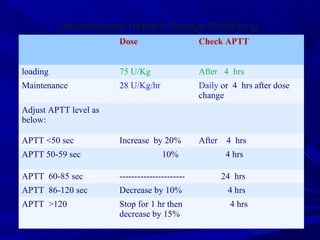
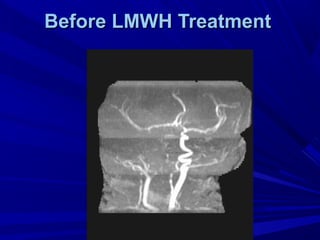
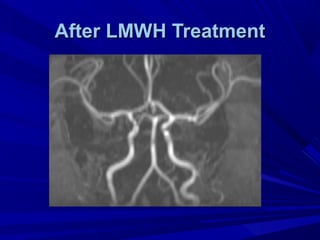
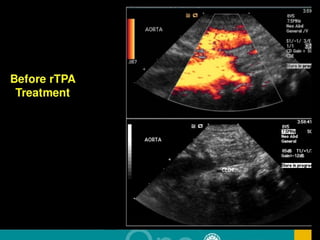
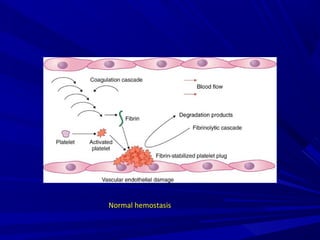
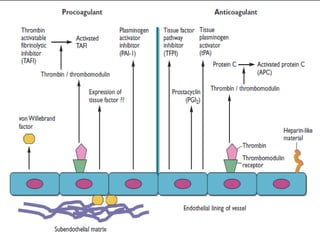
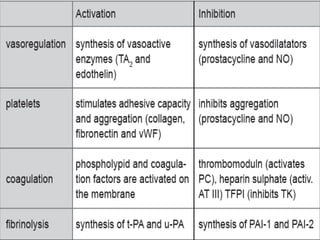
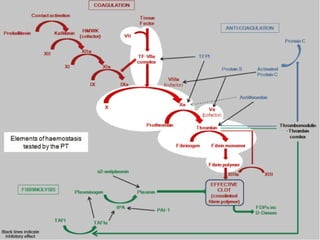
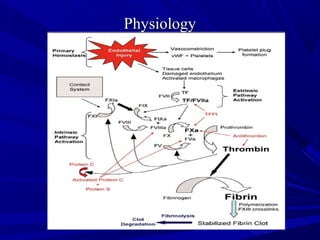
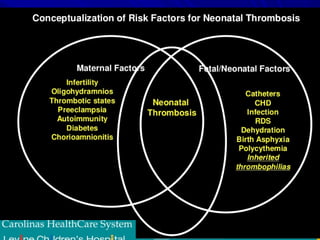
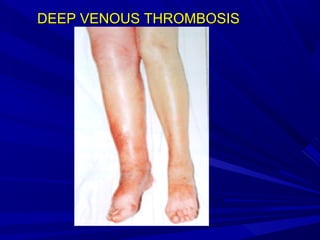
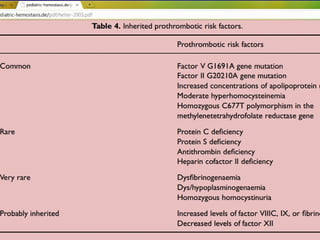
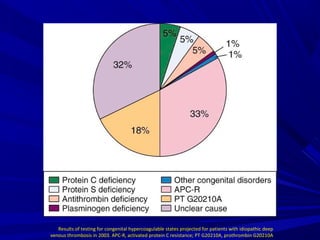
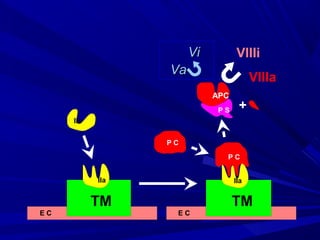
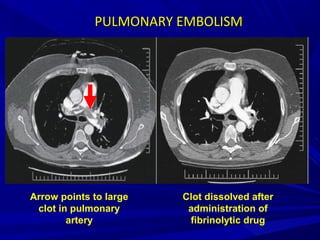
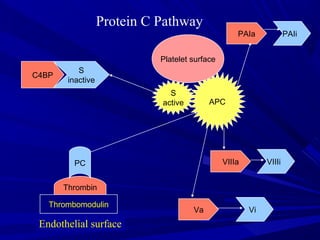
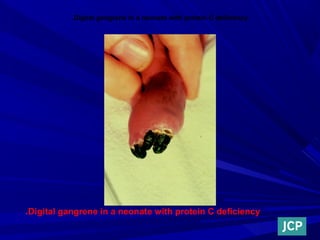
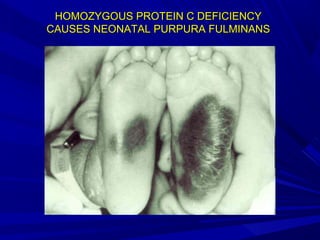
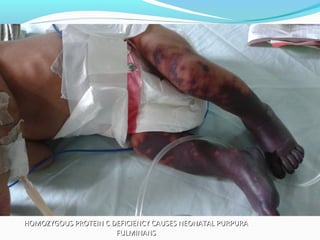
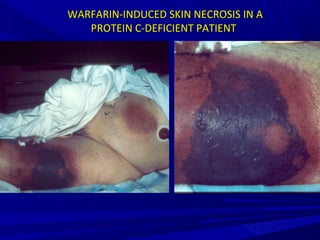
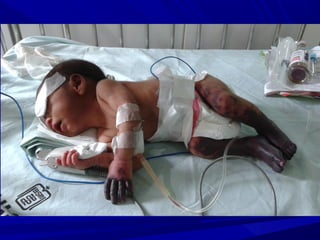
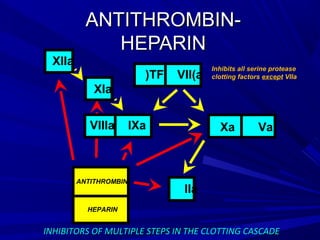
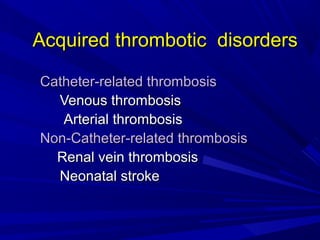
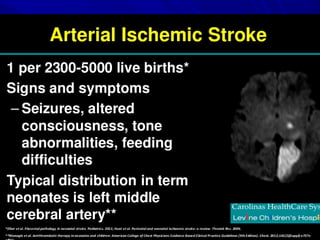
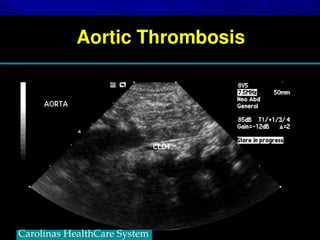
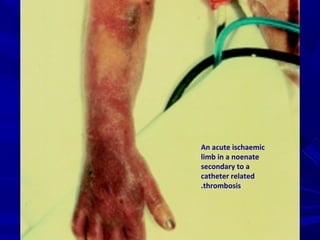
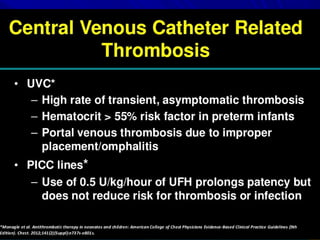
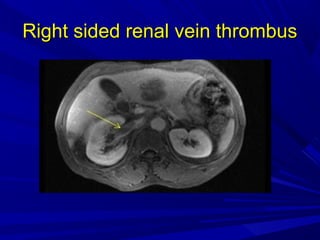
