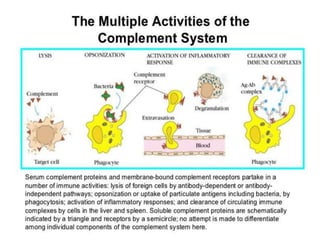
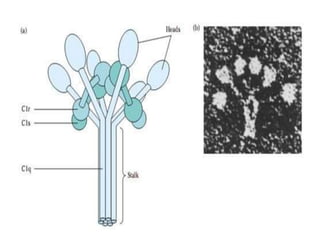
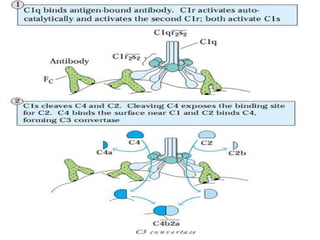
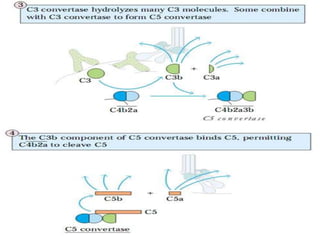
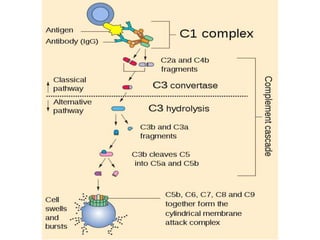
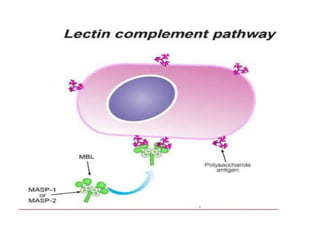
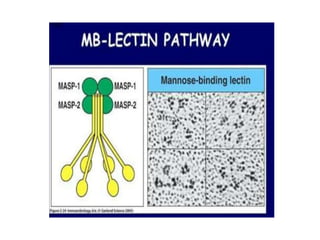
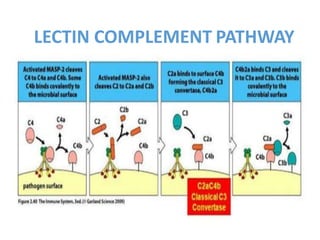
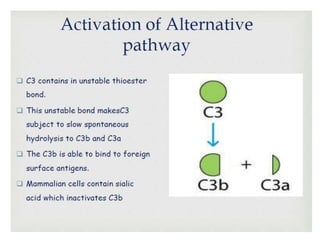
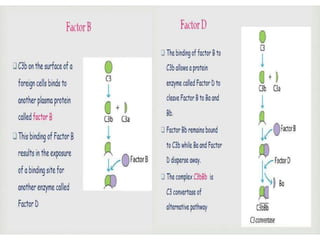
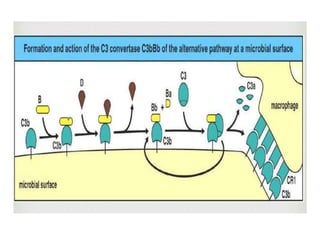
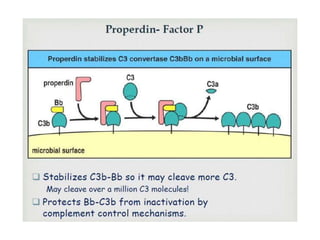
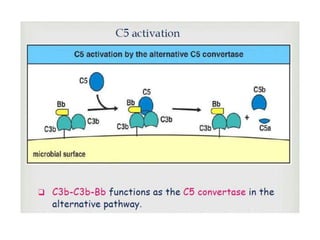
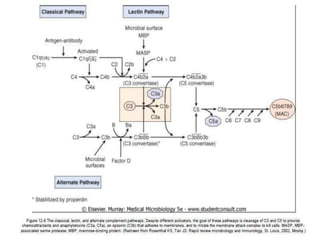
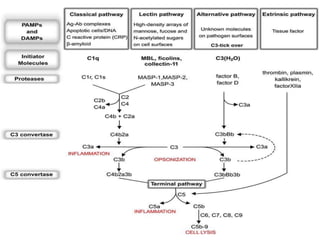
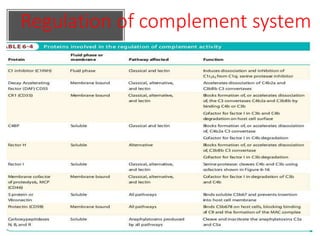
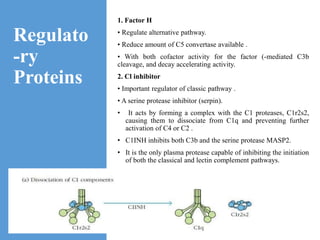
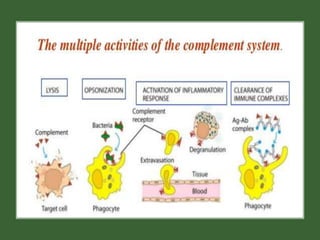
This document provides an overview of the complement system submitted by five MSc students to their professor. It describes that complement was first discovered in 1890 and plays a major role in innate immunity through lysis of cells, opsonization, and inflammation. It summarizes the three pathways of complement activation (classical, lectin, and alternative), components and regulation of the complement system, and consequences of complement activation including cell lysis, inflammation, opsonization, viral neutralization, and solubilization of immune complexes.