States of matter
•Download as PPTX, PDF•
5 likes•2,809 views
The document discusses the kinetic theory of matter and the states of matter. It explains that the kinetic theory states that particles in matter are in constant, random motion and that the arrangement and movement of atoms determines the state of matter. It then describes the three states of matter - solid, liquid, and gas - and how temperature affects the motion of particles and ability to change states.
Report
Share
Report
Share
Recommended
Separating mixtures (Grade 7)

Mixtures
Ways of separating mixtures
Grade 7 Enhanced Science - Chemistry
Periodic Table of Elements Grade 7 1st Quarter 

presentation guide on periodic table of elements for grade 7 first quarter
Acids, Bases, and Salts

K to 12 Grade 7 Acids, Bases, and Salt. A chemistry lesson in first quarter. It includes learning competencies, sample exercises, and videos.
Recommended
Separating mixtures (Grade 7)

Mixtures
Ways of separating mixtures
Grade 7 Enhanced Science - Chemistry
Periodic Table of Elements Grade 7 1st Quarter 

presentation guide on periodic table of elements for grade 7 first quarter
Acids, Bases, and Salts

K to 12 Grade 7 Acids, Bases, and Salt. A chemistry lesson in first quarter. It includes learning competencies, sample exercises, and videos.
Force and its effects

Force and its effects for class 8. this ppt provides an easy way to understand force and the effects of force by giving various daily life examples. It consists of animations through which students can easily understand.
Mixtures

A mixture forms when two or more substances are combined such that each substance retains its own chemical identity. Everywhere around us are made up of mixtures. We can see them in nature, along the surface of the earth, in the oceans and in the foods we eat. There are infinite numbers of mixtures that can be combined into homogeneous or heterogeneous.
States of Matter and changes of state

States of Matter for highschool, solid, liquid, gas, freezing point, melting point, characteristic properties, physical properties, chemical properties.
Matter, Properties, & Phases

Lesson teaches students about Matter (grades 6-8) & talks about the atomic models & the history behind the way it has been established through physics.
This Lesson Also Includes:
1. Physical & Chemical Changes
2. States of Matter & Phase Changes in Matter
3. Molecular Movements in a Solid, Liquid, & Vapor
Hetrogenous and homogenous mixture ppt

Heterogeneous and Homogeneous Mixtures are discussed in this presentation. High School chemistry, physical science, environmental science, earth systems, and material science students will benefit from this presentation. All essential introductory concepts are presented here.
Physical States Of Matter

Basics of solid, liquid and gas including info about gas and temperature relationship for 8th grade science - based on McDougal Littell series
More Related Content
What's hot
Force and its effects

Force and its effects for class 8. this ppt provides an easy way to understand force and the effects of force by giving various daily life examples. It consists of animations through which students can easily understand.
Mixtures

A mixture forms when two or more substances are combined such that each substance retains its own chemical identity. Everywhere around us are made up of mixtures. We can see them in nature, along the surface of the earth, in the oceans and in the foods we eat. There are infinite numbers of mixtures that can be combined into homogeneous or heterogeneous.
States of Matter and changes of state

States of Matter for highschool, solid, liquid, gas, freezing point, melting point, characteristic properties, physical properties, chemical properties.
Matter, Properties, & Phases

Lesson teaches students about Matter (grades 6-8) & talks about the atomic models & the history behind the way it has been established through physics.
This Lesson Also Includes:
1. Physical & Chemical Changes
2. States of Matter & Phase Changes in Matter
3. Molecular Movements in a Solid, Liquid, & Vapor
Hetrogenous and homogenous mixture ppt

Heterogeneous and Homogeneous Mixtures are discussed in this presentation. High School chemistry, physical science, environmental science, earth systems, and material science students will benefit from this presentation. All essential introductory concepts are presented here.
What's hot (20)
Viewers also liked
Physical States Of Matter

Basics of solid, liquid and gas including info about gas and temperature relationship for 8th grade science - based on McDougal Littell series
Matter (states of) grade 5 (teach)

