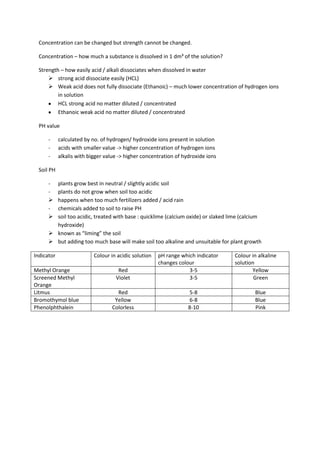
The document describes different types of acids and bases. It defines acids as substances that produce hydrogen ions (H+) in aqueous solution, and bases as substances that produce hydroxide ions (OH-). Examples of common acids include sulfuric acid, nitric acid, hydrochloric acid and ethanoic acid. Examples of bases are metal oxides and metal hydroxides. The document also discusses the reactions of acids and bases, including metal-acid reactions, and the uses of some common acids and bases.