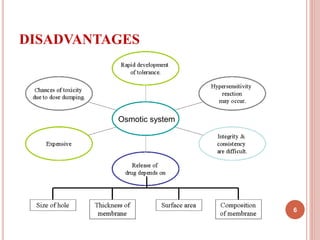
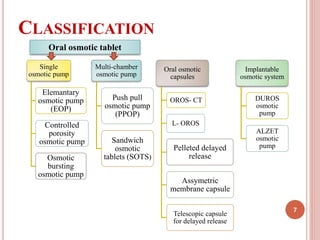
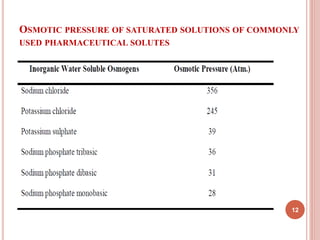
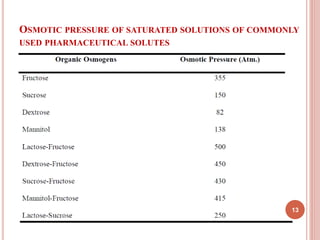
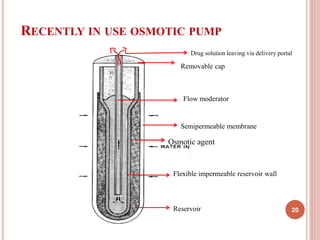
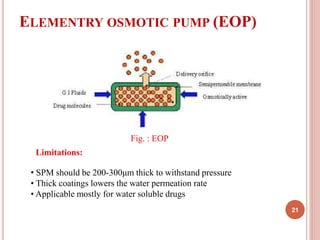
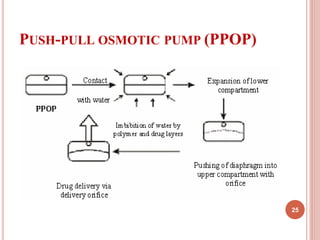
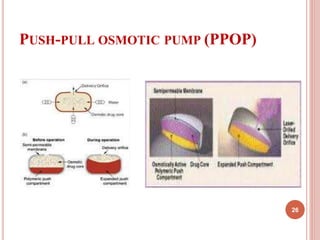
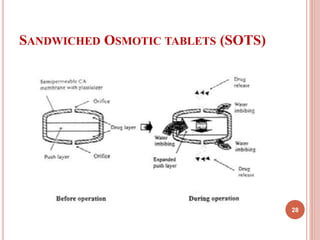
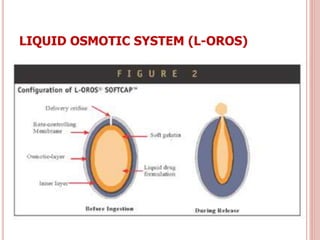
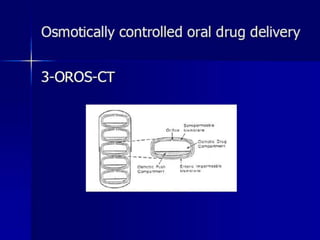
The document discusses osmotic drug delivery systems (ODDS). It begins by defining osmosis and describing how osmotic pressure drives the movement of water across a semi-permeable membrane. It then discusses the advantages of ODDS such as zero-order delivery kinetics and independence from gastric pH. The document classifies several types of ODDS including elementary osmotic pumps, controlled porosity pumps, and push-pull pumps. It also describes components like the semi-permeable membrane and osmotic agents. In vitro evaluation methods and factors affecting ODDS performance are briefly covered.