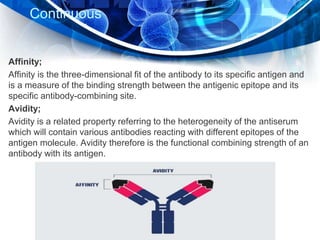
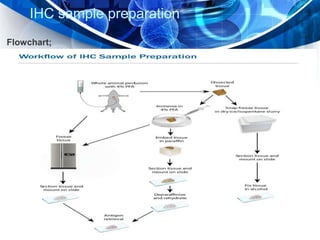
Immunohistochemistry is a technique that combines immunology and histology to detect antigens in tissues using the antibody-antigen interaction. It allows determination of antigen expression and location within tissues at the cellular level. The process involves using labeled antibodies that bind specifically to antigens in tissue sections or cell samples. This binding is then visualized through enzymatic reactions or use of fluorescent dyes, colloidal gold, or radioactive labels. Immunohistochemistry is widely used in research and clinical diagnosis and has various applications including disease diagnosis, prognosis, and therapeutic monitoring.