Teaches matter and the three states of matter at about the 5th grade level.
Viewers also liked (7)
Similar to States of matter
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign: Make a prediction relating heat to the movement of molecules. How does this relate to fluorescent lighting? Gas Liquid
Solution
generally the ,
Solids - vibrational (and rotational); moving very slowly.
Liquids - vibrational, rotational, and translational; moving quickly.
Gases - vibrational, rotational, and translational; moving very quickly.
Explanation:
Particles are always moving. No matter what. There is a certain point where they move so slowly it is considered minuscule, but they are in fact moving.
Thus, in solids, liquids, and gases, you can expect particles to move in a certain trend. That trend is average kinetic energy of the particles and the forces holding them together (attraction).
Solids
When you have little to no heat, the particles are moving very slowly, so slowly that they atomic attractions result in bonds that are difficult to break without adding energy. At this stage, this is known as a solid.
In a solid, the particles are vibrating. Only some solids have the ability to rotate. They cannot translate regardless.
Liquids
When you add energy (heat) to a solid, you \"excite\" the particles so much that they break their bond; their attraction is too weak to hold them together - yet strong enough to keep a form
As a result, you get a liquid.
In a liquid, the particles are not only vibrating (their bonds are not present but they still vibrate), but they are also rotating and translating. The particles are moving much faster than in a solid.
Gases
When you add even more energy to the substance, you increase the kinetic energy of those particles so much, that they lose their state form, becoming a gas.
In a gas, particles have vibrational, rotational, and translational motion, allowing them to \"bounce\" off of one another. Particles in a gas state move much faster than a liquid.
Light is a form of energy that can be released by an atom. It is made up of many small particle-like packets that have energy and momentum but no mass. These particles, called light photons, are the most basic units of light.
When the atom gains or loses energy, the change is expressed by the movement of electrons. When something passes energy on to an atom -- heat , for example -- an electron may be temporarily boosted to a higher orbital (farther away from the nucleus). The electron only holds this position for a tiny fraction of a second; almost immediately, it is drawn back toward the nucleus, to its original orbital. As it returns to its original orbital, the electron releases the extra energy in the form of a photon, in some cases a light photon
.
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

Anything which occupies space or volume, has mass and can be perceived by our senses is called matter.
385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt

bjbsjnskamaskjdfkmnfdkkqw.lejdkdkncdjckdsjwj
Similar to States of matter (20)
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign- Make a prediction relating heat to the movement of molecules.docx
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

ICSE Class VIII Chemistry Matter Around Us - TopperLearning
385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt

385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt
More from amandayoung313
More from amandayoung313 (20)
States of matter
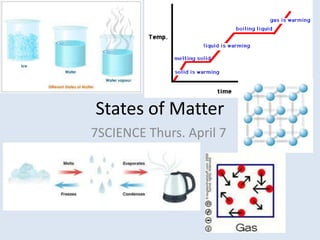
- 1. States of Matter 7SCIENCE Thurs. April 7
- 2. Review: Kinetic Theory of Matter What is the kinetic theory of matter? All particles in matter are in constant, random motion What determines state of matter (solid, liquid or gas) ? Arrangement Movement (motion) of atoms
- 3. Kinetic Theory of Matter These energy produced by these moving particles is called kinetic energy The higher the temperature, the faster the particles are moving and more likely they will collide into other particles When particles collide, the faster-moving particles (hotter) transfer some energy to the slower-moving particles (cooler)
- 4. States of Matter: SOLID The lower the temperature, the slower the particles are moving (less energy) Solids: The particles have little motion, and are shaking in a specific arrangement/shape
- 5. States of Matter: Melting/Freezing Points If temperature ↑, the particles will shake faster and at a certain temperature they will break free of their shape This temperature is called the melting point solid liquid When the opposite happens (liquid solid) this temperature is called the freezing point
- 6. States of Matter: LIQUID In a liquid the particles are free to move and take the shape of its container Ex. Pour a glass of water: Forces hold the particles together as a liquid while gravity holds them at the bottom of the glass
- 7. State of Matter: GAS If temperature in the liquid increases, the particles begin to shake faster and escape the arrangement and forces of the liquid This temperature is called the boiling point and is called vaporization Particles are free to move around inany direction or shape