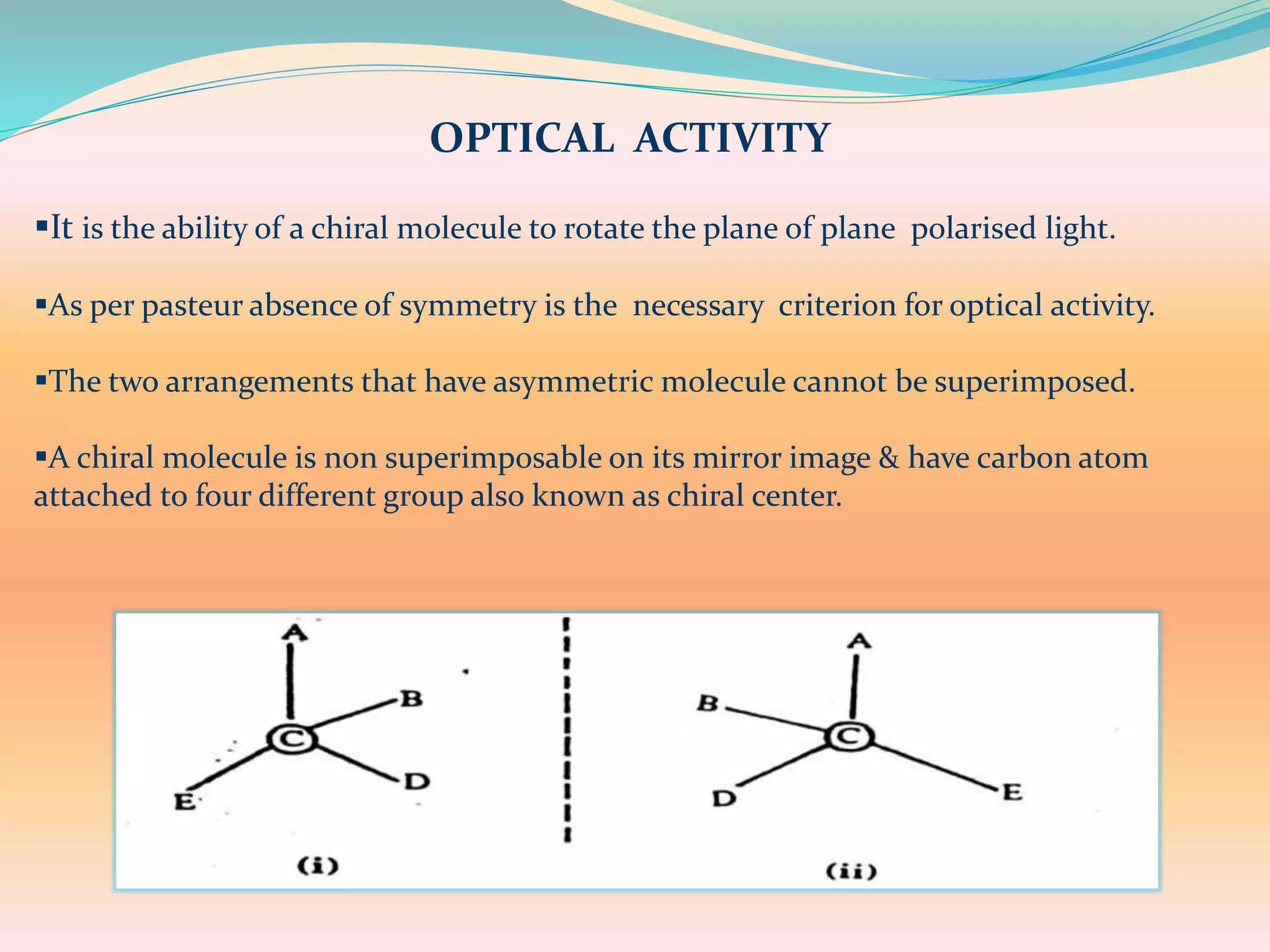
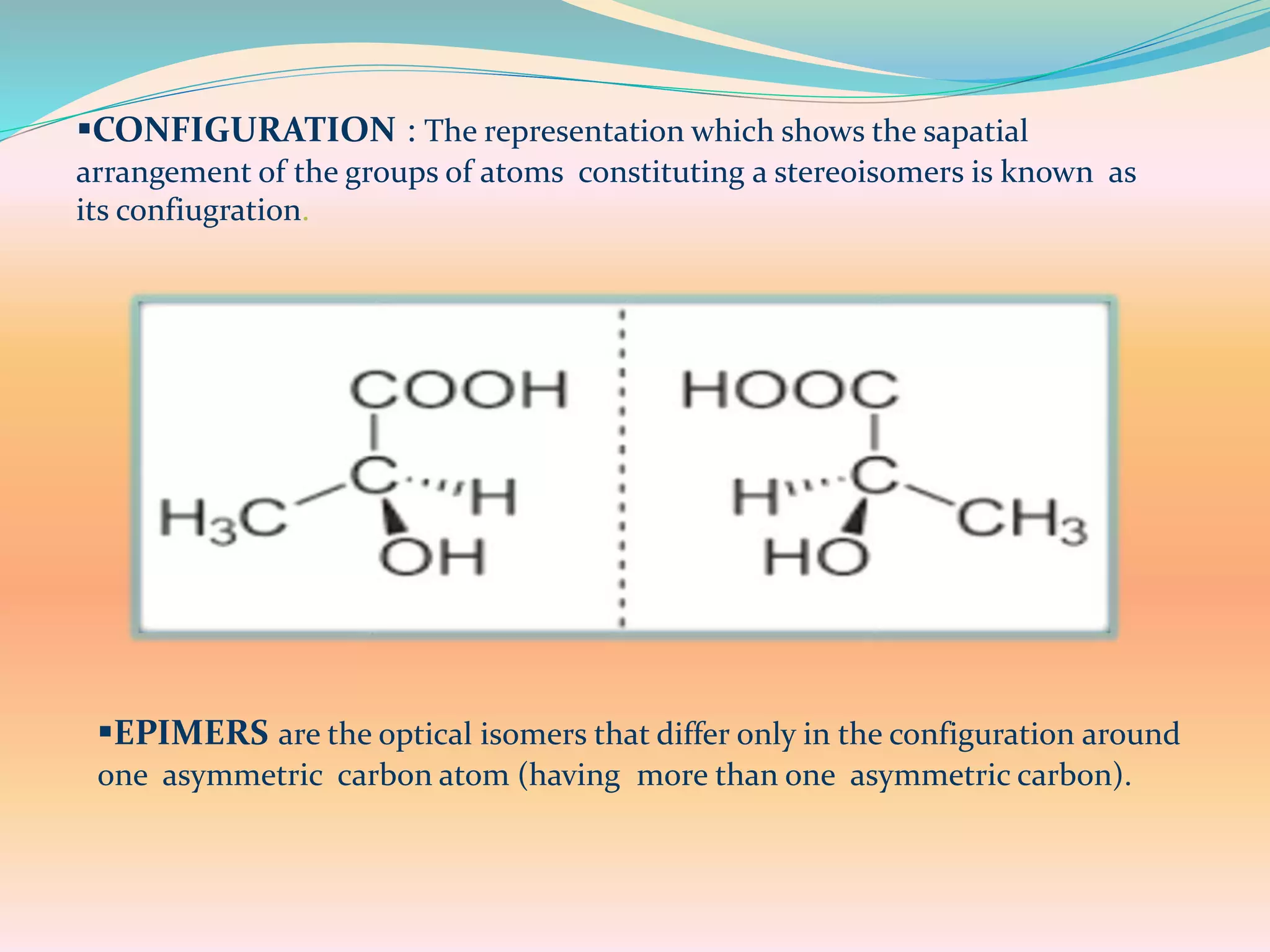
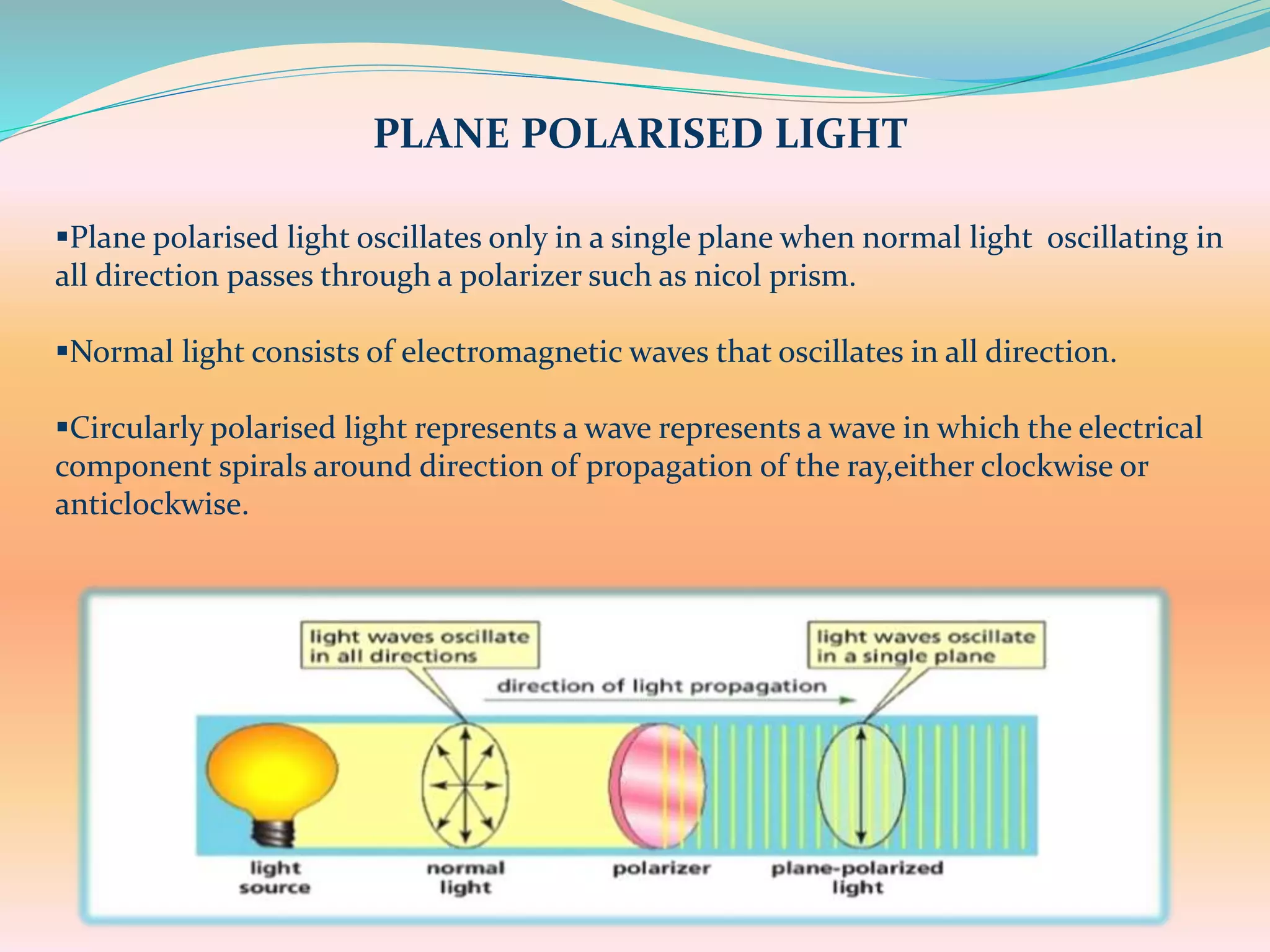
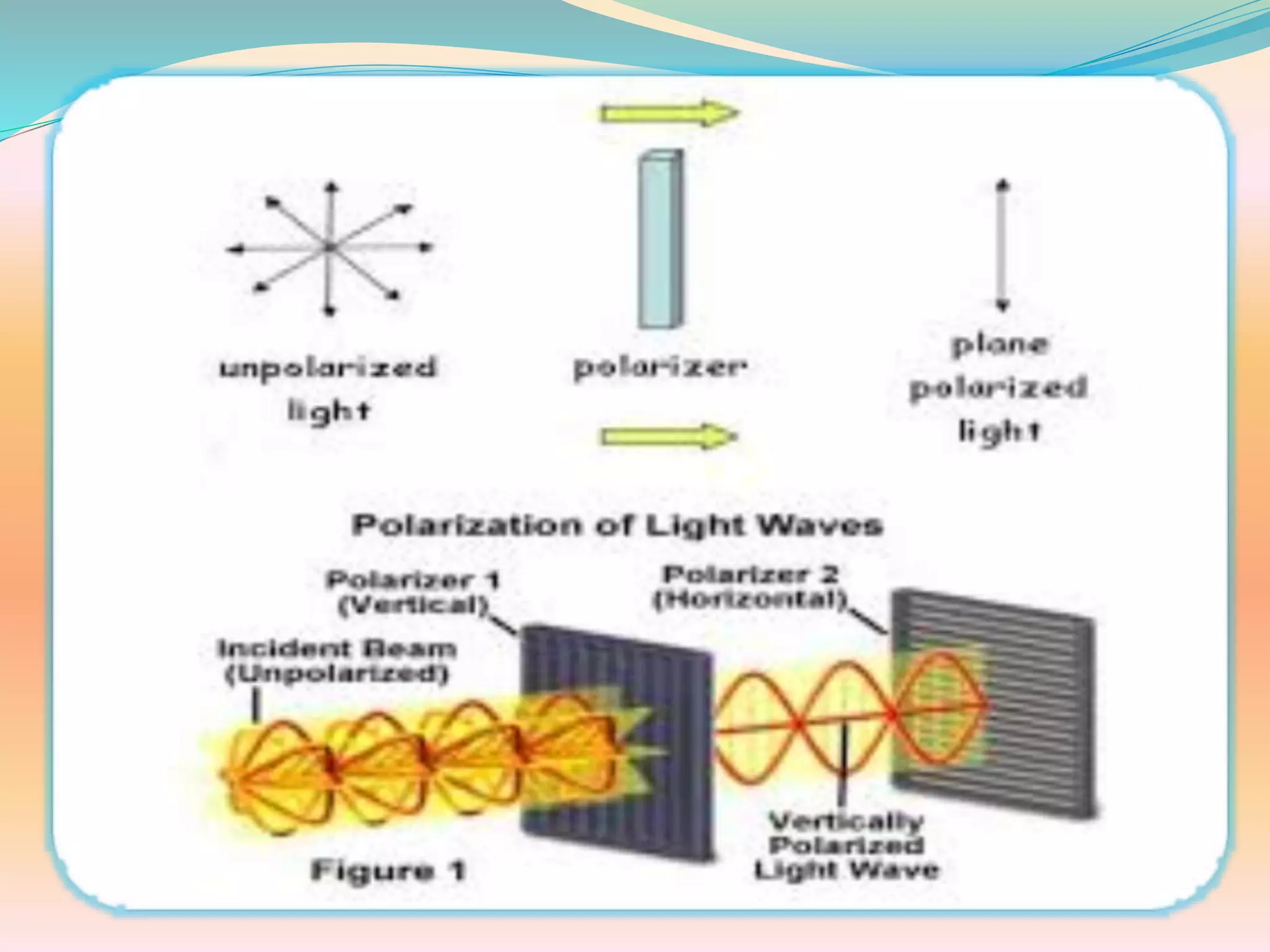
The document discusses polarimetry, the study of the rotation of polarized light by optically active substances, and its applications in qualitative and quantitative analysis. It covers various concepts such as optical activity, chiral molecules, and the functioning and construction of polarimeters. Different types of polarimeters and their applications, including measuring specific rotation and differentiating optical substances, are also described.