquiries received about poisoning
with cosmetic products between 1980 and 2010
revealed that this was largely an urban problem
predominantly involving adult females.
• Nearly two thirds of exposures involved para-
phenylenediamine, a chemical used in hair dyes,
artificial henna and some tattoo inks, which can
easily be bought from herbalists as a product called
Takaout Roumia.
• The majority of these exposures were the result of
intentional self-poisoning, with a mortality rate of
around 18%.
• The poisons centre data also highlighted problems
with skin lighteners of unknown formulation and hair
straighteners containing acetone[7].
E. Hazardous cosmetic products](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-29-320.jpg)
![Toxicovigilance: a new approach for toxicological screening, quality
control and regulation of irrational herbal drugs
It is clear that the herbal industry needs to follow strict guidelines
and that regulations are needed.
The food and drug administrations that regulate prescription drugs
only review a herbal product if the item is suspected of being
harmful or if the label contains a medical claim[10].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-30-320.jpg)
![Toxicity of herbal drugs
• .
For several reasons it is not possible to establish absolute safety
standards for herbal preparations based solely on epidemiological
studies:
First, these types of studies would be costly
Second, there is little published data in countries where the major
use of medicinal plants occurs and thus general standards based on a
limited number of reports would have little meaning.
Third, the exact identification of the products implicated in side
effects claimed for medicinal plants is usually lacking[10].](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-31-320.jpg)

![1. K.D.Tripathi, Essentials of medical pharmacology, Introduction, Routes of drug
administration: 2.
2. Goodman & Gilman's The Pharmacological Basis of Therapeutics > Section I. General
Principles > Chapter 4. Drug Toxicity and Poisoning.
3. Toxicology Education Foundation. (2017). History of Toxicology. [online] Available at:
http://toxedfoundation.org/history-of-toxicology/ [Accessed 30 Jul. 2017].
4. silverstars, b. (2017). Toxicology and Its Types. [online] Biotecharticles.com. Available at:
http://www.biotecharticles.com/Toxicology-Article/Toxicology-and-Its-Types-16.html
[Accessed 27 Jul. 2017].
5. xwwsSfu.ca. (2017). About the MET Program - Department of Biological Sciences -
Simon Fraser University. [online] Available at: http://www.sfu.ca/biology/graduate/about-
met.html [Accessed 27 Jul. 2017].
6. Acmt.net. (2017). ACMT - Introduction to Medical Toxicology. [online] Available at:
http://www.acmt.net/overview.html [Accessed 27 Jul. 2017].
7. Who.int. (2017). WHO | Toxicovigilance. [online] Available at:
http://www.who.int/ipcs/poisons/centre/toxicovigilance/en/ [Accessed 24 Jul. 2017].
8. Anon, (2017). [online] Available at: http://poison control centres and toxicovigilance -
CIDBIMENA [Accessed 25 Jul. 2017].
9. Avula Ravali, Alapati Sahithi et al; A REVIEW ON TOXICOVIGILANCE; ejpmr,
2016,3(3), 142-145.
10. Neetesh K Jain, Suman Jain et al; Toxicovigilance: a new approach for toxicological
screening, quality control and regulation of irrational herbal drugs; JIPBS, Vol 3 (1), 033-
035, 2016.
References](https://image.slidesharecdn.com/toxicologytoxicovigilance-171028173443/85/Toxicology-Toxicovigilance-33-320.jpg)
