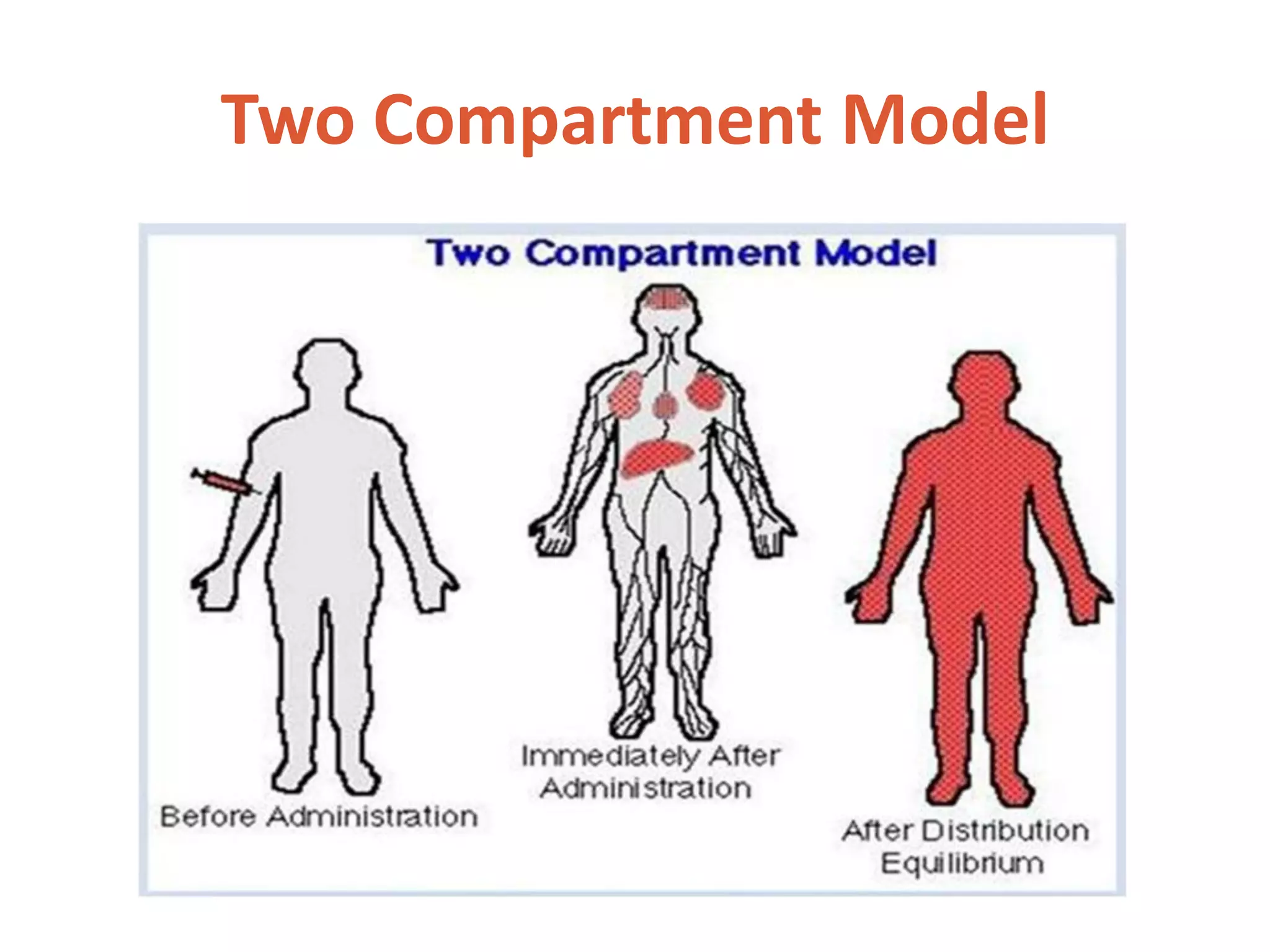
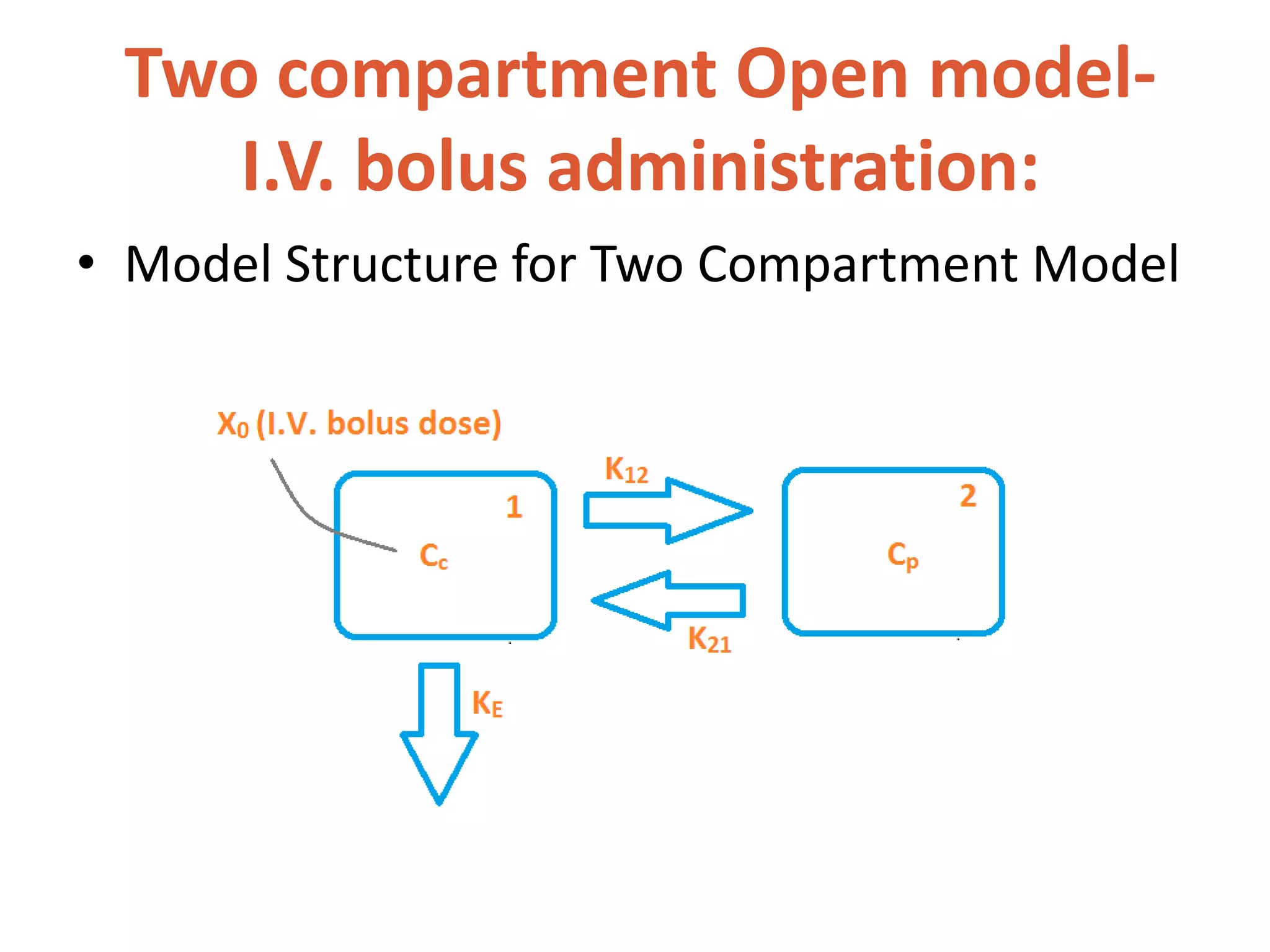
This document discusses multicompartment models, including two-compartment models. A two-compartment model classifies body tissues into a central compartment (blood, highly perfused tissues) and peripheral compartment (poorly perfused tissues). Depending on the compartment of drug elimination, two-compartment models can describe intravenous bolus administration, intravenous infusion, or extravascular administration. Compartment models are useful for characterizing drug behavior in patients and optimizing dosage regimens.