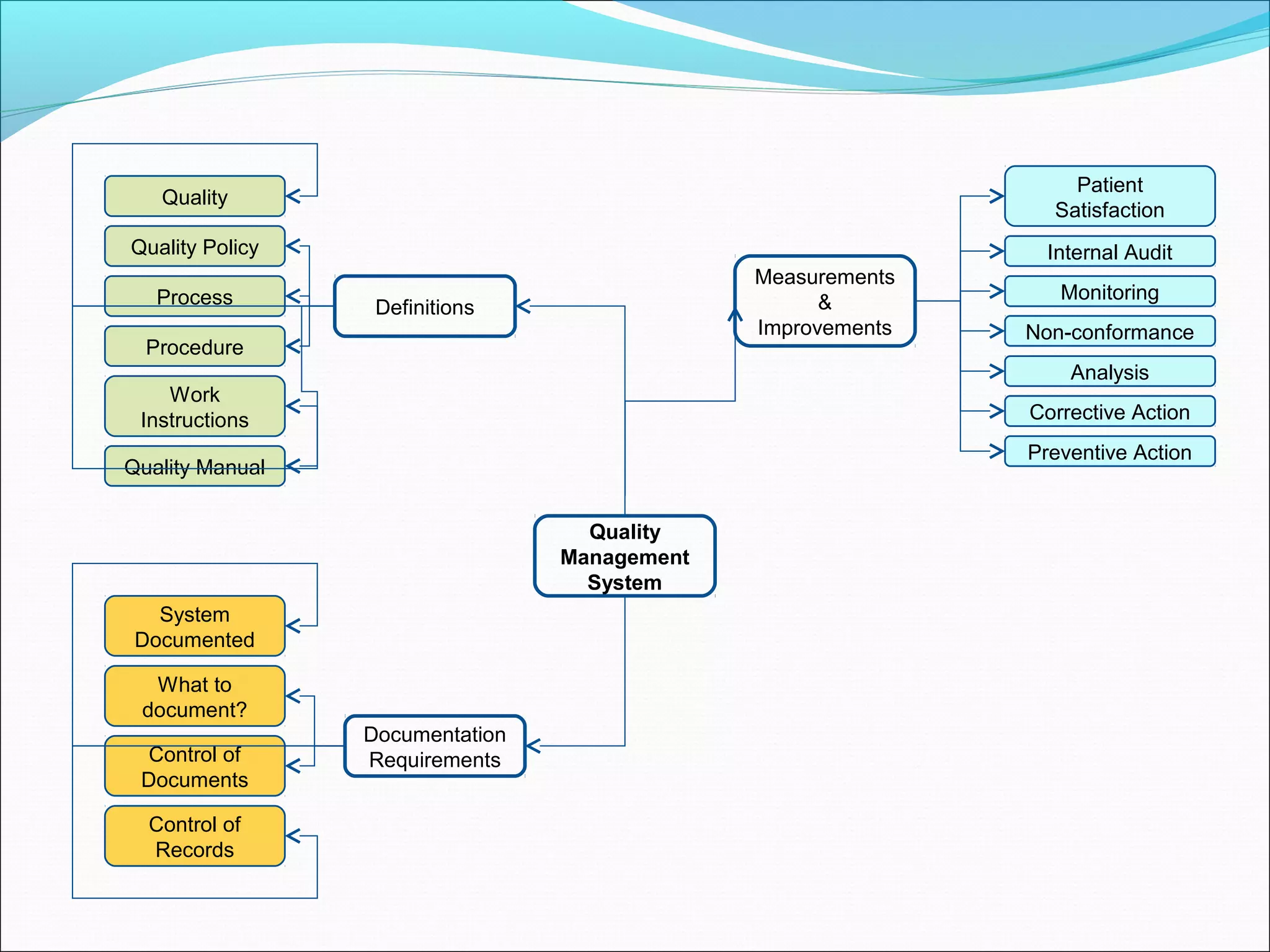
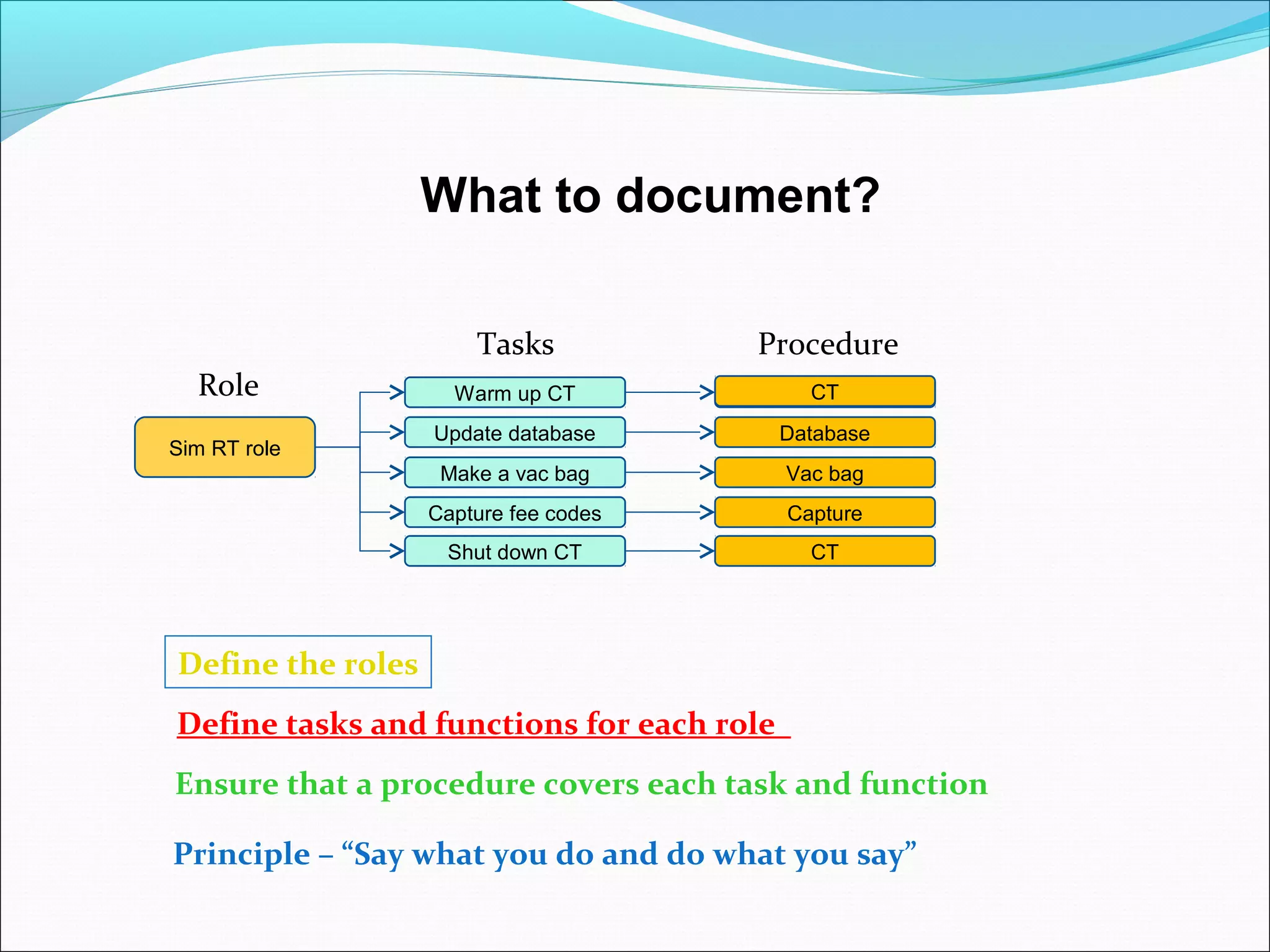
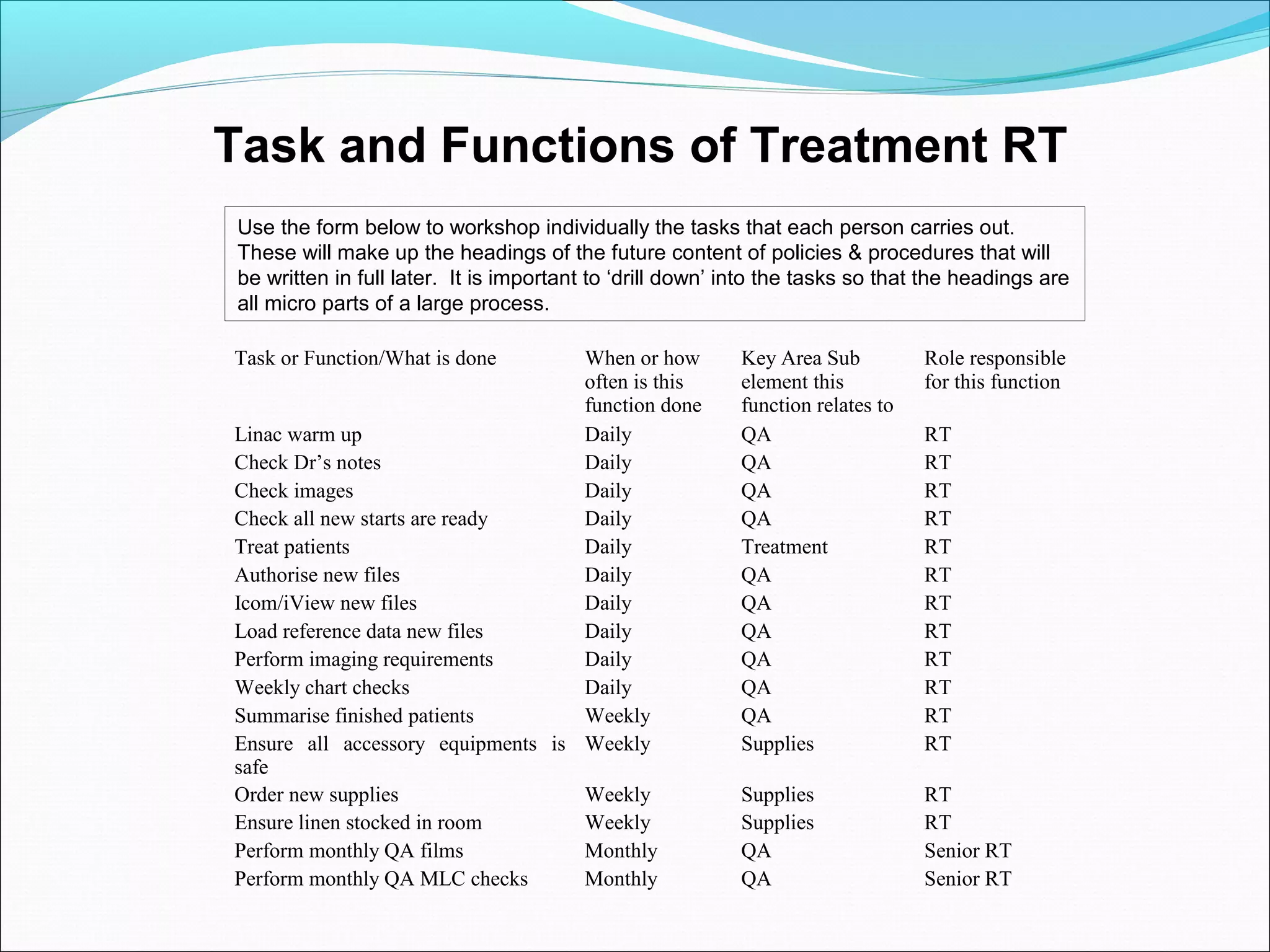
