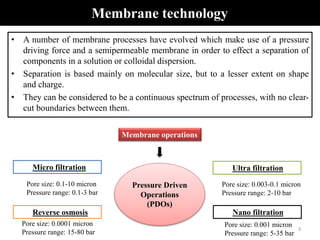
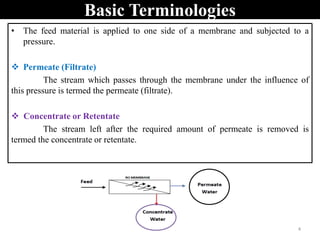
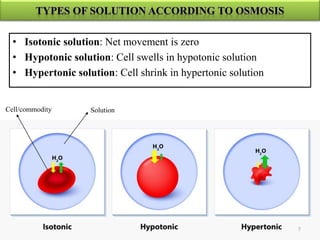
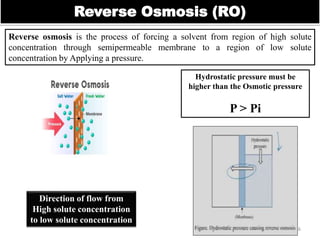
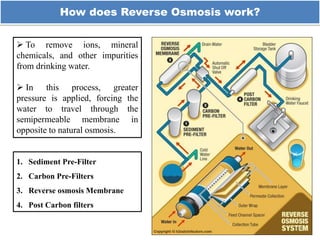
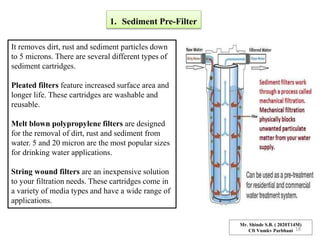
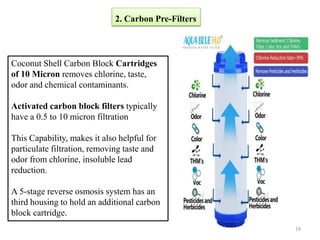
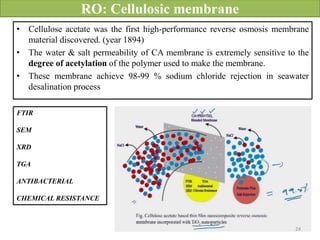
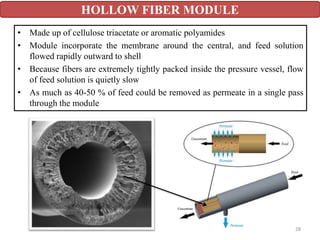
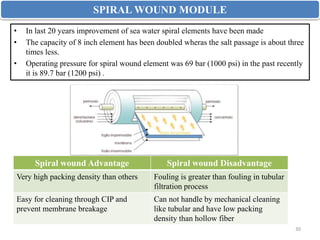
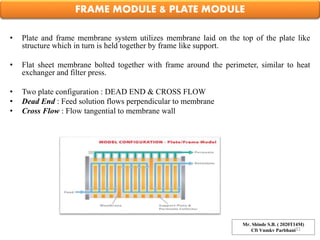
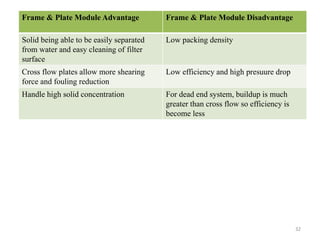
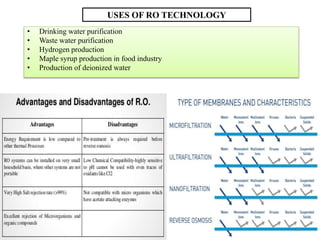
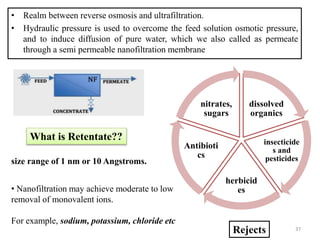
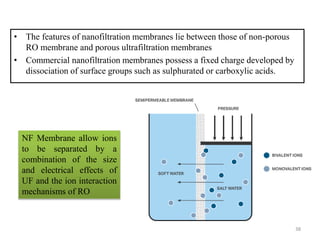
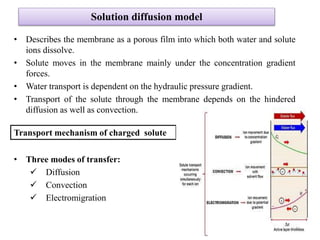
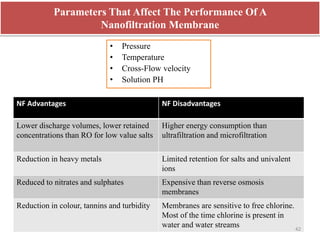
The document discusses membrane technology, specifically focusing on reverse osmosis (RO) and nanofiltration (NF) processes used in food engineering and water purification. It details the principles of osmosis, osmotic pressure, and the operational parameters of various membrane types, including their advantages and applications in industries such as wastewater treatment and drinking water purification. Additionally, the document explores the theoretical framework for membrane function, key processing parameters, and comparisons between different membrane configurations.