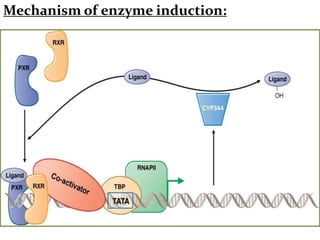
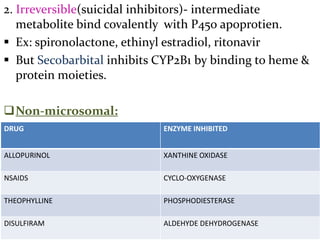
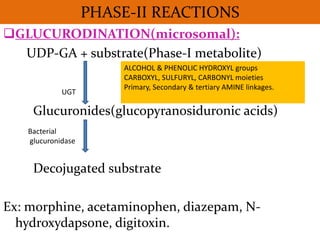
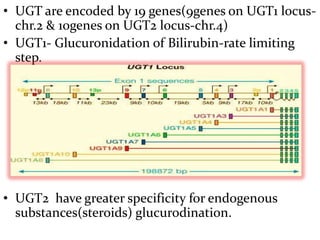
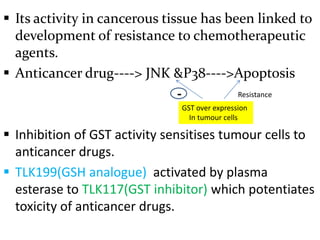
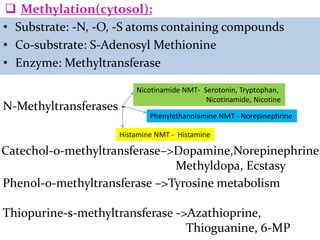
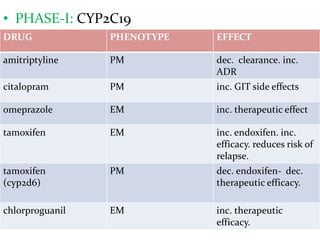
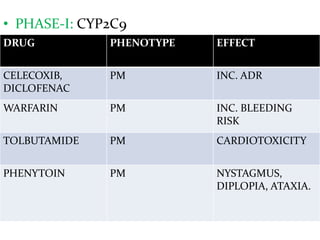
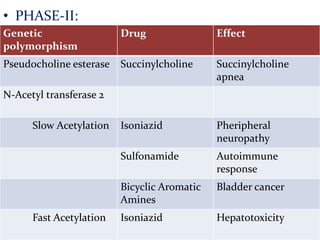
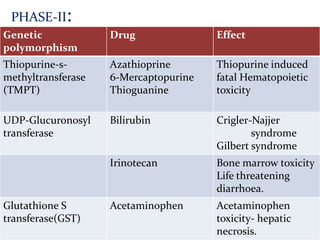
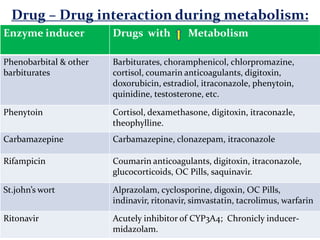
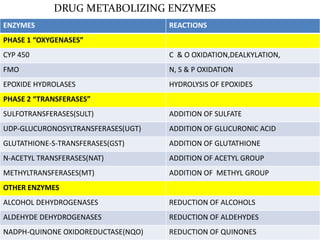
This document provides an overview of drug metabolism and biotransformation. It defines biotransformation as the biochemical alteration of drugs or xenobiotics by enzymes. The liver is identified as the major site of biotransformation. Drug metabolism occurs in two phases - phase I involves reactions like oxidation, reduction and hydrolysis. Phase II involves conjugating reactions. Factors like enzyme induction and inhibition can influence the extent of drug metabolism. Cytochrome P450 enzymes and phase I and II enzymes involved in biotransformation are also discussed.
















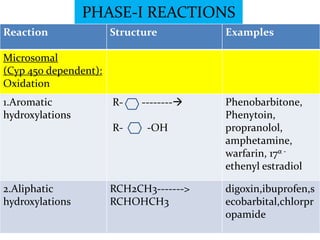
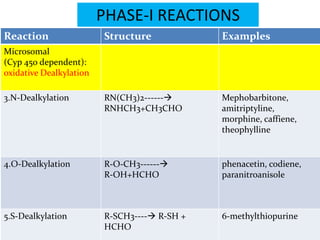
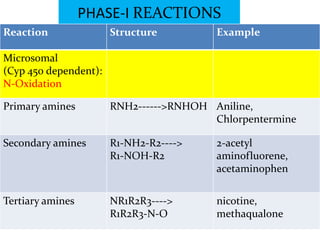
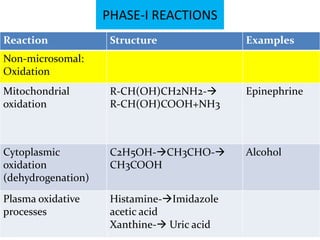
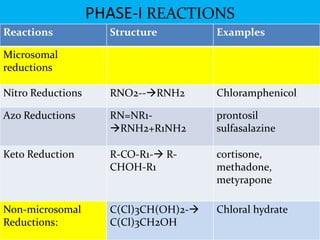
![PHASE-I REACTIONS
Reaction Structure Examples
Microsomal(cyp 450
dependent):
S-Oxidation R1-S-R2---> cimetidine,
R1 -SO-R2 chlorpromazine,
omeprazole, thioridazine
Deamination R-CHNH2-R----> Amphetamine, diazepam
R-COR +NH3
Desulfurisation R1-PS-R2---> Parathion,
R1-PO-R2 thiopental
Dechlorination CCl4---> [CCl3-]---> CHCl3 carbontetrachloride](https://image.slidesharecdn.com/vijay-biotransformation-121124223500-phpapp01/85/Vijay-biotransformation-25-320.jpg)