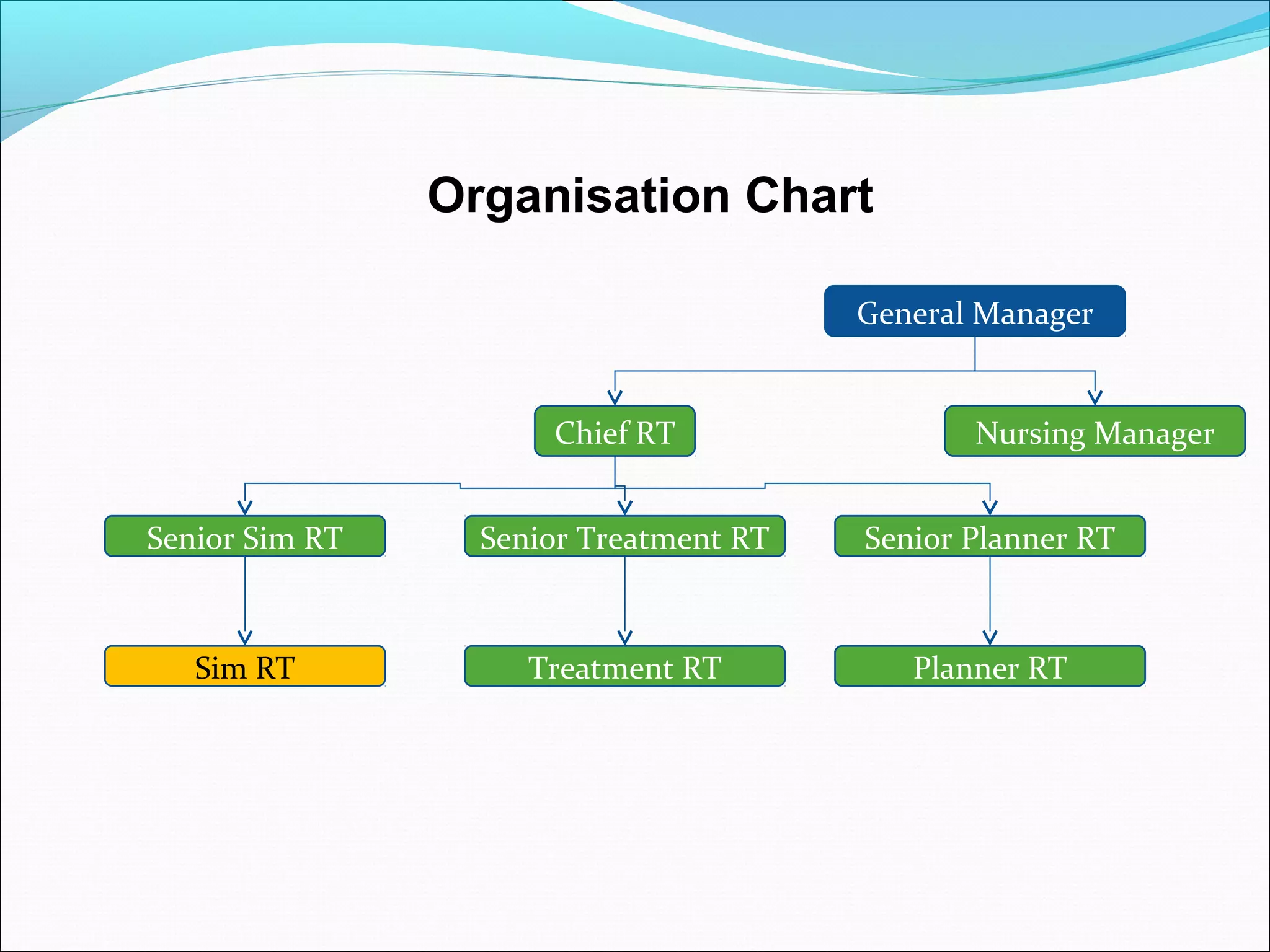
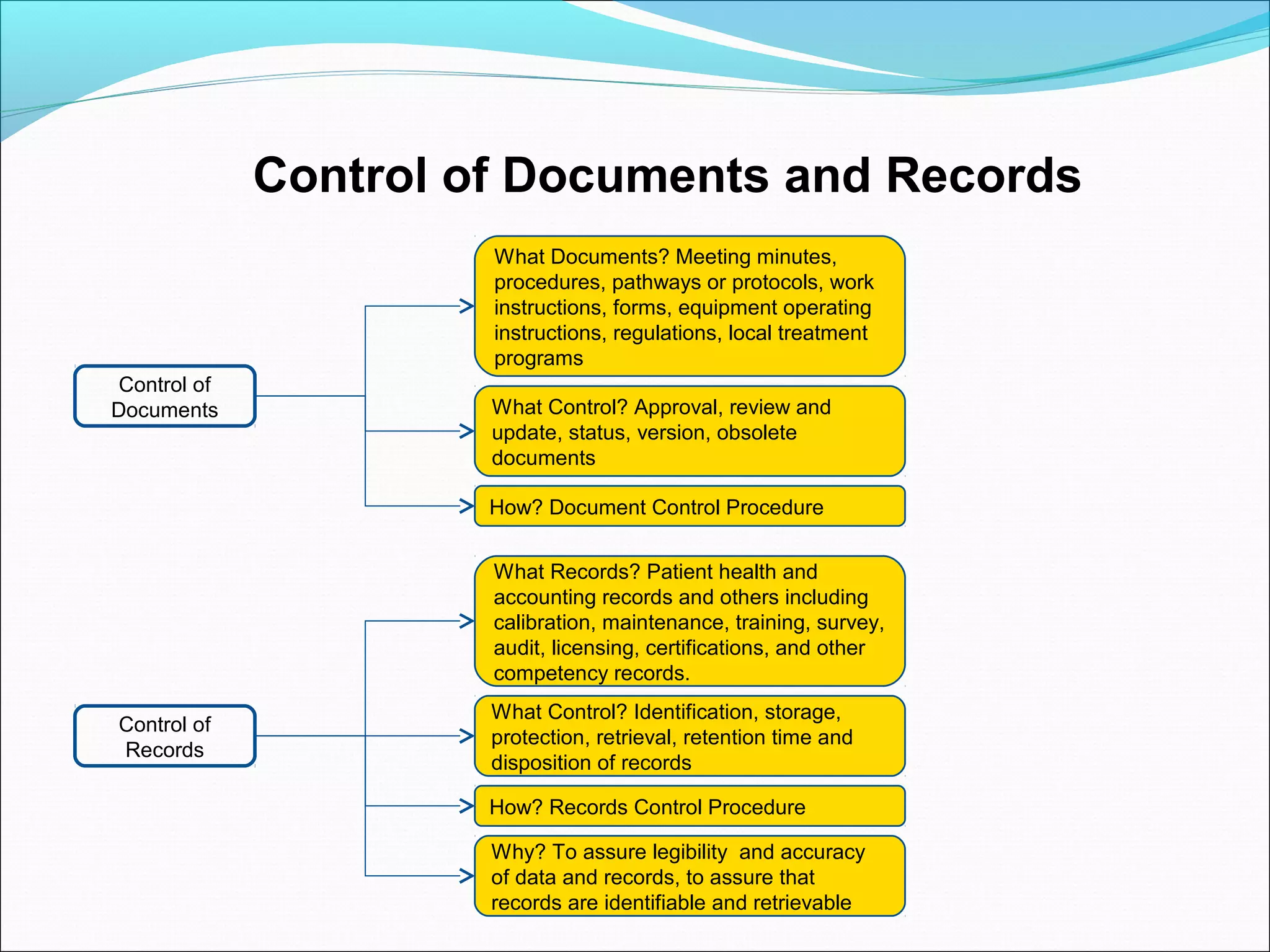
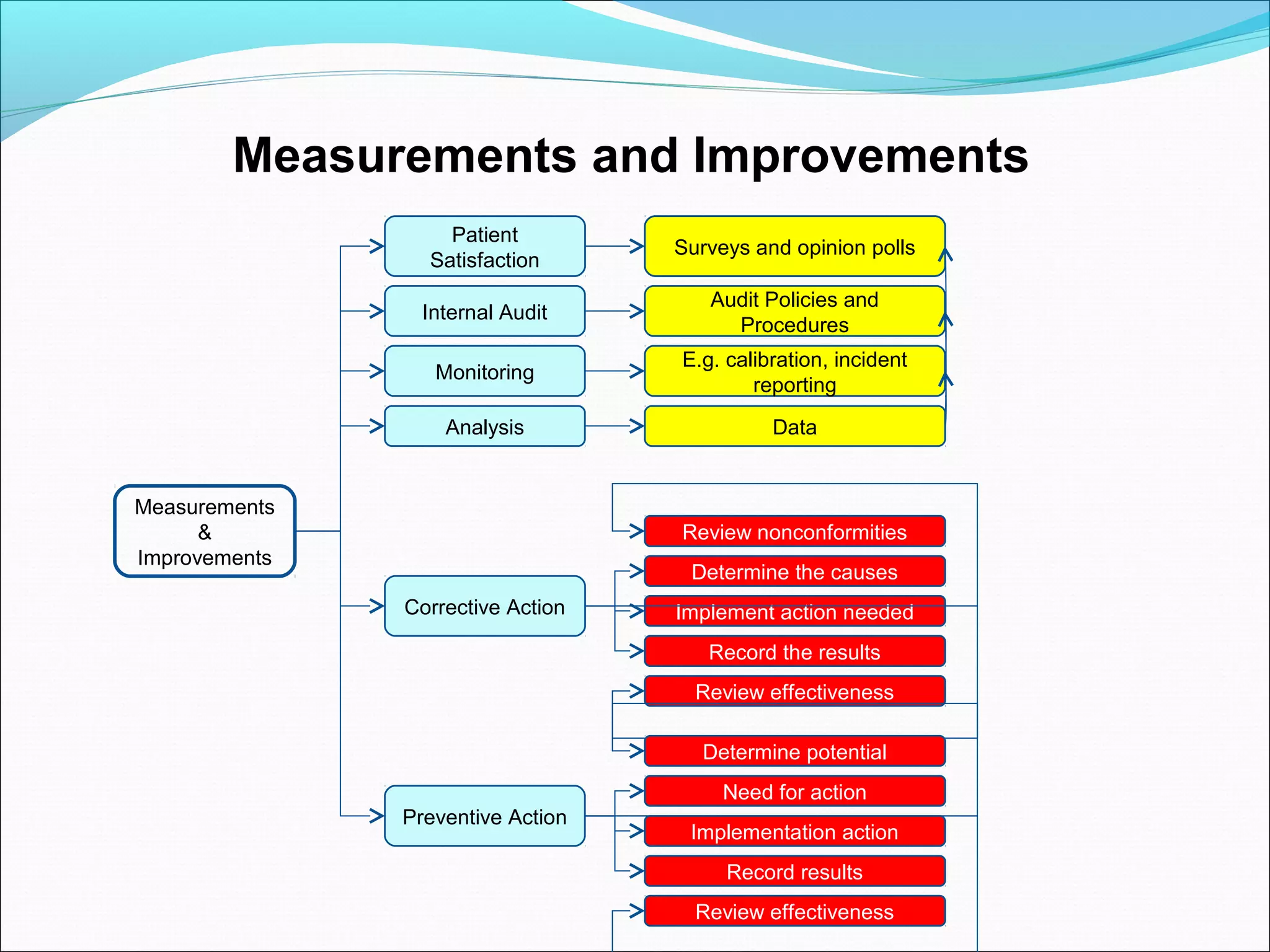