This document discusses neonatal thrombophilia and procoagulant disorders. It begins by defining thrombophilia as a clinical tendency to thrombosis or molecular abnormalities that predispose to thromboembolic disease. It then discusses various inherited and acquired hypercoagulable states including factor V Leiden mutation, prothrombin gene mutation, protein C deficiency, protein S deficiency, and antithrombin deficiency. It notes that thrombophilia in neonates is a significant problem often associated with predisposing disorders and triggers like sepsis.

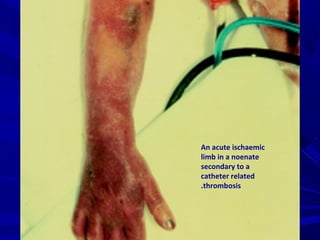




















































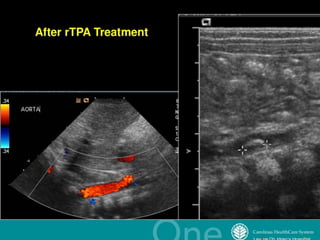
![Choice of therapyChoice of therapy
**MassiveMassive venous thrombi or arterialvenous thrombi or arterial
thrombi:thrombi:
ThrombolysisThrombolysis
SurgerySurgery
[[NBNB- Oral anticoagulant drugs – not- Oral anticoagulant drugs – not
recommnaded for neonate]recommnaded for neonate]](https://image.slidesharecdn.com/procoagulantdisorders-151025151651-lva1-app6892/85/Procoagulant-disorders-in-neonates-Updated-88-320.jpg)