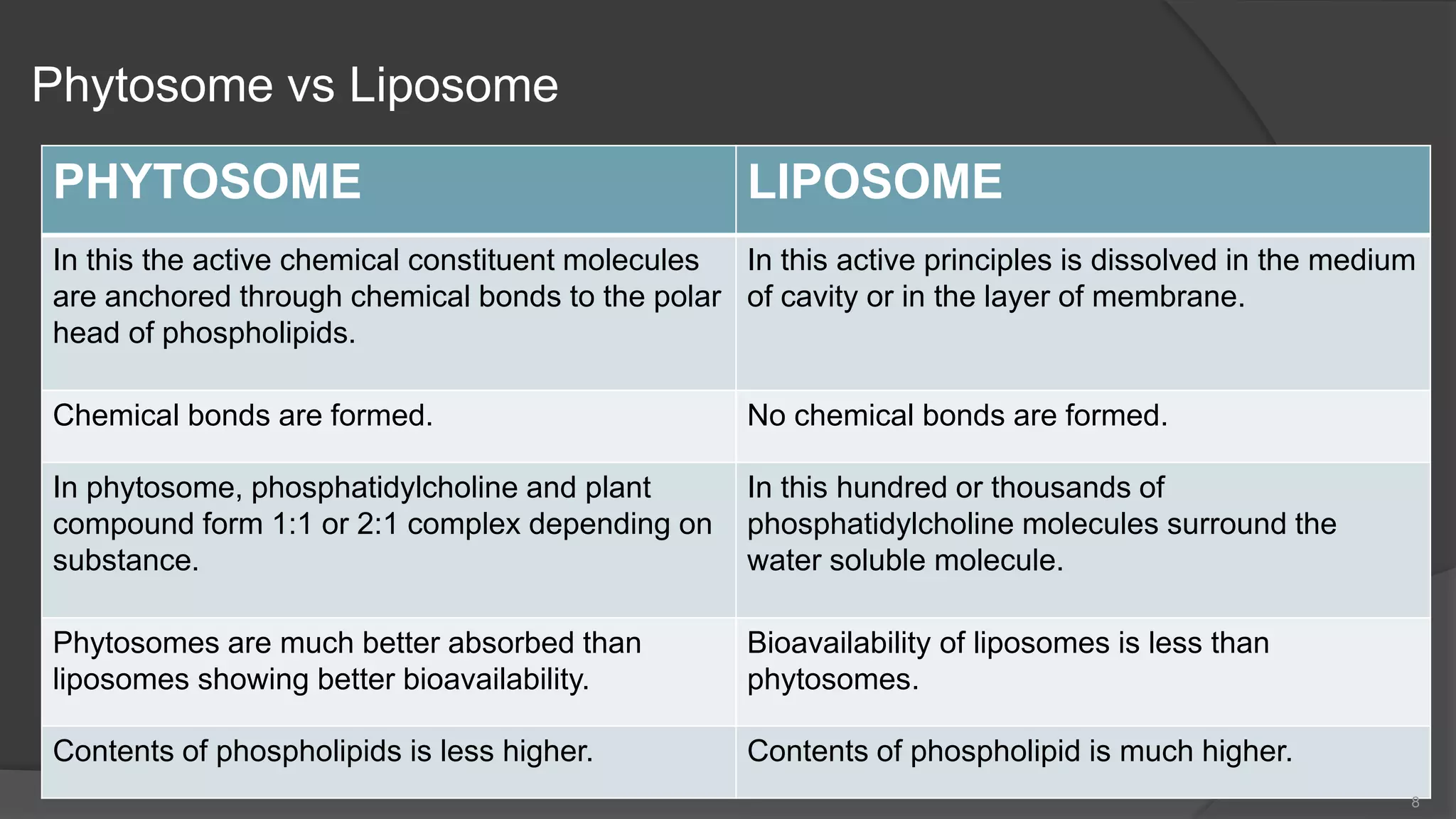
This document discusses phytosomes, which are herbal extracts bound to phospholipids. Phytosomes have several advantages over traditional herbal extracts, including enhanced absorption and bioavailability. The document outlines the structure and properties of phytosomes, comparing them to liposomes. It also describes the preparation process, evaluation methods, and applications of various phytosome formulations. Common phytosomes include silymarin (milk thistle) for liver health, grape seed for antioxidants, green tea for antioxidants and chronic diseases, and curcumin for anti-inflammatory effects. Phytosomes allow herbal constituents to be absorbed more effectively and produce better results than conventional herbal extracts.