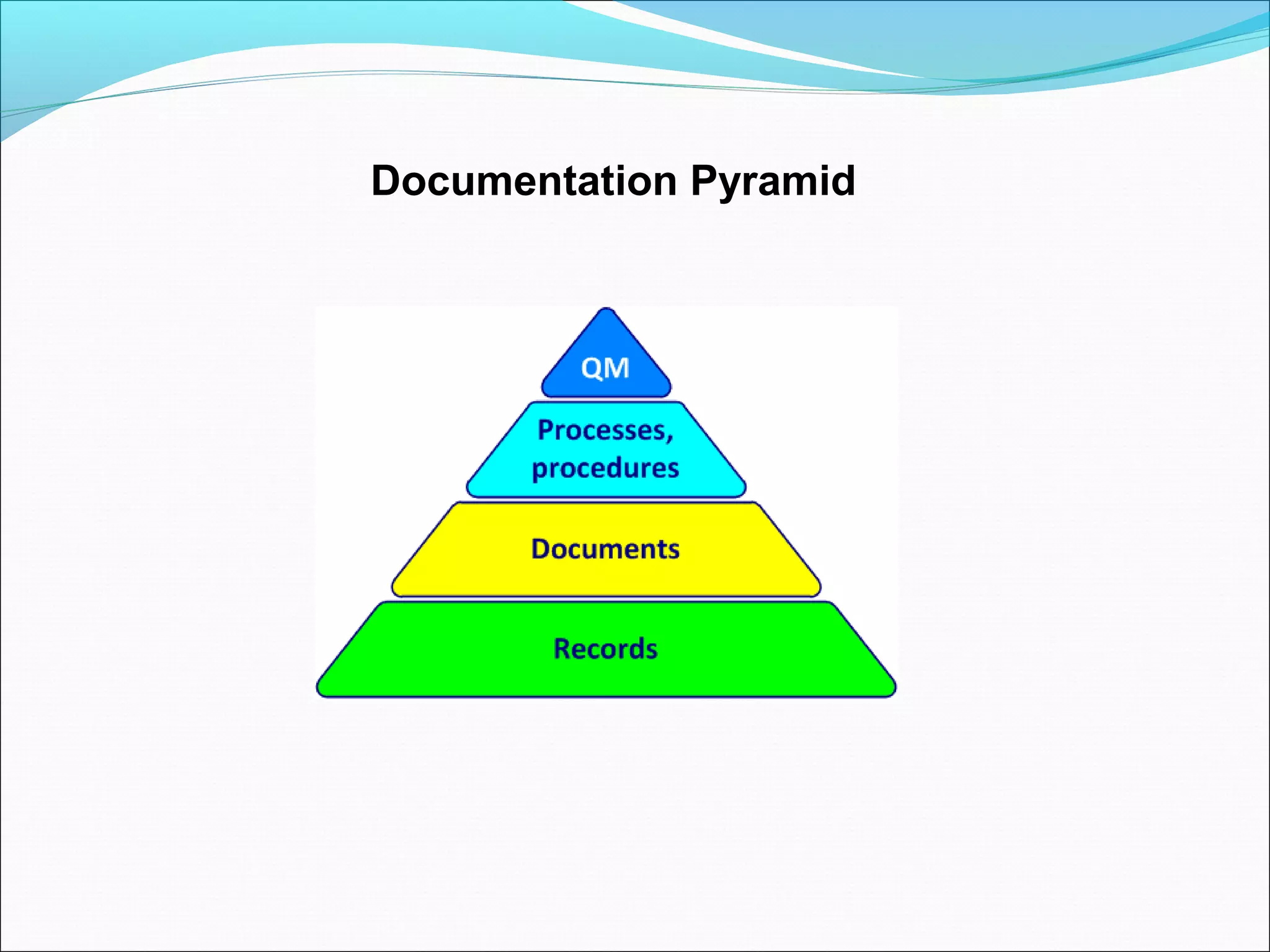
Quality management systems (QMS) based on ISO 9001 principles will be mandatory for all hospitals and day surgeries in Western Australia starting January 1, 2013. The document outlines the importance of documentation, defining processes, and roles within the QMS to ensure consistency, patient satisfaction, and compliance with national standards. It emphasizes that without proper documentation, procedural effectiveness cannot be assured, thereby necessitating a comprehensive and documented approach to quality management.