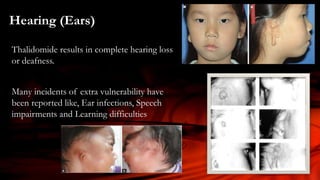
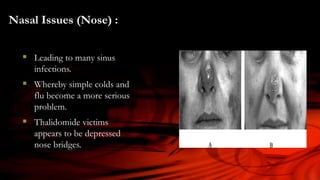
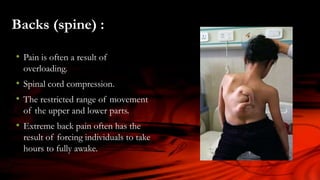
The document discusses the thalidomide tragedy in Canada, detailing how the drug, marketed in the late 1950s as a safe treatment for insomnia and morning sickness in pregnant women, led to severe birth malformations and disabilities. While many countries acted to compensate victims, Canadian patients were left to handle their grievances privately and faced significant obstacles in seeking justice. It also highlights ongoing health issues faced by thalidomide survivors and critiques the governmental negligence in addressing the repercussions of the drug.